Clear Sky Science · en
The antitumor mechanism of oclacitinib in canine lymphoma
Why this matters for dogs and their families
Lymphoma is one of the most common cancers in pet dogs, and while chemotherapy can help many patients, it does not always work and can bring significant side effects. Oclacitinib, a pill already widely prescribed to itchy dogs with allergic skin disease, has recently shown surprising benefits in some dogs with skin lymphoma. This study asks a simple but important question: how, exactly, might this everyday allergy drug act against cancer cells, and can we predict which dogs are most likely to benefit?
From allergy medicine to cancer fighter
Oclacitinib was developed to calm overactive immune signals that drive atopic dermatitis. It does this by blocking a family of molecular switches called JAKs, especially JAK1, which relay messages from inflammatory molecules on the cell surface to the nucleus. Veterinarians began noticing that some dogs with a form of skin lymphoma improved when given oclacitinib, but the reasons were unclear and responses were inconsistent. The researchers set out to study this in a controlled way, using laboratory-grown lymphoma cells from dogs, along with samples from real canine patients, to see when and how oclacitinib can actually kill cancer cells rather than just soothe the skin. 
Testing lymphoma cells in the lab
The team examined one canine cutaneous (skin) lymphoma cell line and eight additional high-grade lymphoma cell lines representing both T-cell and B-cell cancers. When they exposed these cells to oclacitinib at concentrations similar to those reached in treated dogs, about half of the cell lines showed clear slowing of growth, and several underwent cell death. Sensitive cells accumulated in a resting phase of the cell cycle and displayed hallmarks of programmed cell death, including activation of an executioner protein called caspase-3. In contrast, other lymphoma lines hardly responded at all within the same concentration range, highlighting that lymphoma in dogs is not a single disease but a collection of biologically distinct cancers.
Zeroing in on a vulnerable signaling route
To find out what separated sensitive from resistant tumors, the scientists focused on the JAK/STAT signaling route, a common survival pathway in many blood cancers. They found that only the lymphoma cell lines that responded to oclacitinib showed strong baseline activation of two key players: JAK1 and STAT5. When these sensitive cells were treated with oclacitinib, the activity of STAT5 dropped sharply, while overall levels of the proteins stayed the same. A second JAK1-blocking drug used in human medicine, filgotinib, produced a similar pattern of growth inhibition and STAT5 shutdown. Deep gene-activity profiling revealed that oclacitinib dampened sets of genes linked to JAK/STAT signaling and reduced expression of c-Myc, a master growth driver commonly hijacked in cancers, helping explain the observed cell cycle arrest and cell death. 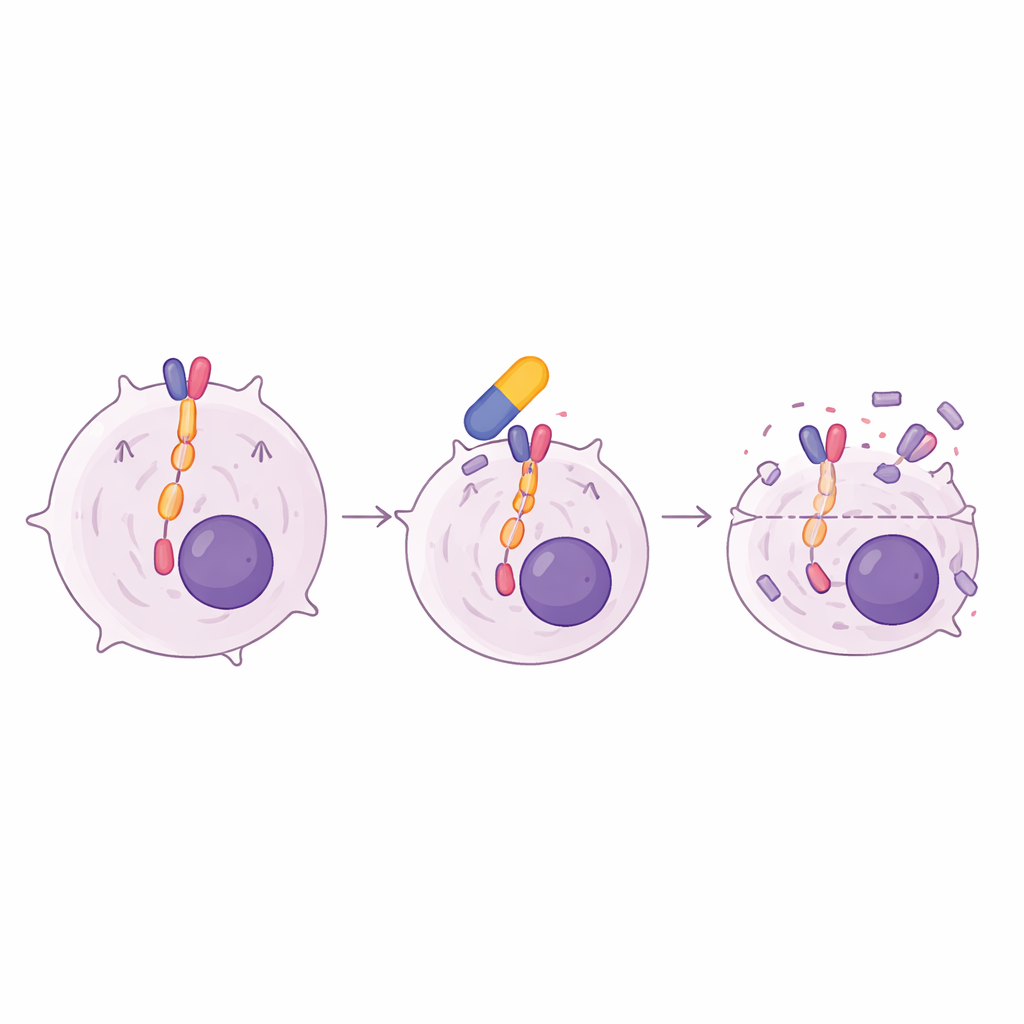
Linking lab findings to real canine patients
The researchers then turned to biopsy samples from dogs with cutaneous lymphoma that had been treated with oclacitinib in clinics. Using staining methods that reveal activated proteins, they found that dogs whose tumors showed activated JAK1 and STAT5 were more likely to experience partial or complete responses to the drug. Some non-responders still had these signals turned on, suggesting that additional escape routes can sustain certain tumors, but the overall pattern pointed to a strong association. Importantly, many lymph node and intestinal lymphoma samples from other dogs also displayed this same JAK1/STAT5 activation pattern, raising the possibility that oclacitinib’s benefits might extend beyond skin lymphoma to a broader set of aggressive canine lymphomas.
What this could mean for future treatment
Taken together, the work shows that oclacitinib can directly hinder and sometimes kill certain canine lymphoma cells by switching off a key survival pathway centered on JAK1 and STAT5 and lowering the activity of growth-promoting genes such as c-Myc. Normal lymphocytes from healthy dogs were far less affected at clinically relevant doses, though high concentrations did slow the expansion of activated immune cells, underscoring the need to balance anti-cancer effects with potential immune suppression. The study suggests that testing tumors for activated JAK1 and STAT5 could help veterinarians identify dogs most likely to benefit from oclacitinib and similar drugs. While more clinical trials are needed, this widely available allergy medication may become part of a targeted treatment toolkit for canine lymphoma, guided by simple molecular markers.
Citation: Harada, M., Inanaga, S., Sakurai, M. et al. The antitumor mechanism of oclacitinib in canine lymphoma. Sci Rep 16, 8427 (2026). https://doi.org/10.1038/s41598-026-40066-9
Keywords: canine lymphoma, oclacitinib, JAK1 STAT5 pathway, targeted cancer therapy, veterinary oncology