Clear Sky Science · en
Müller glial and microglial responses coupled to recovery of cone photoreceptors following limited cone ablation in zebrafish retina
Why Healing Fish Eyes Matter
The back of the eye, called the retina, turns light into sight. In people, once those light-sensing cells are badly damaged, they rarely grow back, leading to permanent vision loss. Zebrafish are different: they can naturally repair parts of their retinas. This study looks at what happens when only a small fraction of the zebrafish’s color-sensing cells are briefly stressed or lost, revealing how support cells and immune cells spring into action to help the retina recover. Understanding this choreography could one day inspire new ways to protect or repair human vision.
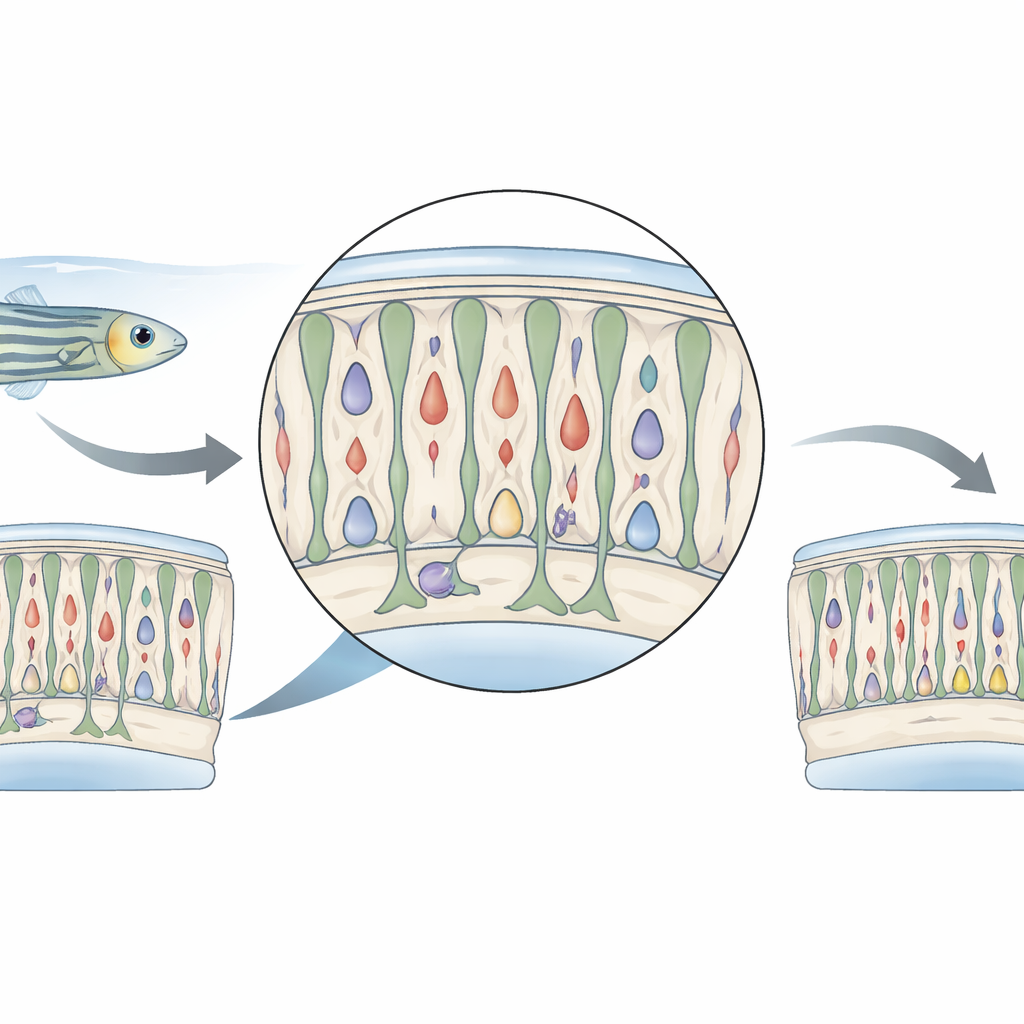
A Gentle Nudge to the Eye
The researchers worked with genetically engineered zebrafish whose cone photoreceptors—the cells that detect color and daylight—carry an enzyme that can turn a harmless drug into a toxin, but only inside those cones. Adult fish were soaked for 24 hours in the drug metronidazole, then returned to clean water. Instead of wiping out large numbers of cones, this treatment killed only a small subset. Most cones survived but showed signs of temporary damage: their appearance changed, their fluorescent signal weakened, and a key cone gene, gnat2, was turned down. The fish themselves behaved as if their vision was dulled right after treatment, but these problems faded within a few days as the retina began to recover.
Color Cells Under Stress
To see what was happening inside individual cells, the team used single-cell RNA sequencing, which reads out which genes are switched on in thousands of single cells at once. They focused on two to five days after drug removal, a window when cones seemed to be bouncing back rather than dying. Surviving cones dialed down many of the genes that handle light detection and signal processing, suggesting they were temporarily “powering down” their visual machinery. At the same time, they turned up genes linked to dealing with harmful oxygen by-products and maintaining protein quality. This pattern points to a stress-response mode in which cones concentrate on damage control and restoration rather than on peak visual performance.
Support Cells Enter Repair Mode
The retina’s main support cells, called Müller glia, responded strongly even though only limited cone death was detected. These glial cells stretch from the bottom to the top of the retina, directly touching the light-sensing cells, and are known in zebrafish to act as a source of new neurons after injury. In this study, Müller glia ramped up genes typically linked to reactivity and tissue remodeling, and many of them re-entered the cell cycle, a sign that they began dividing. The authors observed new progenitor-like cells appearing in the middle layer of the retina, then moving toward the outer layer that houses the photoreceptors. Some of these repair responses were accompanied by switches in genes that are usually active in stem cells, indicating that Müller glia were partially reverting to a more flexible, regenerative state, even though the amount of outright cell loss was modest.
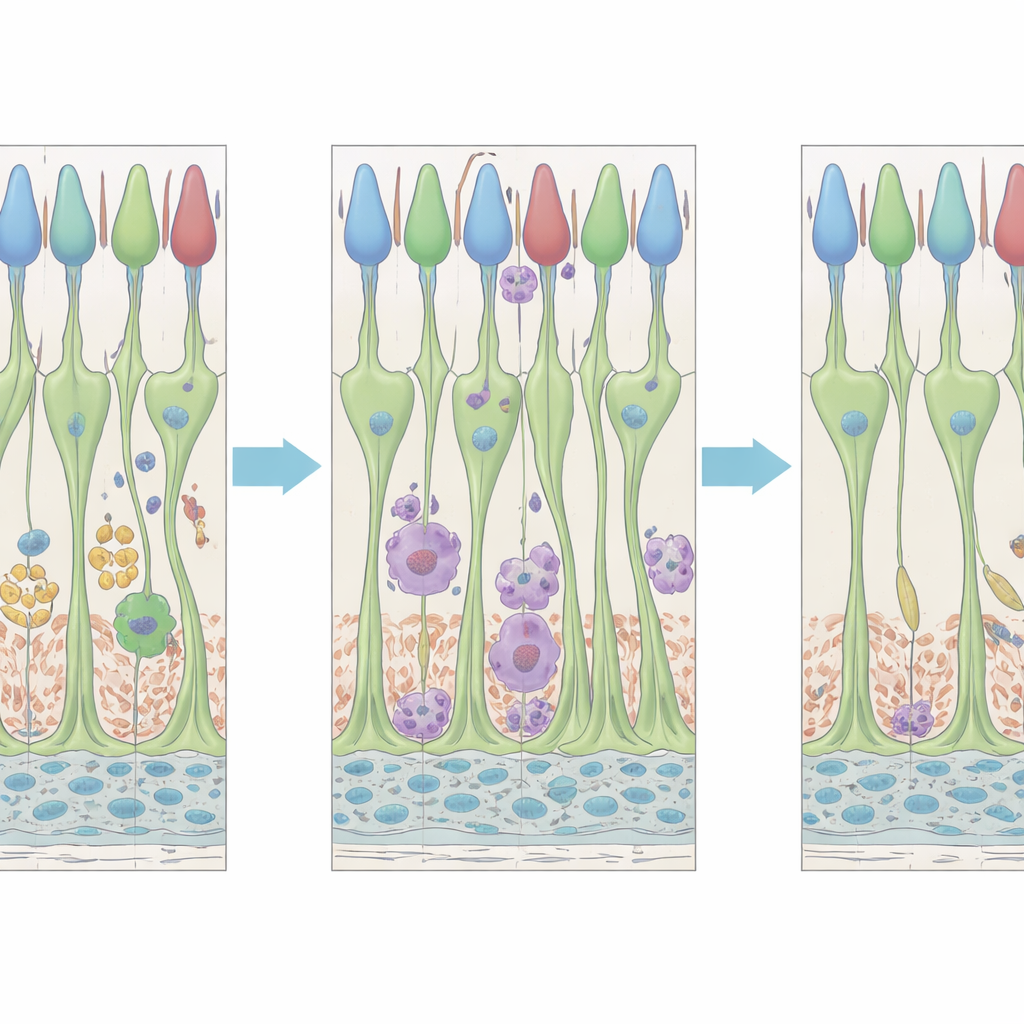
Immune Cells with Two Waves of Action
Resident immune cells in the retina, called microglia and other closely related myeloid cells, also showed a two-stage response. Within about two days after treatment, microglia changed shape, clustering around dying cones and likely helping to clear away debris. Later, around four to five days, a second wave of altered microglial behavior emerged: cells appeared near the outer segments of cones and in the space close to the pigmented layer behind the retina, at a time when cones were showing strong signs of recovery. Gene-expression patterns suggested that there are at least two distinct microglial-like subpopulations with different molecular signatures, hinting that some immune cells may specialize in cleaning up dying cells, while others may focus on supporting stressed but surviving neurons.
What This Means for Protecting Sight
This work shows that in zebrafish, even a limited and brief disturbance to color-sensing cells is enough to rally a coordinated response from both support cells and immune cells. Cones temporarily downshift their visual function while turning on internal repair programs. Müller glia become reactive, divide, and produce progenitor cells, and microglia respond in timed waves linked first to cell death and later to stress recovery. Together, these responses appear to help the retina restore structure and function after a mild insult. For human eye diseases that involve slow, chronic damage rather than sudden injury, understanding how zebrafish mobilize glia and immune cells in response to sublethal stress could point toward therapies that boost the eye’s own capacity to stabilize or even rebuild vulnerable photoreceptors.
Citation: Weimar, H.V., Farre, A.A., Rumford, J.E. et al. Müller glial and microglial responses coupled to recovery of cone photoreceptors following limited cone ablation in zebrafish retina. Sci Rep 16, 9058 (2026). https://doi.org/10.1038/s41598-026-40141-1
Keywords: zebrafish retina, cone photoreceptors, Müller glia, microglia, retinal regeneration