Clear Sky Science · en
Artificial Intelligence-based characterization of therapeutic response in fluid types and volumes influencing retinal function in neovascular age-related macular degeneration
Why this matters for everyday eyesight
As people age, one of the leading threats to central vision is a condition called age-related macular degeneration. In its "wet" form, leaky blood vessels in the back of the eye cause tiny pockets of fluid that blur sight. Modern injections can help, but not everyone responds the same way, and many patients still lose vision over time. This study explores whether a new, automated way of measuring these fluid pockets with artificial intelligence (AI) can reveal who benefits most from treatment and why timing matters.
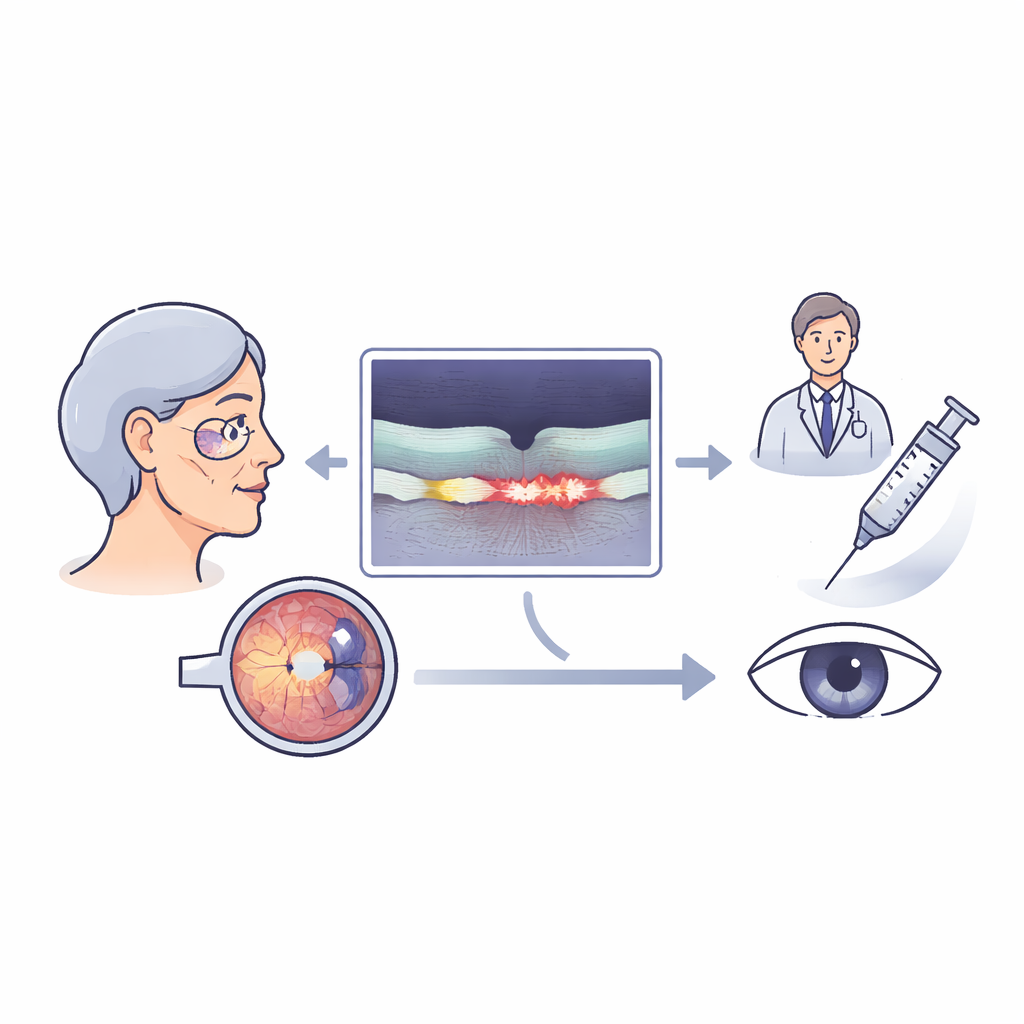
Seeing the problem inside the eye
The light-sensitive part of the eye, the retina, can swell when fragile new blood vessels leak. This leaking fluid can collect within the retina, just under it, or beneath a supportive cell layer. Doctors already look for these changes using a scan called optical coherence tomography, which creates cross-sectional images of the retina. Traditionally, they judge these scans by eye, deciding whether fluid is present or not. But this rough approach does not capture how much fluid is there or where exactly it sits, even though both the amount and location of fluid are closely tied to how well a person can see.
Letting computers measure the fluid
The researchers used an approved AI tool that can automatically trace and measure different fluid pockets in three key zones: within the retinal tissue, under the light-sensing cells, and in a pocket called a pigment epithelial detachment. They applied this tool to 285 eyes with active wet macular degeneration that were part of a larger clinical trial. Some eyes had never been treated before, while others had already received many anti-leakage injections over nearly two years in routine care. All eyes were scanned and tested for visual acuity at the start of the study and again one month after an injection.
Fresh versus previously treated eyes
At the outset, almost every eye had a raised pocket under the supporting cell layer, and most also had fluid under the retina; about half had fluid within the retinal tissue itself. Eyes that had never been treated before carried more fluid inside the retina but had slightly worse vision. After one injection, these treatment‑naive eyes gained on average about four to five letters on a standard eye chart, while previously treated eyes gained only about one letter. The biggest difference lay in the behavior of fluid within the retina: in newly treated eyes, the volume of this inner fluid dropped sharply, both in absolute terms and as a percentage of what was present at baseline. By contrast, fluid under the retina and in the raised pockets changed more modestly and to a similar degree in both groups.
What the fluid patterns reveal
When the team looked at fluid in a simpler, yes-or-no way, they saw that most pockets of inner fluid either stayed very small or dried up with treatment, especially in first‑time patients. Fluid under the retina and in raised pockets tended to linger, although it often shrank in size. These patterns support earlier work suggesting that inner retinal fluid is especially harmful to vision and more tightly linked to active, early disease, while some residual fluid under the retina may be less damaging and slower to clear. In previously treated eyes, lower starting levels of inner fluid and weaker response may reflect longer-standing damage, partial scarring, or a type of "wearing off" of responsiveness to the drug.
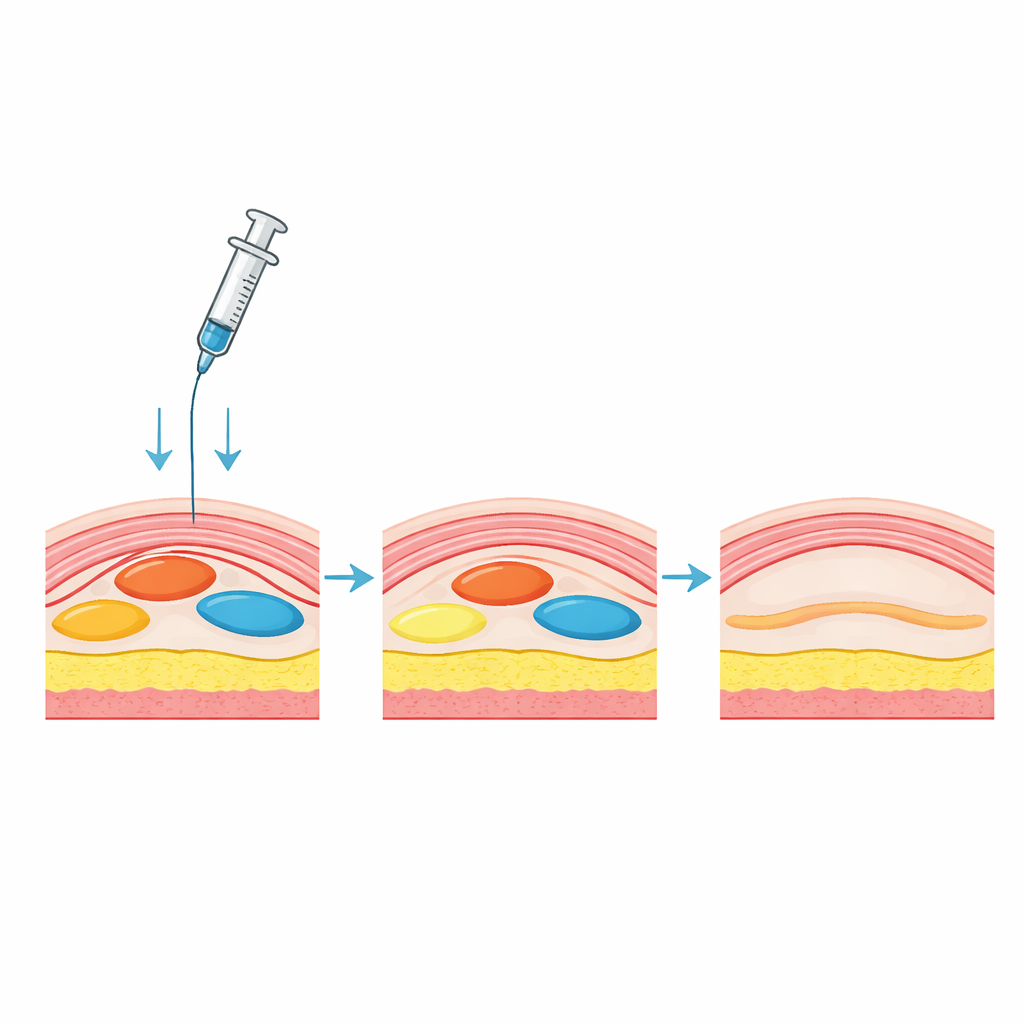
How this could change care
For a person living with wet macular degeneration, the findings underscore two practical messages. First, starting treatment early—when inner retinal fluid is still high and the disease is in a brisk, active phase—appears to bring the greatest short-term gains in vision. Second, precise, automated measurements of where and how much fluid is present can help doctors move beyond a simple "wet or dry" judgment. AI tools like the one used here can provide quick, objective fluid maps that may guide more personalized decisions about dosing intervals, drug choice, and when it is safe to tolerate small, stable pockets of fluid. While this study focused on the first month of therapy and cannot yet answer long-term questions, it points toward a future in which eye injections are steered not just by what doctors see, but by detailed, computer‑derived measures that capture the true dynamics of disease.
Citation: Frank-Publig, S., Buehl, W., Mares, V. et al. Artificial Intelligence-based characterization of therapeutic response in fluid types and volumes influencing retinal function in neovascular age-related macular degeneration. Sci Rep 16, 9466 (2026). https://doi.org/10.1038/s41598-026-40138-w
Keywords: age-related macular degeneration, retinal fluid, optical coherence tomography, anti-VEGF therapy, artificial intelligence imaging