Clear Sky Science · en
High-performance Au–MoS₂–graphene multilayer SPR biosensor with superior sensitivity and precision
Why this tiny sensor matters
Doctors, environmental scientists, and even food safety inspectors all need tools that can spot trace amounts of chemicals or biomolecules quickly and reliably. This paper presents a new type of optical sensor, built from ultra-thin layers of gold and advanced 2D materials, that can detect extremely small changes in liquid samples such as blood, sugar solutions, or water-based buffers. By squeezing light into a nanometer-scale region at a metal surface, the device promises faster, more precise tests for disease markers and pollutants.
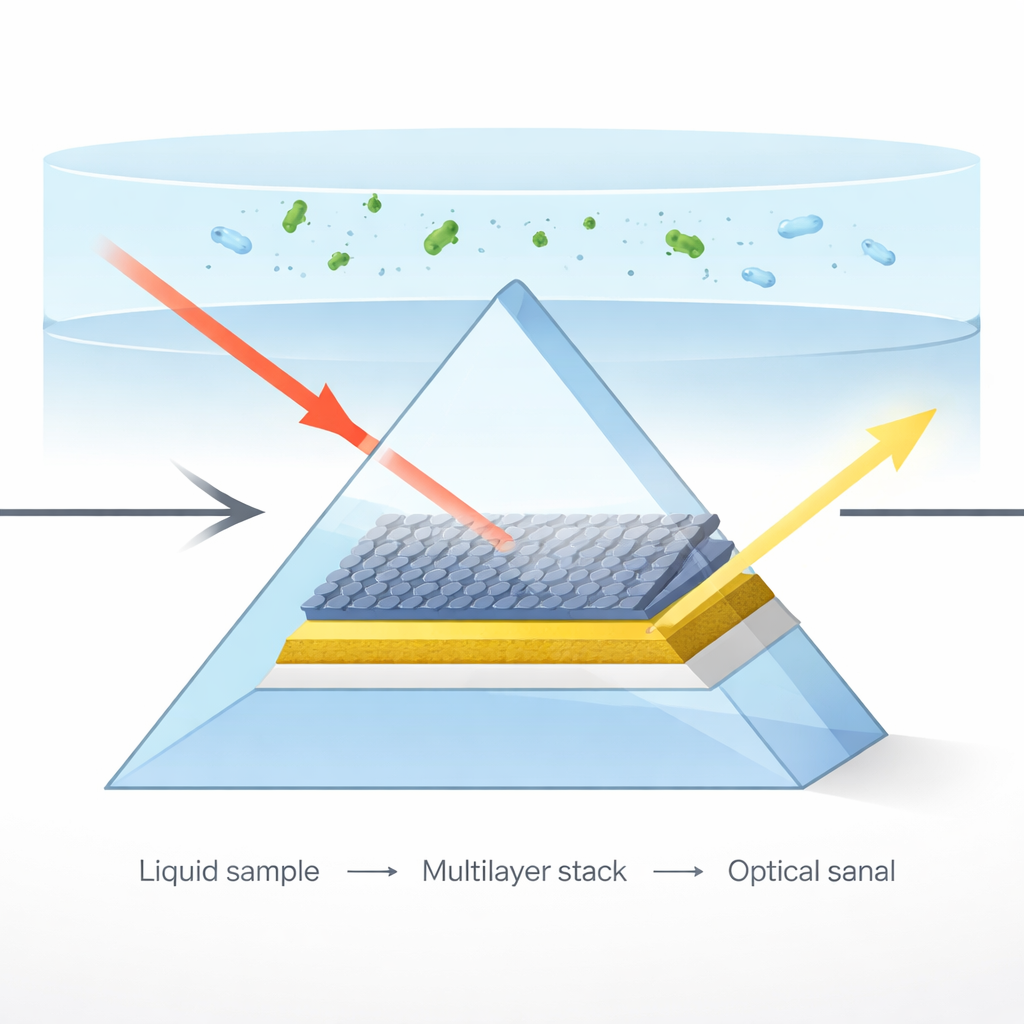
Listening to light at a surface
The sensor relies on a phenomenon called surface plasmon resonance, where light skims along a metal surface and couples to ripples of electrons at that boundary. When a liquid sample touches this surface, even a tiny change in the liquid’s optical properties shifts the angle at which the resonance occurs. By shining a laser through a glass prism onto a thin gold film and monitoring the reflected light, the device can translate these shifts into information about what is dissolved in the liquid, such as sugar concentration or changes in blood composition.
Stacking smart materials for stronger signals
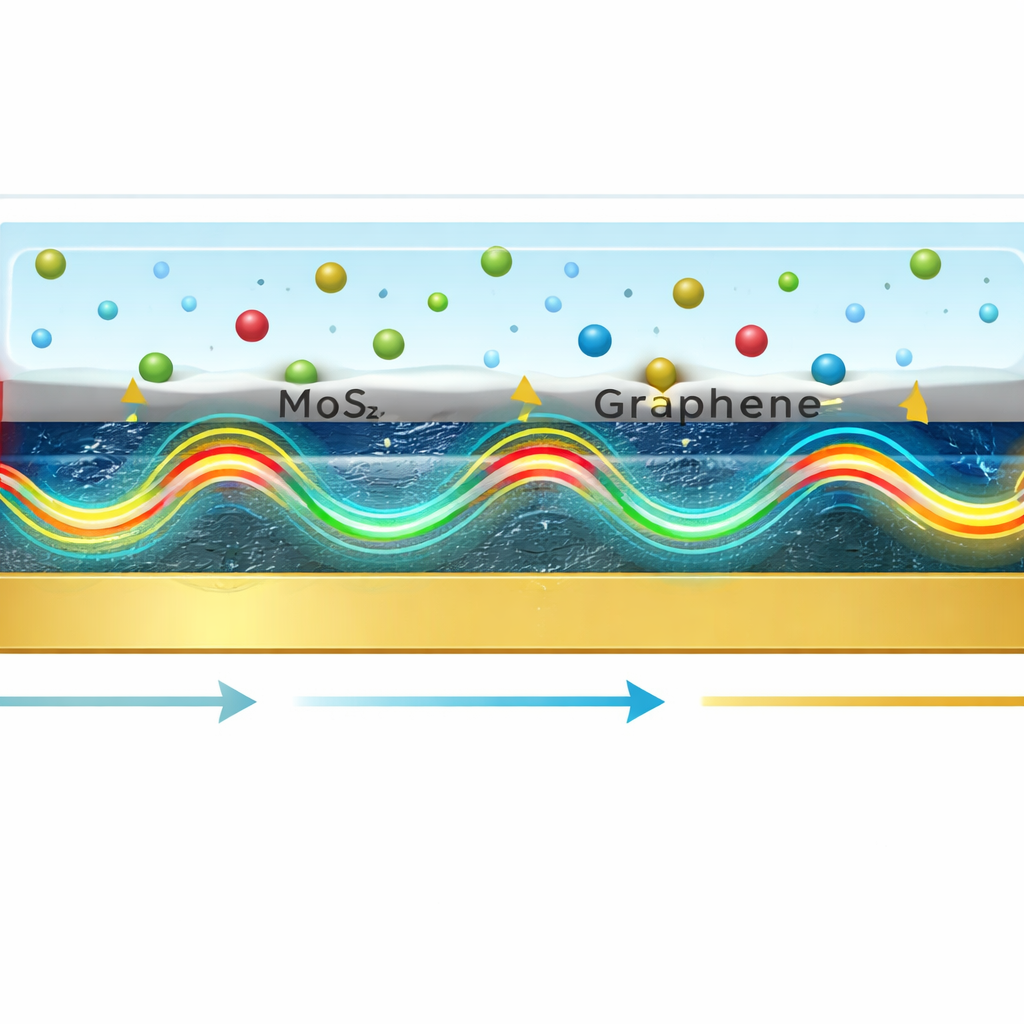
Instead of using bare gold alone, the researchers designed a multilayer stack: a glass prism, a gold film, a few atomic layers of molybdenum disulfide (MoS₂), and three layers of graphene, with the test liquid sitting on top. Each material has a specific job. Gold efficiently launches the surface ripples of electrons. MoS₂, with its high optical density and strong interaction with light, squeezes and concentrates the electromagnetic field near the surface. Graphene adds enormous surface area for molecules to stick to and excellent electrical and optical properties that further strengthen the light–matter interaction. Computer simulations revealed that an arrangement with five MoS₂ layers and three graphene layers produced the most intense and sharply confined field at the sensing surface.
Turning tiny changes into clear readings
To judge performance, the team simulated how the reflected light angle shifts when the sensor is exposed to different liquids: standard laboratory buffer (PBS), sucrose solution, blood, and glycerol. They calculated several figures that together describe how well the sensor performs—how large the angle shift is for a given change in the liquid, how narrow and sharp the resonance dip appears, and how precisely small shifts can be distinguished. Across all tested samples, the sensor achieved sensitivities close to 80 degrees per refractive index unit, with especially high precision for sucrose and glycerol, where the resonance features were very narrow. These numbers match or improve upon many previously reported multilayer sensors, showing that the three-material stack offers real gains in both sensitivity and measurement clarity.
Tuning the layers for best performance
The authors also explored how changing the thickness of the graphene and MoS₂ layers affects the signal. Very thin graphene brings the confined light field close to the liquid, increasing sensitivity, while making it too thick pushes the field inward and weakens the response. A similar balance appears with MoS₂: adding a few layers boosts field confinement, but beyond an optimal thickness, extra absorption damps the resonance and broadens the signal. The simulations further examined how different illumination angles and wavelengths influence sensitivity, identifying ranges where the device responds most strongly to small changes in the sample.
From computer model to real-world tests
Although this work is based on numerical modeling, the authors discuss realistic fabrication pathways using standard techniques for depositing gold and growing or transferring MoS₂ and graphene. They highlight practical challenges—such as keeping surfaces extremely smooth, controlling layer thickness to within a few nanometers, and ensuring that the fragile 2D materials do not wrinkle, oxidize, or peel. With careful control over these steps and integration into microfluidic channels for handling tiny volumes of liquid, they argue that the multilayer sensor could be built and used in the lab.
What this means for everyday applications
In simple terms, this study shows how carefully stacking three advanced materials can turn a familiar optical effect into a highly capable chemical “ear,” able to hear faint whispers of change in complex liquids. The gold layer launches the signal, MoS₂ amplifies and focuses it, and graphene provides a welcoming surface for target molecules, together producing sharper, more sensitive readings than many earlier designs. If realized experimentally, such sensors could help doctors detect disease markers earlier, allow faster quality checks in the food and pharmaceutical industries, and enable portable systems for tracking water pollution—all by watching how a narrow beam of light reflects from an engineered surface only a few nanometers thick.
Citation: Bahmani, E., Kaatuzian, H. & Shafagh, S.G. High-performance Au–MoS₂–graphene multilayer SPR biosensor with superior sensitivity and precision. Sci Rep 16, 8428 (2026). https://doi.org/10.1038/s41598-026-39993-4
Keywords: surface plasmon resonance, biosensor, graphene, MoS2, refractive index sensing