Clear Sky Science · en
Clinically interpretable extracellular vesicle gene model for Non-Invasive liver cancer diagnosis
Why a Blood Test for Liver Cancer Matters
Liver cancer is one of the deadliest cancers in the world, largely because it is often discovered too late for curative treatment. Today’s tools for finding it early—imaging scans and liver biopsies—are expensive, sometimes risky, and not always accurate. This study explores a different idea: whether a simple blood sample, analyzed with smart computer models, could reveal early signs of liver cancer by reading tiny genetic messages that diseased cells send into the bloodstream.
Tiny Messengers Floating in Our Blood
All cells in our body regularly release microscopic bubbles, known as extracellular vesicles, into blood and other body fluids. These vesicles carry proteins, fats, and bits of genetic material that reflect the state of the cells they came from. Cancer cells also shed such vesicles, but their cargo looks different from that of healthy cells. Because vesicles travel in the circulation, they can be collected from a simple blood draw rather than a needle inserted into the liver. The authors used a large public database, exoRBase 3.0, which contains detailed measurements of genetic material found in vesicles from hundreds of people with and without liver cancer.
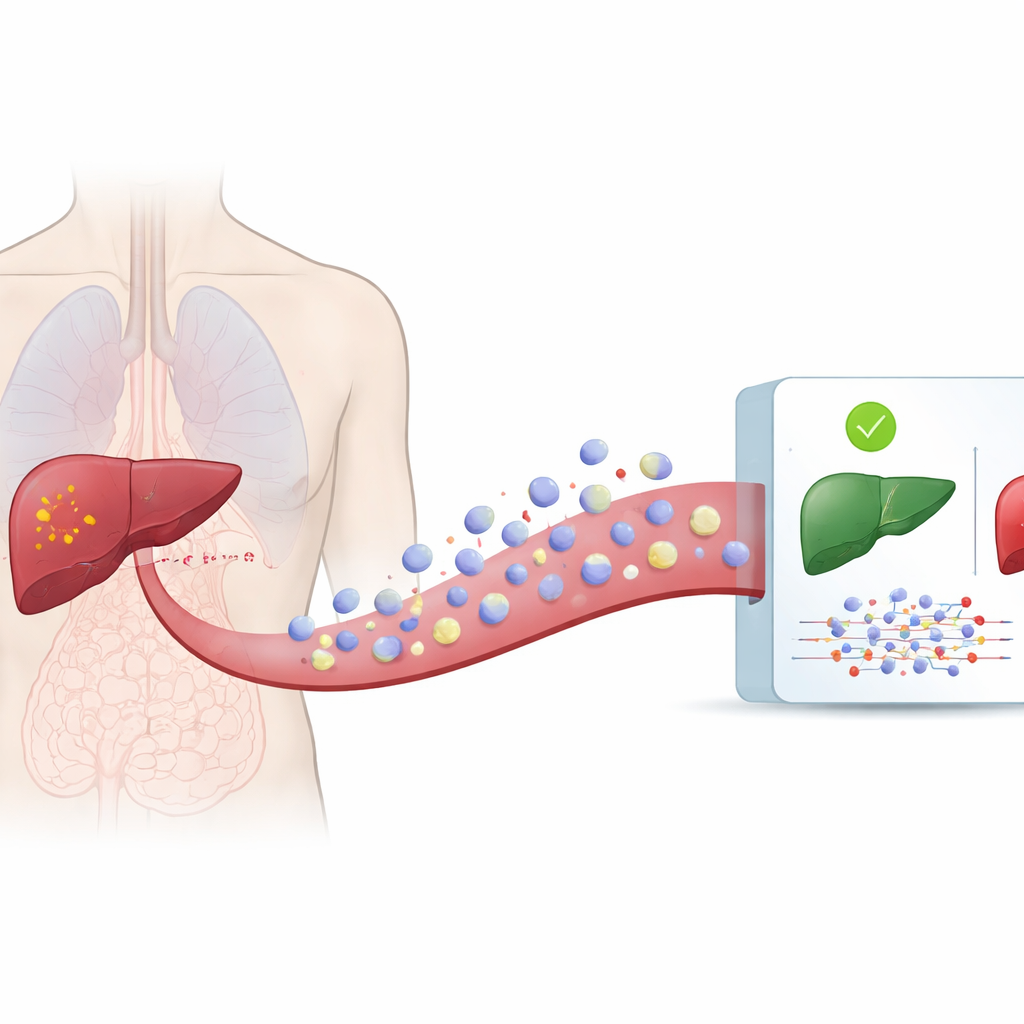
Teaching Computers to Read Vesicle Signals
From this database, the team gathered information on two types of RNA molecules—messenger RNA, which carries instructions for making proteins, and long non-coding RNA, which helps regulate cell behavior. They analyzed samples from 112 patients with liver cancer and 118 healthy individuals. After cleaning and standardizing the data, they trained six different computer models to tell cancer and non-cancer samples apart based on patterns in vesicle RNA. These methods ranged from traditional statistical tools to more flexible deep neural networks that can uncover complex patterns.
Finding a Small but Powerful Gene Panel
The deep neural network achieved the best performance within this dataset, correctly separating cancer from healthy blood samples much of the time. However, a model that relies on thousands of measurements is hard to understand and impractical for routine testing. To address this, the researchers applied a method called SHAP, which helps identify which inputs matter most for a model’s decisions. This allowed them to narrow the signature down to just ten specific messenger RNAs carried by vesicles. One of them, MTRNR2L8, stood out as making the strongest contribution to the model’s predictions, with others like HBB, PF4, FTL, and S100A9 also playing key roles. Even with only these ten RNAs, the model still performed well, suggesting that a relatively small, focused panel might be enough for a blood-based test.
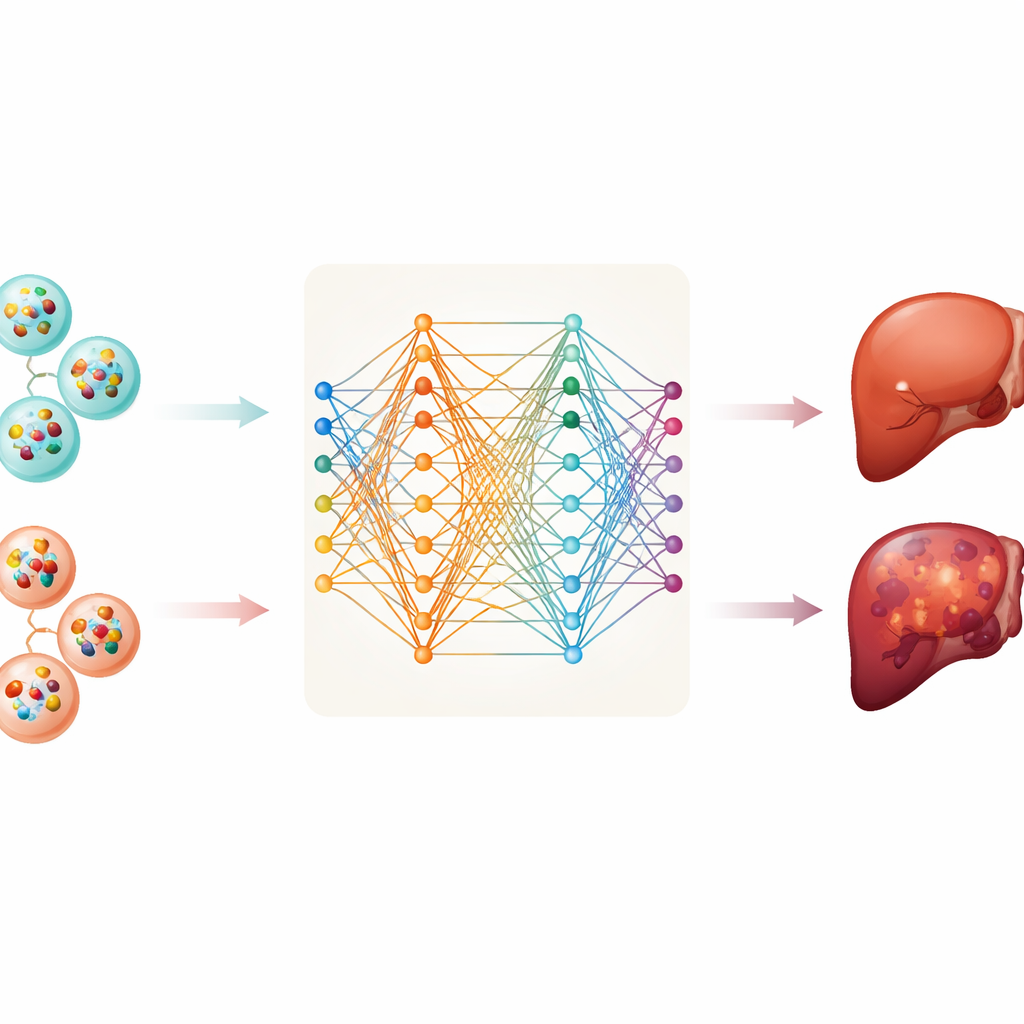
Opening the “Black Box” of Artificial Intelligence
Doctors are often wary of computer models that give answers without clear reasons. To build trust, the team emphasized transparency. SHAP was used not only to rank the importance of each gene overall, but also to show, for a given patient, how each gene pushed the prediction toward “cancer” or “healthy.” The researchers also tried a newer type of network, the Kolmogorov–Arnold Network, which represents relationships between inputs and outputs with explicit mathematical curves. This approach confirmed that the same ten genes carry strong signals and illustrated how their combined behavior shapes the final decision, offering a more interpretable view of what the model has learned.
From Laboratory Concept to Possible Clinic Tool
As a proof of concept, the authors built an online demonstration platform where users can enter vesicle gene measurements and see the model’s predicted probability of liver cancer along with a visual breakdown of which genes influenced the outcome. They stress, however, that the work is still at an experimental stage. The model has so far been tested only on data from the same public database, and real-world patients often have mixed liver diseases, varied treatments, and technical differences in how samples are collected. Larger, carefully designed studies in independent groups of patients—using standardized methods—will be required before such a test can be used in hospitals or screening programs.
What This Means for Patients
This study shows that a small set of genetic markers carried by tiny particles in the blood could, in principle, help detect liver cancer without invasive procedures. By combining these markers with computer models that doctors can understand and inspect, the work points toward future blood tests that are both accurate and trustworthy. While significant hurdles remain before clinical use, such as confirming the results in many different populations and ensuring that the test is practical and affordable, this research offers a glimpse of how liquid biopsies and interpretable artificial intelligence might one day make earlier, safer liver cancer diagnosis a reality.
Citation: Zhang, Y., Mo, Z., Zhang, L. et al. Clinically interpretable extracellular vesicle gene model for Non-Invasive liver cancer diagnosis. Sci Rep 16, 9054 (2026). https://doi.org/10.1038/s41598-026-40020-9
Keywords: liver cancer, liquid biopsy, extracellular vesicles, machine learning, early diagnosis