Clear Sky Science · en
“Salvia officinalis extract–conjugated magnetite and selenium nanocomposites showed enhanced antibacterial and anti-biofilm activity against multidrug-resistant pathogens”
Why this matters for everyday health
Doctors are increasingly running out of options when bacteria stop responding to common antibiotics. These hard-to-kill microbes often hunker down in slimy communities called biofilms on medical devices, wounds, or tissues, where drugs and the immune system struggle to reach them. This study explores an unconventional strategy that teams up an everyday culinary herb—sage—with ultra-small particles of iron and selenium to create a new line of defense against these stubborn infections.
The rise of hard-to-treat germs
The researchers began by testing how six disease-causing bacteria reacted to a panel of standard antibiotics. Three of these were so-called Gram-positive species and three were Gram-negative, a group known for being especially tough. Most drugs failed dramatically. Many bacteria resisted the majority of antibiotics offered, with some shrugging off more than 90 percent of the medicines tested. Only one powerful hospital antibiotic, meropenem, was able to slow or stop all of them—and even then, its strength against protective biofilms was limited. These findings echo a global pattern: overuse and misuse of antibiotics have helped breed strains that routine treatments can no longer control.
Turning kitchen sage into a stronger weapon
To look for alternatives, the team focused on Salvia officinalis, better known as common sage, a herb long used in cooking and traditional remedies. A simple water-based extract of dried sage leaves showed modest ability to inhibit both Gram-positive and Gram-negative bacteria, working better on the former. But on its own, very high doses of the extract were needed to have an effect. To boost its power, the scientists paired the extract with two kinds of tiny metal particles: magnetite (an iron oxide) and selenium. These nanoparticles, made through a “green” method using vitamin C as a reducing agent, are only a few billionths of a meter across and carry special surface charges that help them interact with biological molecules.
Building herbal–nanoparticle hybrids
The next step was to mix sage extract with each type of nanoparticle, forming two hybrid materials. Detailed measurements showed that the plant molecules coated the particles, creating stable, well-dispersed complexes. The iron-based hybrids tended to be very uniform in size, while the selenium-based ones carried a richer mix of plant compounds, including flavonoids and phenolic acids known for antimicrobial and antioxidant effects. Both hybrids carried strong negative surface charges, which helps keep them from clumping and may also influence how they touch and penetrate bacterial surfaces and biofilms.
Putting the new materials to the test
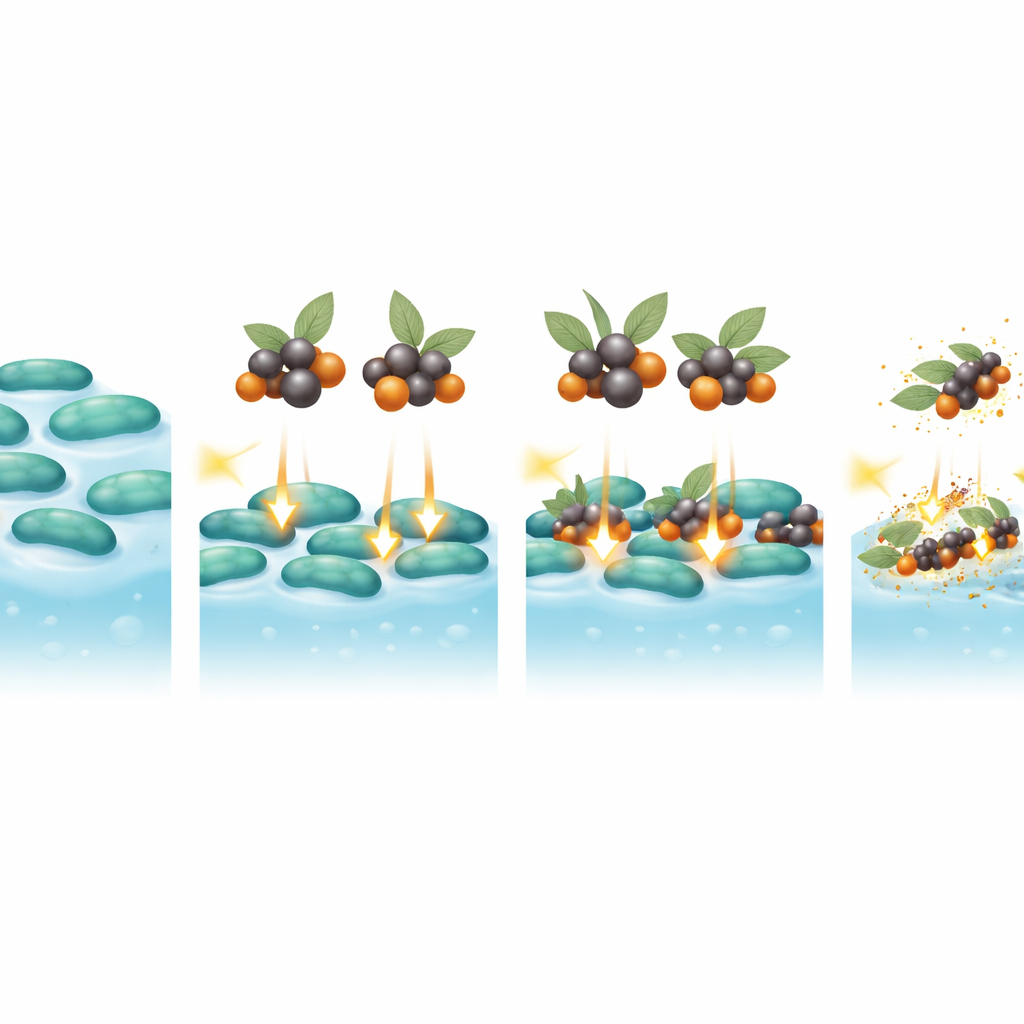
When the team compared plain sage extract, bare nanoparticles, and the two hybrids, the differences were striking. The hybrids needed far lower amounts to stop bacterial growth—often dozens of times less than the individual components alone. In some cases, the minimum amount needed was comparable to, or even lower than, that of meropenem. The selenium–sage hybrid generally performed best, especially against difficult Gram-negative species. Over a 24-hour period, both hybrids sharply reduced bacterial growth compared with untreated cultures or those exposed only to nanoparticles. Electron microscope images revealed that treated bacteria showed ruptured walls, leaking interiors, and distorted shapes, far more severe damage than seen with the antibiotic control.
Breaking down stubborn biofilms
A particularly encouraging finding was the hybrids’ effect on biofilms. At concentrations twice the minimum needed to halt growth, the sage–nanoparticle hybrids cut biofilm formation by roughly one-third to more than one-half, often outperforming meropenem. This was true for both Gram-positive and Gram-negative bacteria, though the latter remained somewhat more resilient. The results suggest that the hybrids can penetrate the protective slime layer, interfere with its structure, and attack the bacteria both outside and inside the biofilm. The combination of plant chemicals and reactive metal surfaces appears to act through multiple overlapping routes, making it harder for bacteria to adapt.
What this could mean for future treatments
Overall, the study shows that fusing a familiar medicinal herb with engineered nano-sized particles can create potent new agents against drug-resistant bacteria and their biofilms. While these findings are still from laboratory tests and not yet ready for the clinic, they point to a promising direction: using safe plant extracts to guide and strengthen nanomaterials, producing therapies that rival or even beat last-resort antibiotics in some situations. With further safety testing and animal studies, such herbal–nanoparticle blends could one day help doctors treat stubborn infections, protect medical devices, and extend the useful life of existing antibiotics.
Citation: Enan, G., El-Wafa, N.A., El-Saber, M.M. et al. “Salvia officinalis extract–conjugated magnetite and selenium nanocomposites showed enhanced antibacterial and anti-biofilm activity against multidrug-resistant pathogens”. Sci Rep 16, 9201 (2026). https://doi.org/10.1038/s41598-026-39983-6
Keywords: antibiotic resistance, biofilms, nanoparticles, sage extract, selenium nanocomposites