Clear Sky Science · en
Oncofetal chondroitin sulfate positive circulating tumor cells as prognostic biomarkers in early-stage melanoma
Why tiny traces in blood can matter
Skin cancer is often curable when caught early, yet some people with seemingly small, local tumors still develop life-threatening disease years later. This study explores whether a simple blood test can reveal which early-stage melanoma patients are already at higher risk, by hunting for rare cancer clues drifting in the bloodstream long before scans or standard lab tests show trouble.
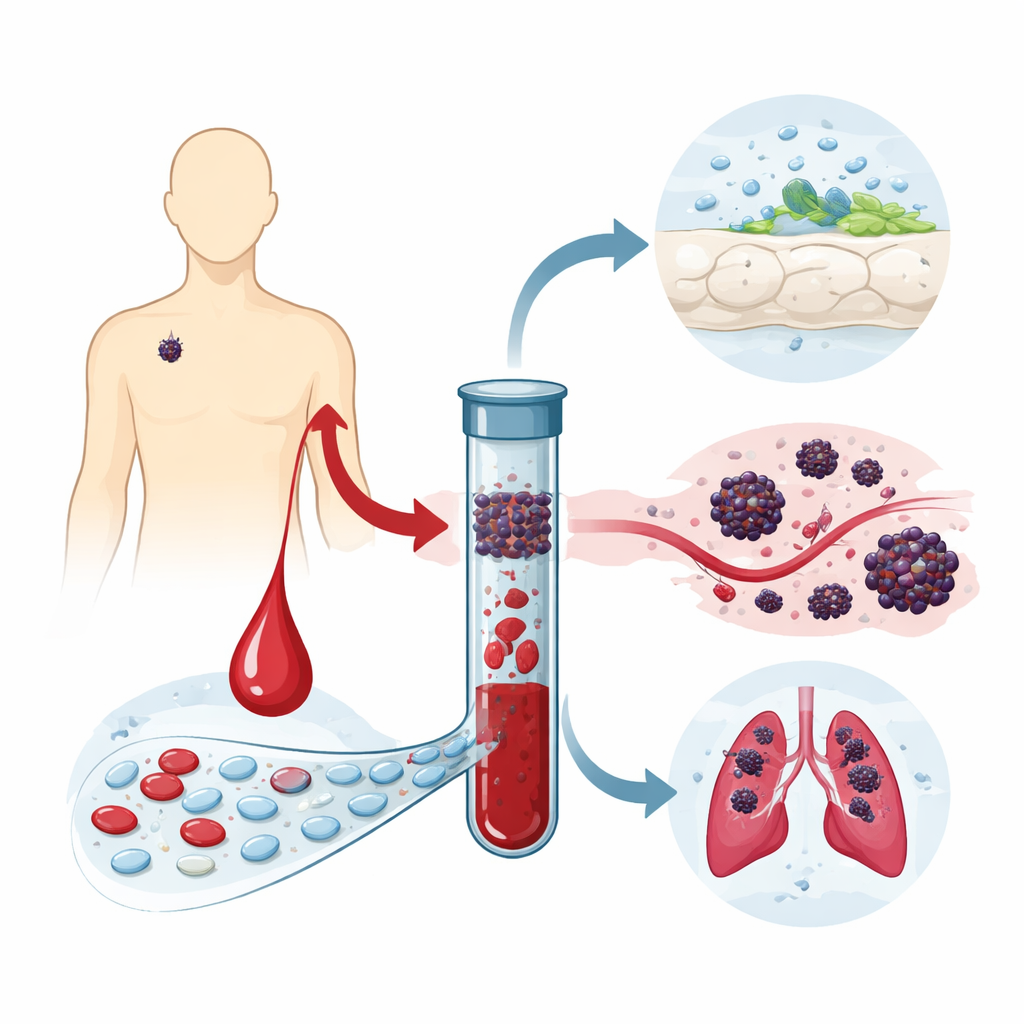
A closer look at early skin cancer
Melanoma is the most dangerous form of skin cancer. Many patients are diagnosed while the tumor is still confined to the skin and their lymph nodes appear clear, suggesting a good outlook. Still, about one in ten will later develop spread of the cancer. Doctors currently rely on features like how thick the tumor is under the microscope to estimate risk, but these measures do not fully explain why some patients progress and others do not. The idea behind this work is that cancer cells and fragments of tumor DNA may already be escaping into the blood, turning melanoma into a whole-body disease much earlier than we can see with standard tests.
Finding wandering cancer cells in a blood sample
The researchers focused on 92 people with early-stage melanoma (stages I and II) whose nearby sentinel lymph node tested negative, meaning no visible spread. Each person gave a single blood sample around the time of diagnosis. From this sample, the team tried to detect two different signals. First, they looked for whole cancer cells that had broken away from the tumor and entered the bloodstream, known as circulating tumor cells. To catch these extremely rare cells, they used a trapping method based on a malaria protein called rVAR2 that sticks to a sugar-like structure, oncofetal chondroitin sulfate, found on many cancer cells but not on normal blood cells. After enrichment with magnetic beads, they stained the captured cells with glowing markers that highlight melanoma and tumor-initiating features, allowing trained observers to count suspected tumor cells under a microscope.
Reading fragments of tumor DNA in blood
At the same time, the team isolated free-floating DNA fragments from the same patients’ blood. Using a high-sensitivity gene panel that scans 29 cancer-related genes, they searched for mutations typical of melanoma. When at least one clearly harmful or likely harmful gene change was present, the sample was considered positive for tumor DNA. This approach, often called a liquid biopsy, is already used to track advanced cancers, but its usefulness in very early melanoma, where the total amount of tumor material is tiny, has been uncertain.
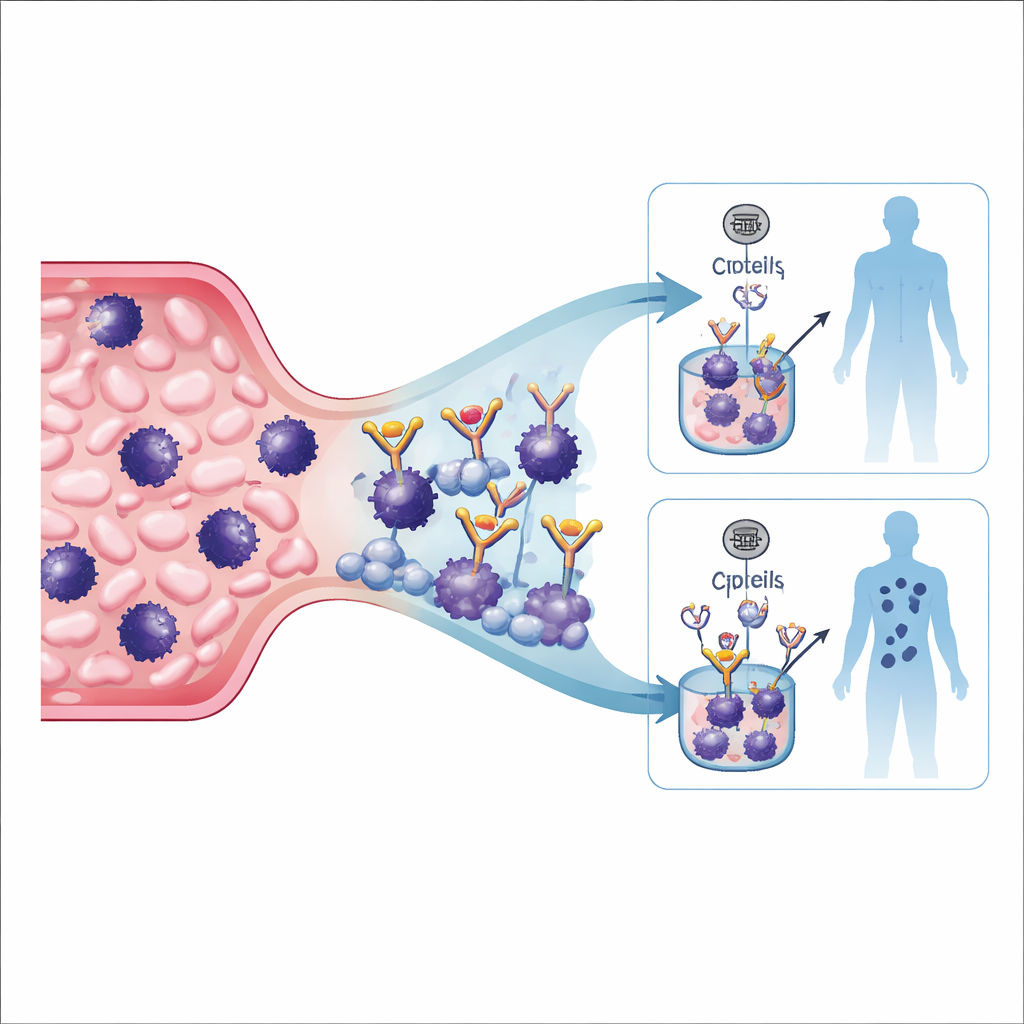
What the blood signals revealed about future risk
Over a median of about two and a half years of follow-up, the patients were monitored for tumor progression or death from melanoma. Circulating tumor cells were found in 21 of the 92 patients—nearly 23 percent—despite their disease being labeled early and their sentinel nodes negative. Having just one or more of these cells in the initial blood sample was linked to a significantly higher chance of later progression or melanoma-related death. In contrast, only six patients had clearly abnormal tumor DNA in their blood, and this signal alone did not strongly predict outcome, likely because early tumors shed very little DNA. However, when the presence of tumor cells and tumor DNA were evaluated together, patients with either signal present faced a clearly higher risk of their melanoma worsening, even after accounting for age, sex, ulceration, and tumor thickness.
What this could mean for patients
This study suggests that, for some people, melanoma may behave like a body-wide disease earlier than current tests reveal. Detecting even a handful of tumor cells in a routine blood draw, especially when combined with sensitive DNA analysis, could help flag patients who need closer follow-up or may benefit from more intensive treatment while their disease still appears limited. Although larger studies and more standardized lab methods are needed before such tests become part of everyday care, the work points toward a future in which a simple tube of blood could sharpen risk estimates, guide personalized therapy, and potentially catch dangerous spread of melanoma before it is visible by other means.
Citation: Sunzenauer, J., Rammer, M., Stöckl, L. et al. Oncofetal chondroitin sulfate positive circulating tumor cells as prognostic biomarkers in early-stage melanoma. Sci Rep 16, 10034 (2026). https://doi.org/10.1038/s41598-026-40072-x
Keywords: melanoma, liquid biopsy, circulating tumor cells, tumor DNA, cancer prognosis