Clear Sky Science · en
Evaluation of bacteriophage efficacy against Pseudomonas aeruginosa in ex vivo and in vitro canine skin systems
Why skin infections need new answers
Stubborn skin infections are not just a nuisance for pets and their owners—they can become long‑lasting, painful problems that resist standard antibiotics. One frequent culprit is Pseudomonas aeruginosa, a hard‑to‑kill bacterium that thrives in slimy biofilms on wounds. This study explores whether viruses that infect bacteria, called bacteriophages or simply phages, can safely clear these infections in dog skin models, pointing to new options that may one day help both animals and people.
A tough germ that loves slime
Pseudomonas aeruginosa is an opportunistic germ that strikes when the body’s defenses are weakened, causing serious lung, blood, and wound infections in hospitals and veterinary clinics. It often grows in biofilms—thin, sticky layers of bacteria glued together by a protective matrix. Inside these films, the microbes become much harder to reach with antibiotics and can better dodge the immune system, turning treatable infections into chronic ones. Because multidrug‑resistant Pseudomonas is now on the World Health Organization’s high‑priority list, researchers are urgently searching for alternatives to traditional drugs.
Viruses that hunt bacteria
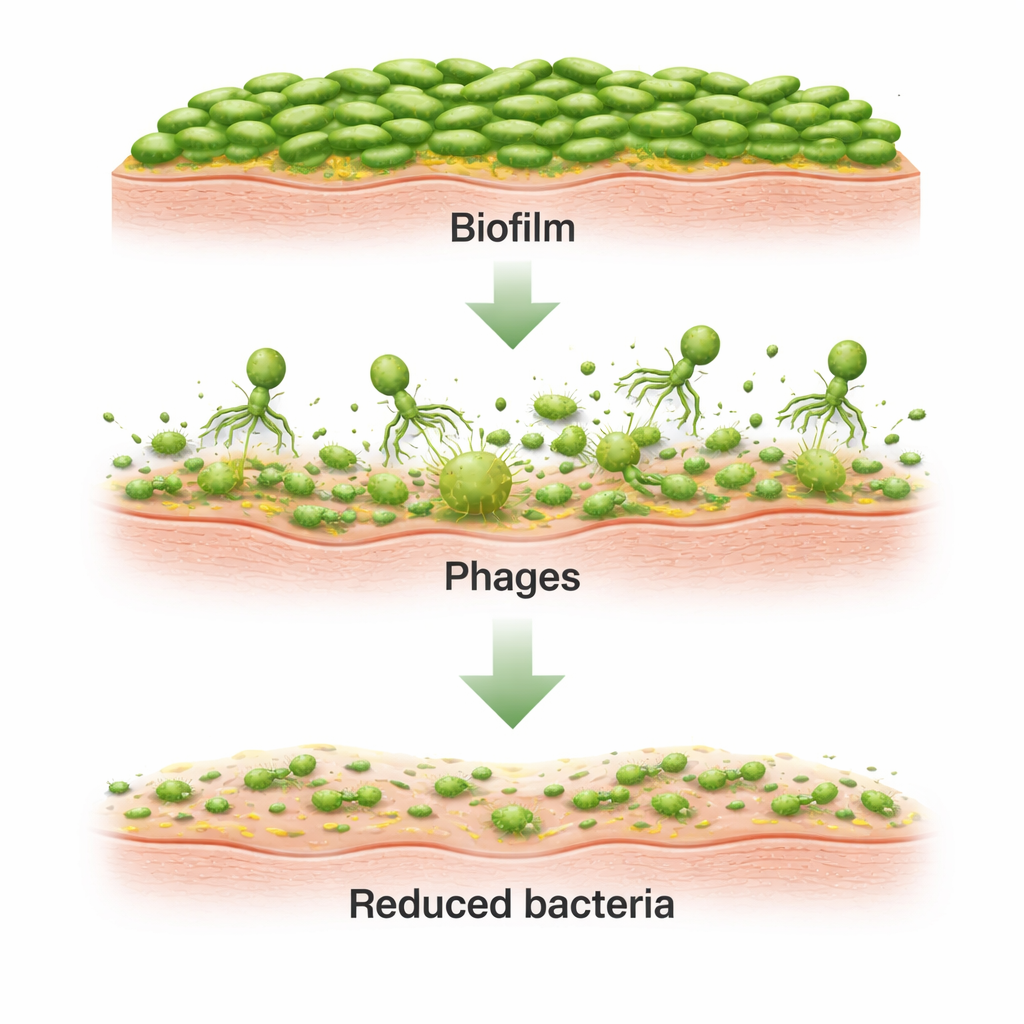
Bacteriophages are naturally occurring viruses that infect and kill bacteria while leaving animal and human cells unharmed. The team focused on two well‑characterized phages, JG003 and PTLAW1, chosen because they are strongly lytic and carry genes that may help break down biofilms. First, the researchers tested these phages on laboratory‑grown Pseudomonas biofilms formed on plastic plates. Using fluorescent dyes and 3D confocal microscopy, they showed that each phage alone—and especially the pair together—substantially reduced the total biofilm volume and increased the share of bacteria with damaged membranes, signaling effective attack on the bacterial community.
Putting phages onto real dog skin
Biofilms on plastic are far simpler than real skin, which has layers, hair follicles, glands, and a tough outer barrier. To recreate more lifelike conditions, the researchers used two canine skin systems. In an ex vivo setup, thin discs of dog skin were mounted in Franz‑type diffusion cells—glass devices with a donor chamber above and a receptor chamber below the skin. After infecting the surface with Pseudomonas for 16 hours, they applied a cocktail of the two phages. Over the next eight hours, bacterial counts inside the skin fell by about four orders of magnitude compared with untreated samples. Microscopy confirmed that dense clusters of bacteria and biofilm matrix on the skin surface were largely broken up after phage exposure, while the phages themselves remained active but did not cross through into the lower chamber.
A living skin model that can feel inflammation
Next, the scientists grew a living “epidermal equivalent” from dog skin cells on permeable inserts, mimicking the outer layers of skin including a proper cornified surface. This allowed them not only to measure bacterial survival but also to track inflammatory signals. When these skin equivalents were infected with a small number of Pseudomonas cells, then treated with the phage cocktail three hours later, bacterial levels plummeted—no bacteria were detectable in most treated samples. At the same time, levels of the pro‑inflammatory messenger CXCL8, which rise sharply during infection, dropped back toward normal both at the gene and protein level. Importantly, phage exposure alone did not harm the skin cells or disrupt the barrier structure.
What this means for future wound care
Taken together, these experiments show that carefully selected phages can meaningfully chip away at Pseudomonas biofilms, lower bacterial loads, and calm early inflammation in realistic dog skin models, without obvious side effects. While the treatments did not completely sterilize heavily infected skin and were tested mainly on early‑stage biofilms, the work provides a reproducible preclinical framework to refine phage doses, timing, and combinations—possibly alongside antibiotics. For veterinarians managing difficult canine wounds, and for doctors facing similar infections in humans, phage‑based sprays or creams may eventually offer a targeted way to tackle biofilm‑driven infections that no longer yield to standard drugs.
Citation: Dalponte, A., Filor, V., Nerlich, A. et al. Evaluation of bacteriophage efficacy against Pseudomonas aeruginosa in ex vivo and in vitro canine skin systems. Sci Rep 16, 7167 (2026). https://doi.org/10.1038/s41598-026-40091-8
Keywords: bacteriophage therapy, Pseudomonas skin infection, biofilm, canine wound model, antibiotic resistance