Clear Sky Science · en
The role of IL-17 in orchestrating macrophage polarization via NF-κB, mTOR/HIF-1α and pyroptosis in diabetes
Why This Matters for People with Diabetes
Type 2 diabetes is often described as a problem of blood sugar, but behind the numbers lies a slow-burning fire of inflammation that damages organs over time. This study looks at a powerful immune signal called IL-17 and shows how it can act like an amplifier, turning the body’s defender cells into overzealous attackers in a high-sugar environment. By understanding and dialing down this signal, the research points to new ways to protect people with diabetes from long-term complications.
Immune Cells at a Crossroads
Our immune system relies on cells called macrophages, which patrol tissues and either promote healing or stoke inflammation depending on the situation. In type 2 diabetes, these cells are frequently pushed into a more aggressive, damage-promoting mode. The authors focused on IL-17, a messenger molecule of the immune system that is elevated in many inflammatory diseases. They asked how IL-17 behaves when blood sugar is high, and whether blocking it could shift macrophages back toward a healthier balance.
Setting Up a High-Sugar Mini-World in the Lab
To explore this, the researchers used a human immune cell line and coaxed it into becoming macrophages. They then exposed these cells to a normal or high level of sugar, added IL-17, and in some experiments applied an IL-17–blocking antibody similar in spirit to drugs already used for skin and joint diseases. They measured how the cells grew, which genes they switched on, and which proteins they produced, focusing on markers of aggressive behavior, inflammatory signal release, and a particularly fiery form of cell death.
How IL-17 Turns Up the Heat
Under high sugar conditions, IL-17 pushed macrophages strongly toward an inflammatory, tissue-damaging state. The cells pumped out higher levels of irritant molecules such as IL-1β, IL-6, and TNF-α, while dialing down IL-10, which normally helps cool inflammation. Inside the cells, key control hubs switched on: one pathway tied to stress and metabolism and another that prepares proteins for harsh, low-oxygen environments. Together, these changes primed a molecular complex that triggers a dramatic form of cell bursting, spilling toxic contents that can injure nearby tissue and worsen insulin resistance.
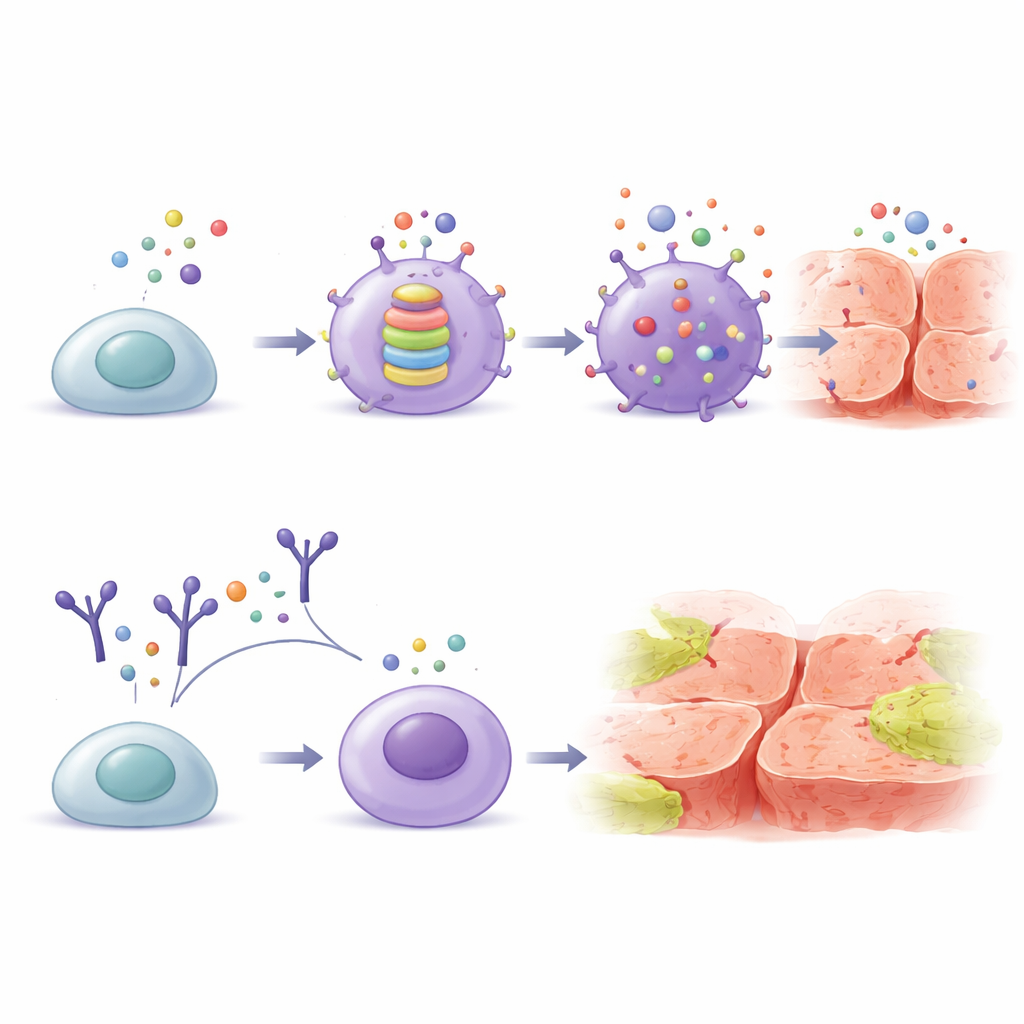
Putting the Brakes on With an Antibody
When the team added an antibody that neutralizes IL-17, many of these harmful changes were partially reversed. The macrophages produced fewer inflammatory signals, the internal stress pathways quieted down, and the hallmarks of explosive cell death were reduced. In simple terms, blocking IL-17 helped shift the cells away from a destructive mode and back toward a more balanced state, even in a high-sugar setting. This suggests that IL-17 is not just a bystander but a central volume knob for immune overreaction in diabetes.
What This Could Mean for Future Treatment
For people living with type 2 diabetes, this work highlights that controlling blood sugar is only part of the story; calming misdirected immune activity may be just as important. The study suggests that drugs which block IL-17, already in use for other conditions, might someday be repurposed to reduce chronic inflammation, protect insulin-producing cells, and slow organ damage in diabetes. At the same time, IL-17 helps the body fight infections, so any treatment would need to be carefully tailored to avoid weakening everyday defenses. Still, this research offers an early but promising glimpse of how targeting a single immune signal could help cool the hidden fire driving diabetic complications.
Citation: Niu, J., Liu, G., He, G. et al. The role of IL-17 in orchestrating macrophage polarization via NF-κB, mTOR/HIF-1α and pyroptosis in diabetes. Sci Rep 16, 8559 (2026). https://doi.org/10.1038/s41598-026-40079-4
Keywords: type 2 diabetes, inflammation, macrophages, IL-17, immunotherapy