Clear Sky Science · en
Green synthesis and enhanced photocatalytic activity of ZnSe nanoparticles capped with Artemisia herba-alba and calligonum plants extracts
Turning Plants into Pollution Fighters
Many of the bright colors in clothes, plastics, and packaging come from synthetic dyes that don’t easily break down in nature. When these dyes end up in rivers and lakes, they can harm ecosystems and pose health risks. This study explores a way to clean such polluted water using tiny particles made from zinc and selenium, built with help from everyday desert plants instead of harsh chemicals. The work shows how green chemistry can turn common vegetation into a tool for purifying water in an energy-efficient, low-cost way.
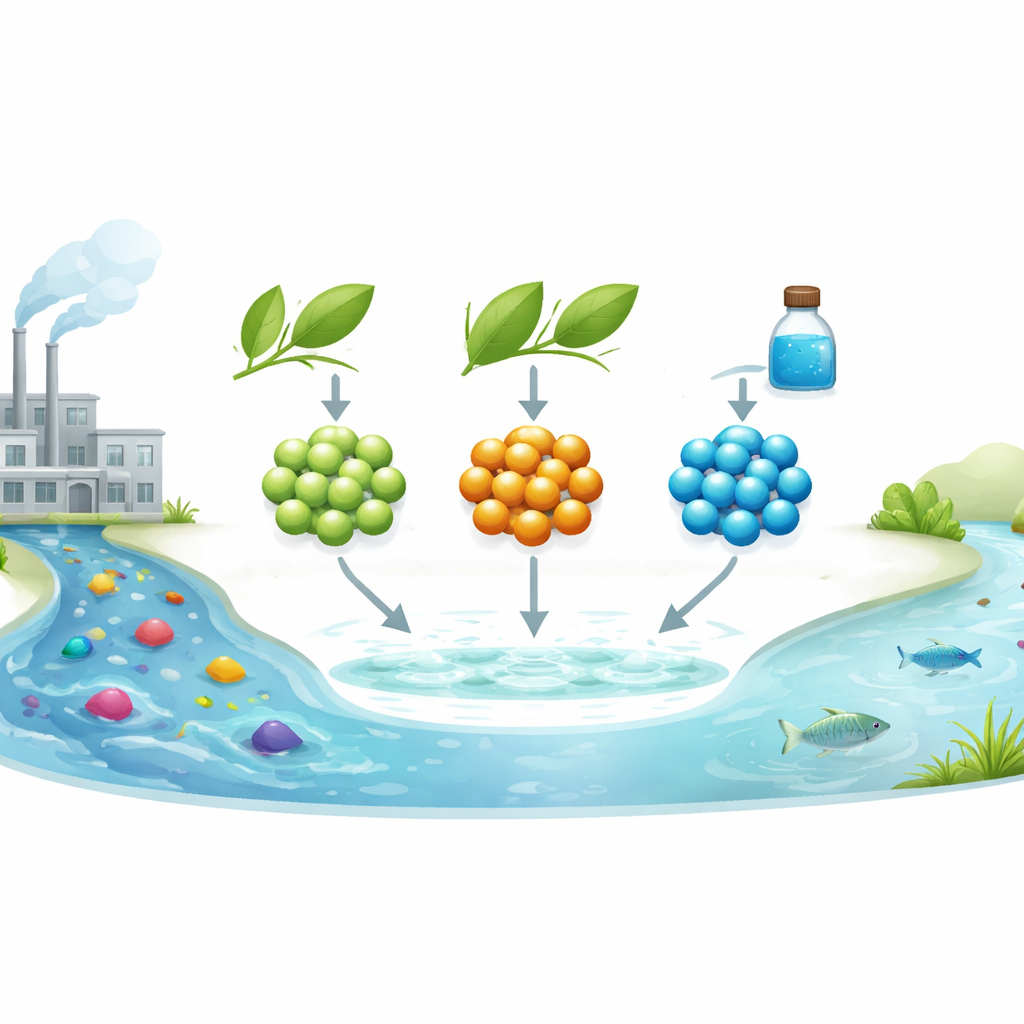
Why Colored Wastewater Is Hard to Treat
Industries such as textiles, paper, and plastics release large amounts of colored wastewater. Many of the dyes they use are designed to resist fading, which also makes them resistant to natural breakdown. Traditional treatment methods often struggle to remove these stubborn molecules completely. A promising alternative is photocatalysis: using light-activated materials that generate highly reactive species able to rip dyes apart into smaller, safer components. The challenge is to create such materials in a way that is both effective and environmentally friendly.
Tiny Particles Built with Desert Plants
The researchers focused on zinc selenide nanoparticles—specks many thousands of times smaller than the width of a human hair. These particles can absorb light and drive chemical reactions. Usually, they are made and stabilized using synthetic molecules like L-cysteine, which help control their size but come from more complex chemical routes. In this work, the team instead used water extracts from two plants common in arid regions, Artemisia herba-alba and Calligonum, to both form and “cap” the nanoparticles. The plant components attach to the particle surface, shaping how big they grow and how they behave in water.
How Plant Coatings Change the Nanoparticles
To understand what they had made, the scientists used a suite of techniques to look at the particles’ crystal structure, shape, and light response. They found all three versions—L-cysteine capped, Artemisia capped, and Calligonum capped—were only a few nanometers across and formed mainly in a hexagonal crystal form, with a small fraction of a cubic form. Under the microscope, the particles made with the synthetic chemical were the smallest and most uniform, while the plant-made particles were slightly larger and less regular in shape. Optical measurements showed that all samples absorbed light more strongly at shorter wavelengths than bulk zinc selenide, a signature of their very small size. However, the plant-coated particles showed more complex light emission patterns, revealing additional “defect” sites and surface states created by the plant compounds on the particle surface.
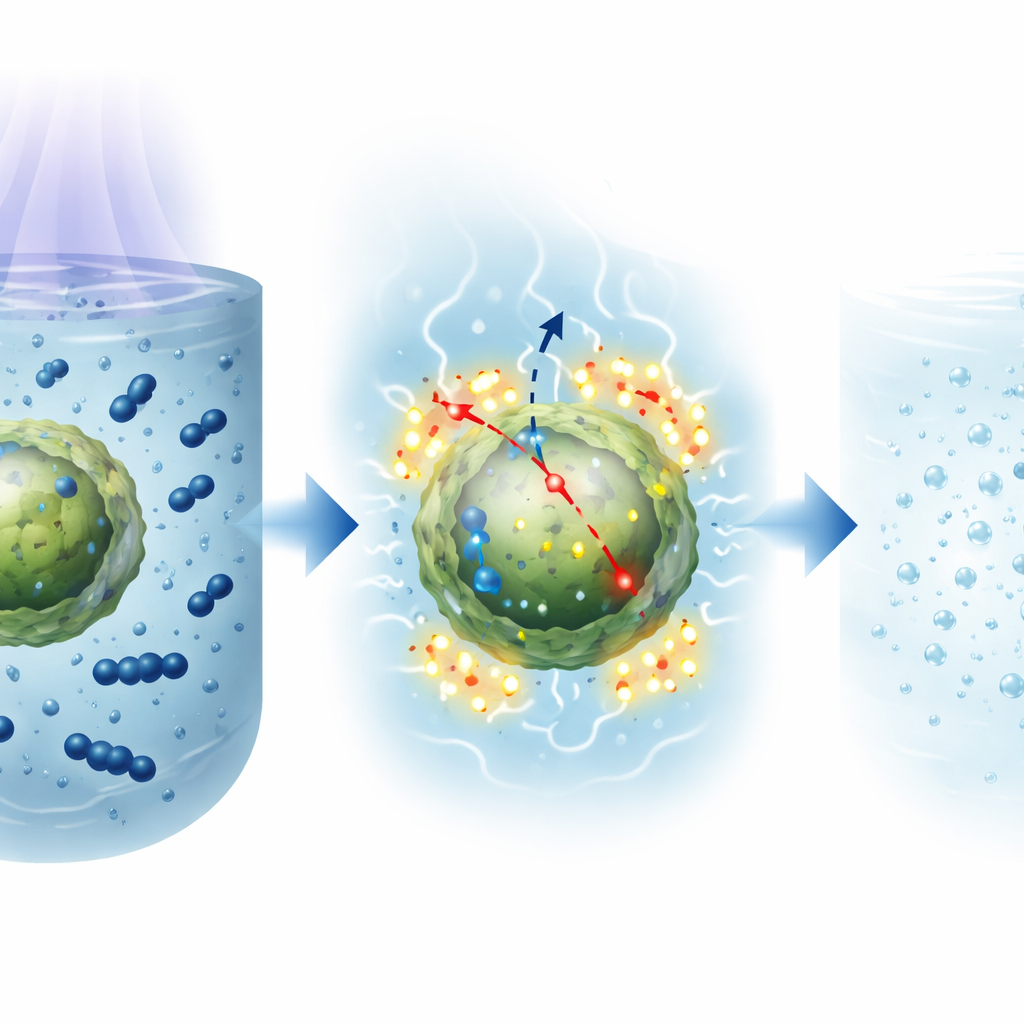
Cleaning a Model Dye from Water
The team then tested how well these nanoparticles could break down methylene blue, a common blue dye often used as a stand-in for real industrial pollutants. They mixed a small amount of each type of nanoparticle with dye-contaminated water and shone ultraviolet light on the mixture. Over three hours, the characteristic blue color steadily faded. The Calligonum-coated particles removed about 40% of the dye, slightly better than the synthetic L-cysteine-coated ones (38%) and clearly more than the Artemisia-coated version (28%). Interestingly, when they analyzed how fast the reaction proceeded, the Artemisia-coated particles showed the highest reaction rate, meaning that once dye molecules reached their surface, they were broken down particularly efficiently. The lower overall removal for Artemisia was linked to weaker initial dye adsorption on its particle surfaces.
How Defects and Plant Molecules Boost Performance
The detailed light-emission studies suggest why the plant-based coatings work so well. The phytochemicals from the extracts—phenols, flavonoids, tannins, and related molecules—introduce controlled imperfections in the particles and form a thin organic shell around them. These features create a variety of energy sites that temporarily trap electrons and holes generated by light. Instead of quickly recombining and wasting their energy as simple light emission, these separated charges live long enough to react with water and oxygen, producing aggressive “oxygen radicals” that attack and break down the dye molecules. In the Calligonum-coated particles, abundant surface defects and good adsorption of dye appear to combine for strong overall cleaning ability, while Artemisia creates especially effective reaction sites but adsorbs less dye to begin with.
From Lab Concept to Cleaner Water
In plain terms, this work shows that simple water extracts from hardy desert plants can replace synthetic chemicals in making powerful, light-driven cleaning agents for polluted water. The plant-coated zinc selenide particles are not only greener to produce, they also perform as well as—or in some ways better than—conventionally made particles in breaking down a stubborn dye. By tuning the natural mixtures used for capping, it may be possible to design low-cost, scalable nanomaterials that help treat industrial wastewater, limit the spread of toxic dyes, and even find use in antibacterial surfaces and solar-driven energy applications.
Citation: Alshammari, A.F., Ouni, S., Bouzidi, M. et al. Green synthesis and enhanced photocatalytic activity of ZnSe nanoparticles capped with Artemisia herba-alba and calligonum plants extracts. Sci Rep 16, 8674 (2026). https://doi.org/10.1038/s41598-026-39998-z
Keywords: green photocatalysis, plant-based nanoparticles, wastewater treatment, zinc selenide nanomaterials, dye degradation