Clear Sky Science · en
Tribolium castaneum with longer duration of tonic immobility have more variations corresponding to the human Parkinson’s disease genomic region
Why a tiny beetle matters for brain disease
Parkinson’s disease is best known for its tremors and difficulty with movement, but at its core it is a problem of brain cells that use the chemical messenger dopamine. This study shows that a common pantry pest, the red flour beetle, can develop Parkinson-like traits after generations of selection for an unusual anti-predator tactic: playing dead. By tying this dramatic behavior to specific gene changes that resemble those seen near human Parkinson’s genes, the work suggests that even a small beetle can help us understand a major human brain disorder.
A beetle that cheats death by standing still
When attacked by predators, some red flour beetles drop motionless and “play dead” in a state called tonic immobility. The researchers previously bred two strains: one that stays motionless for a long time (the L-strain) and one that snaps out of it quickly (the S-strain). Earlier work showed that the long-feigning beetles move more slowly, have odd walking patterns, and carry lower levels of dopamine in their brains. Giving them dopamine, or even caffeine, shortens their death-feigning episodes. In nature, the slow, long-feigning beetles actually survive better against certain spiders and assassin bugs, because staying still makes them harder to catch. Thus, a survival strategy in the wild produces beetles that, in the lab, look surprisingly similar to Parkinson’s patients in how they move.
Scanning beetle DNA for echoes of Parkinson’s
To see whether these Parkinson-like traits were tied to genetic changes similar to those in people, the team compared the full DNA sequences of the long- and short-feigning strains. They looked for small changes in the genetic code, larger insertions and deletions, and changes in how many copies of a gene the beetles carry. They then asked which of these altered beetle genes closely match human genes already implicated in Parkinson’s disease or in the dopamine system. The answer was striking: the long-feigning strain carried many more mutations in beetle versions of human Parkinson-related genes than the short-feigning strain. These included genes involved in stress-responsive “chaperone” proteins and in the energy-producing machinery of mitochondria, both of which are central suspects in human Parkinson’s.
From genes to dopamine and movement
The study went beyond raw DNA sequence and examined how strongly different genes are switched on in the beetle body. Using earlier RNA sequencing data, the authors connected dozens of genes that differed between strains to human pathways for monoamine signaling, a family of brain chemicals that includes dopamine. In long-feigning beetles, enzymes involved in turning the amino acid tyrosine into dopamine showed altered expression and carried subtle sequence changes. This suggests that dopamine is being produced and processed differently in these animals. They also uncovered differences in beetle genes related to insulin and sugar handling that match human and fruit fly genes linked to Parkinson’s and to diabetes—two conditions already known to influence one another in people.
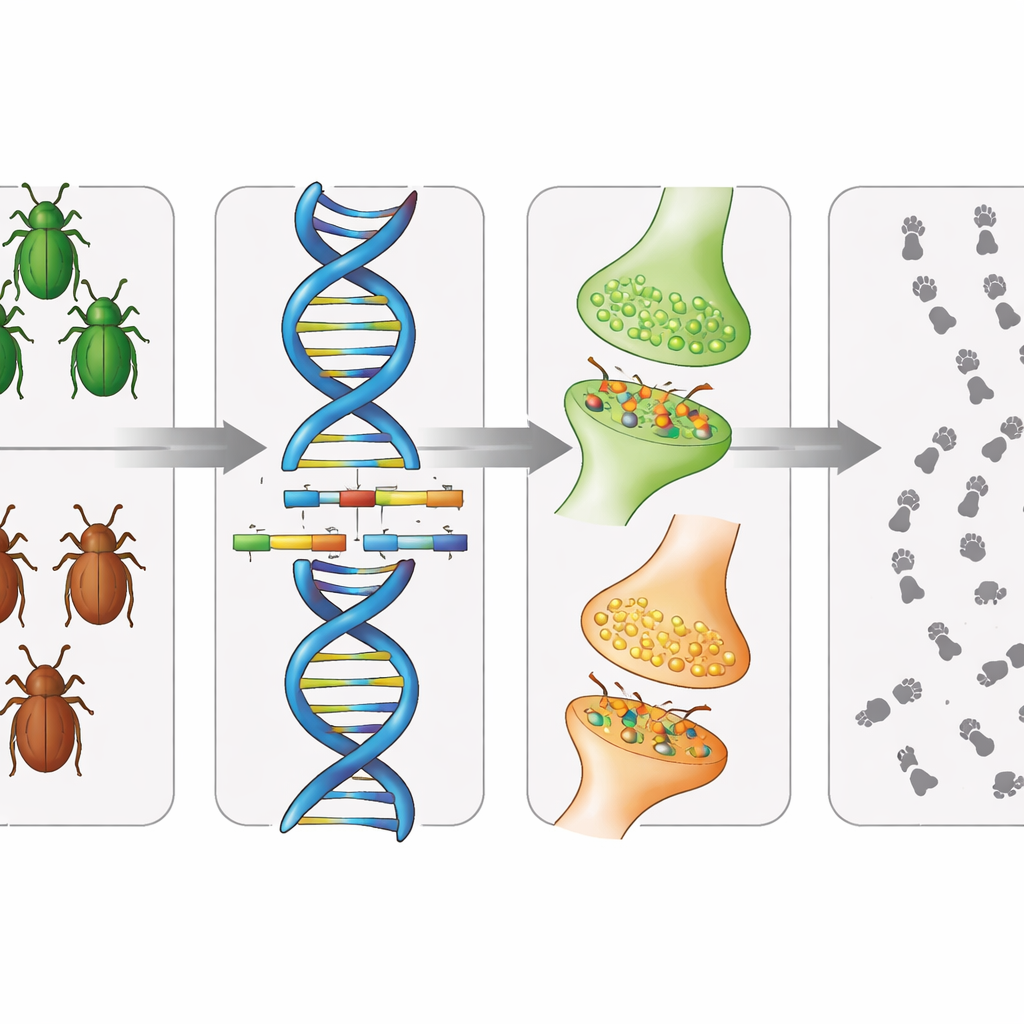
A web of signals, not a single “Parkinson gene”
Importantly, the classic human Parkinson genes such as LRRK2 and PINK1 did not show obvious mutations in these beetles. Instead, the long-feigning strain appears to accumulate a cluster of changes across many genes that feed into dopamine signaling, mitochondrial health, stress responses, and insulin pathways. Network analyses showed that these beetle genes map onto human pathways labeled as different Parkinson subtypes. The authors argue that this mirrors the human situation, where Parkinson’s is better viewed as a syndrome arising from disturbed networks of genes and cellular systems, not from a single faulty switch.
What this means for people and for predators
To a non-specialist, the key message is that a behavior shaped by evolution to outwit predators can accidentally recreate features of a human brain disease. By selecting beetles for how long they “play dead,” scientists ended up with animals whose genes and brain chemistry echo important aspects of Parkinson’s, including low dopamine, stiff and altered walking, and disruptions in energy and insulin signaling. While these beetles do not “have Parkinson’s,” they offer a simple, genetically tractable model to probe how changes across many pathways combine to disturb movement. Such models may help researchers test ideas about how stress, metabolism, and dopamine interact in the human brain, ultimately informing new approaches to prevent or treat Parkinson-like disorders.
Citation: Tanaka, K., Sasaki, K., Yajima, S. et al. Tribolium castaneum with longer duration of tonic immobility have more variations corresponding to the human Parkinson’s disease genomic region. Sci Rep 16, 8840 (2026). https://doi.org/10.1038/s41598-026-40050-3
Keywords: Parkinson’s disease, dopamine, red flour beetle, tonic immobility, insulin signaling