Clear Sky Science · en
Mechanistic and electrochemical investigation of solar light driven organic pollutant degradation using SrFe12O19/NiO Z-scheme heterojunctions
Cleaning Water with Sunlight
Many of the bright dyes in our clothes and the painkillers in our medicine cabinets eventually end up in rivers and lakes, where they can harm fish, wildlife, and even human health. This study explores a sunlight‑powered material that can help scrub two troublesome pollutants—Rhodamine B dye and the common drug ibuprofen—from water. By combining two tiny crystalline solids into a single, smarter particle, the researchers show how we might harness ordinary sunlight to break down stubborn chemicals more efficiently and more sustainably.
Why Everyday Chemicals Linger in Water
Modern industry and healthcare rely on synthetic dyes and pharmaceuticals that are designed to be stable. That stability becomes a problem once these molecules are washed down the drain. Conventional treatment plants struggle to remove them completely, so traces of dyes like Rhodamine B and drugs like ibuprofen are now routinely detected in wastewater and natural waters. Rhodamine B is not just vivid pink; it is linked to nerve damage and respiratory illness, while ibuprofen can disrupt aquatic life over time. Methods such as filtration or adsorption can move these pollutants around but often create new waste streams. A more attractive approach is to break the molecules apart using light‑driven chemistry, turning them into carbon dioxide, water, and other simple species.
Building a Sun-Activated Cleaning Particle
To do this, the team created a new photocatalyst—a material that speeds up chemical reactions when exposed to light—by joining two different metal oxides at the nanoscale: strontium hexaferrite (SrFe12O19, called SFO) and nickel oxide (NiO). On their own, each material can absorb light and generate charges, but they lose much of that energy because electrons and holes quickly recombine. By carefully co‑precipitating the ingredients from solution and then heating them, the researchers made nanometer‑sized particles in which NiO coats or nestles around hexagonal grains of SFO, forming a so‑called Z‑scheme junction. Microscopy, X‑ray diffraction, and surface analysis confirmed that the two crystals sit in intimate contact, while optical measurements showed that the combined material absorbs a broader slice of sunlight and has a narrower effective band gap than either component alone.
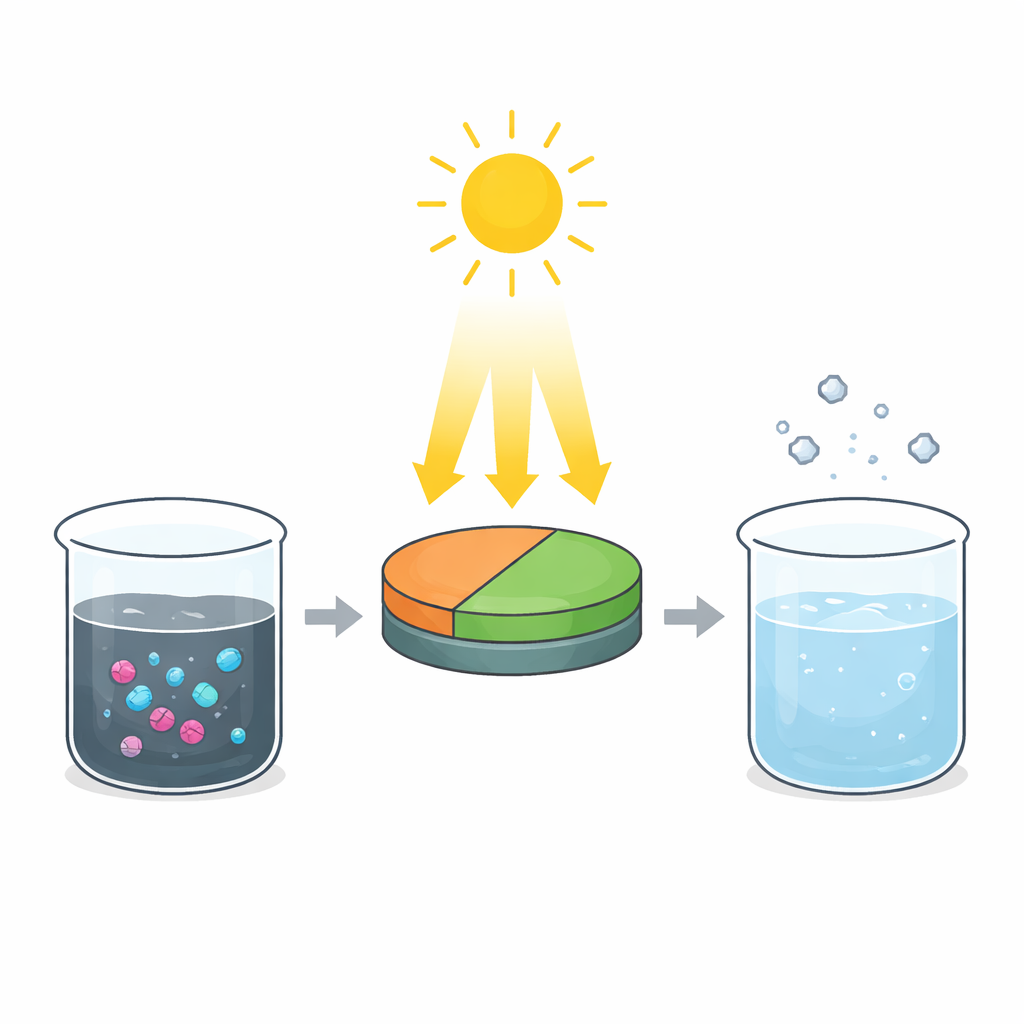
How the New Material Uses Light More Wisely
The key advance lies in how the joined crystals handle the charges produced by sunlight. In many traditional designs, charges simply flow “downhill,” which keeps electrons and holes apart but weakens their chemical punch. In the Z‑scheme arrangement here, low‑energy electrons from SFO recombine with low‑energy holes from NiO right at the interface, leaving the most energetic electrons on the NiO side and the most energetic holes on the SFO side. These high‑energy charges live long enough to react with oxygen and water at the surface, forming highly reactive oxygen‑based species that can attack and dismantle dye and drug molecules. Measurements of light emission from the particles and electrical tests both supported this picture, showing reduced charge recombination and altered current flow when the two oxides work together.
Putting the Photocatalyst to Work
The researchers then tested how well their particles cleaned water under natural sunlight, using Rhodamine B dye and ibuprofen as stand‑ins for broader classes of pollutants. By tuning practical factors like the amount of catalyst, water acidity, pollutant concentration, and exposure time, they found conditions where the composite destroyed about 93% of a moderately concentrated Rhodamine B solution in 100 minutes, and 75% of a dilute ibuprofen solution in 120 minutes. Careful tracking of carbon content in the water revealed that the dye, in particular, was not just faded but largely mineralized to simple inorganic products. Chemical “trap” experiments showed that reactive holes and superoxide radicals were the main species breaking down the molecules, with hydroxyl radicals playing a supporting role. Importantly, the particles could be filtered out and reused several times, with only a gradual loss of performance and minimal leaching of nickel into the water.

Promise and Next Steps for Sunlit Water Treatment
For non‑specialists, the bottom line is that this work demonstrates a small, solid material that can use everyday sunlight to convert hazardous dye and drug residues into far less harmful substances, without needing extra chemicals or electricity. By engineering how two familiar oxides share and shuttle light‑generated charges, the authors achieve stronger and more selective reactions than either material can deliver alone. Although performance still declines over repeated cycles and real wastewater is more complex than laboratory test solutions, the approach points toward compact, magnetically recoverable photocatalysts that could sit in sunlight‑exposed reactors or treatment ponds, quietly helping to clean up the chemical footprint of modern life.
Citation: Pattanaik, R., Kamal, R., Pradhan, D. et al. Mechanistic and electrochemical investigation of solar light driven organic pollutant degradation using SrFe12O19/NiO Z-scheme heterojunctions. Sci Rep 16, 8473 (2026). https://doi.org/10.1038/s41598-026-39997-0
Keywords: solar photocatalysis, wastewater treatment, nanocomposite catalyst, organic pollutant degradation, Z-scheme heterojunction