Clear Sky Science · en
A Flexible Hybrid Quantum-classical Training Framework of Organ-at-Risk and Tumor Segmentation Models for Radiation Therapy Planning
Why smarter scans matter for cancer care
When doctors plan radiation treatments, they must target tumors precisely while sparing nearby healthy organs. Today this careful outlining is often done by hand and aided by artificial intelligence, but standard AI tools can be bloated and fragile: they need huge amounts of labeled data to work well, something hospitals rarely have. This study introduces a new way to train medical imaging models that borrows ideas from quantum computing to make them leaner, more accurate, and better suited to real-world cancer clinics.
The challenge of drawing a clean boundary
In radiation therapy, computers first need to separate “organs at risk” and tumors from surrounding tissue on CT scans. Modern deep learning models can do this automatically, yet they typically contain tens of millions of tunable numbers. With only a few hundred carefully annotated cases available, these oversized models tend to memorize quirks in the training data instead of learning robust anatomy. Existing tricks to shrink models either cut away parts of the network or restrict how its internal weights can vary. Both approaches save memory but often dull the model’s ability to capture the messy, irregular shapes of real tumors.
Borrowing power from quantum circuits
The authors propose a different strategy: instead of directly training every weight in the network, they let compact quantum-inspired modules generate many of those weights. These modules, called variational quantum circuits, are small systems that are tuned using ordinary optimization methods but can produce large collections of parameters from a much smaller set of learned values. In the hybrid quantum–classical training framework, or HQC-TF, some layers in a standard segmentation model are trained in the usual way, while others receive their weights from these quantum-style generators. Crucially, each image channel is paired with its own generator, preserving the independence of features that is essential for clear pattern extraction.
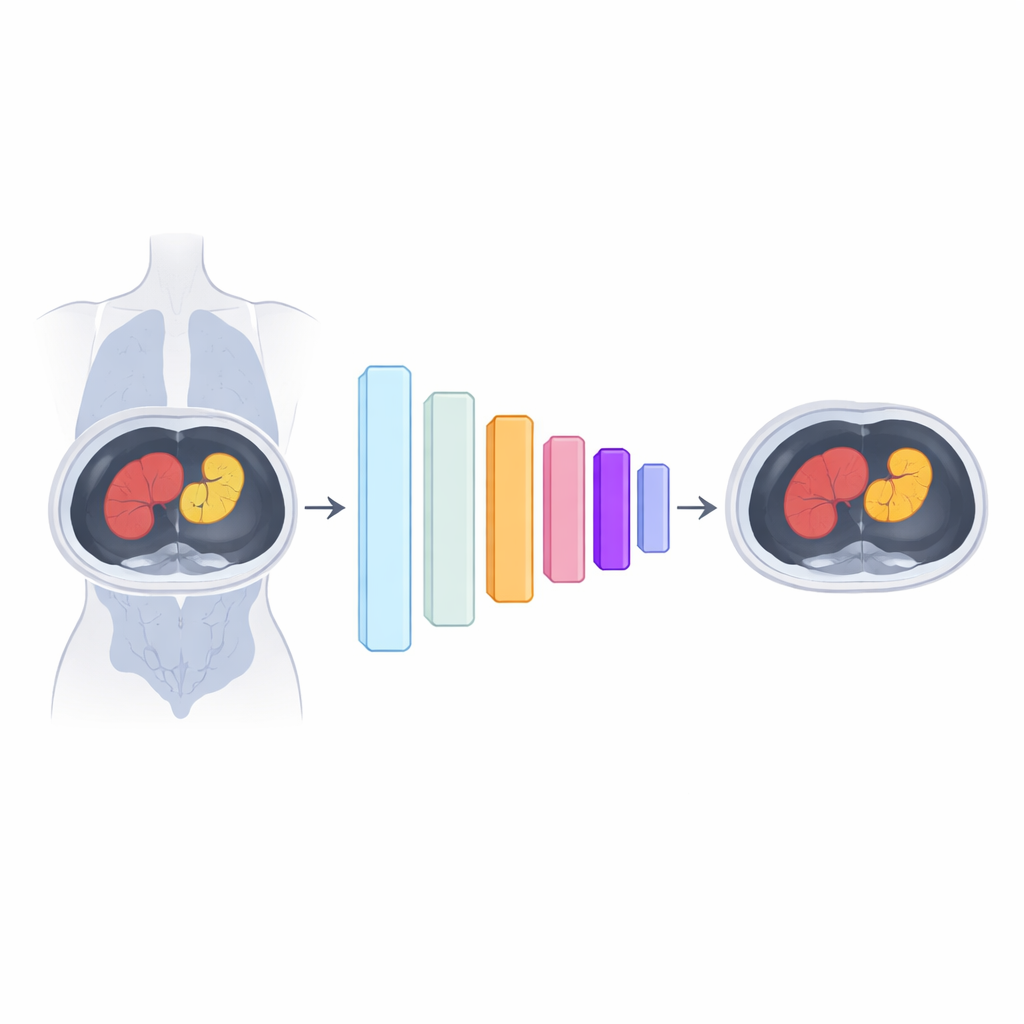
Keeping flexibility while cutting complexity
A key advantage of this design is that it reduces the number of parameters that must be learned without forcing the model into a rigid, low-rank form. Whereas popular compression methods require engineers to guess in advance how much to simplify each weight matrix, the quantum-inspired generators naturally adapt how rich their outputs need to be during training. The authors also explore three simple ways to convert the limited range of quantum probabilities into the broader range of values that a classical network expects, balancing hardware friendliness, parameter count, and numerical precision. Because the quantum modules are used only while the model learns and can be simulated efficiently on existing hardware, the approach is practical even before large, fault-tolerant quantum computers exist.
Putting the framework to the test
To see whether this hybrid setup pays off in practice, the team trained five well-known segmentation models on a public kidney CT dataset that includes both organ and tumor labels. They compared conventional training with versions in which only selected layers were driven by quantum-style generators. Across the board, the hybrid models produced cleaner outlines with less speckled noise and closer agreement with expert labels. Gains were especially strong for the more delicate task of tumor segmentation, where boundaries are small, jagged, and easy to miss. In one case, a popular architecture improved its overlap score for kidney tumors by nearly seven percentage points while using fewer trainable parameters. The authors also showed that sprinkling quantum-generated layers throughout different parts of a network usually helped performance, and that even shallow quantum circuits were enough to achieve these benefits.
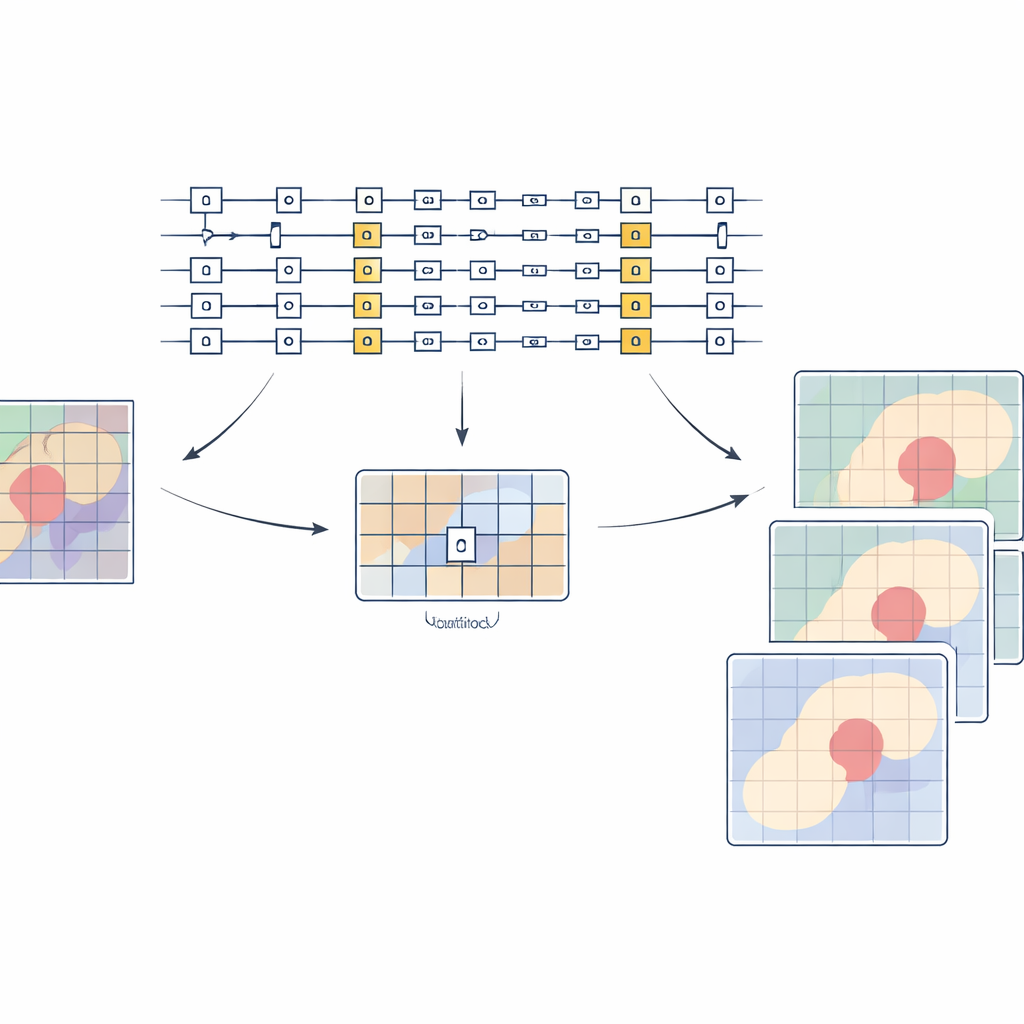
What this means for future clinics
For non-specialists, the main message is that the study offers a way to make medical AI both slimmer and smarter by letting a small quantum-inspired core orchestrate a much larger classical model. Rather than relying on ever-bigger networks and ever-larger datasets, this hybrid training scheme squeezes more value out of scarce annotated scans, particularly for hard-to-see tumors. Because it operates only during training and can run on today’s hardware, it could be woven into existing development pipelines without changing hospital equipment. If extended beyond kidneys to other organs and cancer types, approaches like HQC-TF may help bring reliable, automated contouring closer to everyday use in radiation therapy, supporting clinicians in delivering safer, more precise treatments.
Citation: Sun, Q., Chen, J., Fan, Y. et al. A Flexible Hybrid Quantum-classical Training Framework of Organ-at-Risk and Tumor Segmentation Models for Radiation Therapy Planning. Sci Rep 16, 9265 (2026). https://doi.org/10.1038/s41598-026-40127-z
Keywords: medical image segmentation, radiation therapy, quantum machine learning, deep learning, kidney tumor