Clear Sky Science · en
Skipjack tuna bone derived biocalcium ameliorates C2C12 myotube atrophy through microRNA29b regulation
From Fish Bones to Stronger Muscles
As people age, many quietly lose muscle mass and strength, a condition called sarcopenia that raises the risk of falls, frailty, and loss of independence. At the same time, the seafood industry generates tons of nutrient-rich bone waste that is usually discarded. This study brings those two problems together and asks a surprising question: could finely processed calcium from skipjack tuna bones help protect muscle cells from shrinking and weakening?
Why Muscle Loss Matters
Sarcopenia is driven by more than just inactivity. Aging muscles often live in a constant low-grade inflammatory state, with chemical messengers that nudge cells toward breaking down their own proteins faster than they can rebuild them. Certain medicines, such as the steroid dexamethasone, can mimic or worsen this process and are widely used in research to trigger muscle wasting in the lab. The authors used mouse muscle cells grown in dishes and exposed them to dexamethasone to recreate this kind of atrophy and then tested whether a tuna bone–derived biocalcium powder could shield the cells from harm.
A New Type of Calcium
Unlike typical calcium pills made from simple mineral salts, the skipjack tuna biocalcium used here is a complex mix of natural bone minerals plus collagen and small protein fragments. These extra components help keep calcium soluble and easier to absorb and have already been shown to support bone health in animal studies. The powder is produced by cleaning, defatting, bleaching, and finely milling tuna bones that are normally discarded after canning, turning a waste stream into a concentrated source of bioactive nutrients. This background motivated the researchers to explore whether the same preparation that benefits bone might also help preserve muscle.
Protecting Muscle Cells in the Lab
In the dish, dexamethasone caused the muscle cells to thin out, a clear sign of atrophy. When the researchers added low to moderate doses of tuna biocalcium, the cells maintained a larger diameter and healthier appearance without signs of toxicity. The treatment also reduced nitric oxide and several inflammatory molecules that had been pushed up by dexamethasone. Inside the cells, markers that drive protein breakdown fell, while a central growth controller, mTOR, was nudged in the opposite, protein-building direction. Together these changes point to a broad shift away from self-cannibalization and toward maintenance and repair.
Silencing a Small but Powerful Switch
A major focus of the study was a tiny regulatory molecule called microRNA-29b, which has emerged as a key promoter of muscle wasting in many disease states. MicroRNA-29b normally dampens pathways that help muscle cells grow and regenerate. Dexamethasone pushed this molecule upward, but tuna biocalcium drove its levels down at several stages of its formation. When the team artificially boosted microRNA-29b, muscle-building genes dropped; when they blocked it, those genes rose again, even in the presence of the drug. Computer modeling further suggested that microRNA-29b could physically interact with important muscle-regulating proteins, reinforcing the idea that this small molecule sits at a critical control point that the biocalcium can indirectly influence.
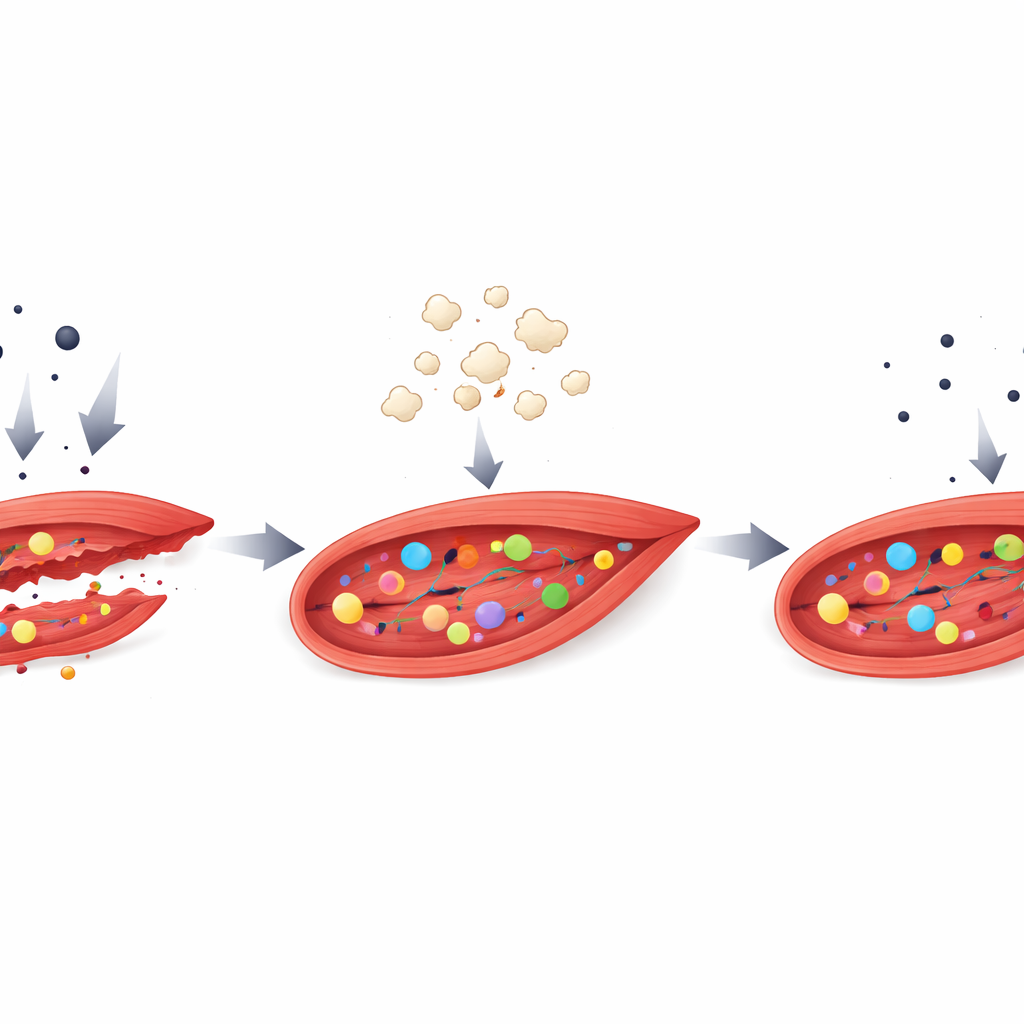
How All the Signals Tie Together
The researchers also examined broader signaling routes known to tip the balance between muscle growth and breakdown. Dexamethasone activated pathways linked with inflammation and stress and increased a factor that switches on protein-degrading genes. Biocalcium treatment generally reduced these stress signals and increased a counteracting growth pathway centered on Akt and mTOR, although the authors note that some of these measurements were limited by technical challenges. Even so, the overall pattern—less inflammation, less protein destruction, more support for rebuilding—matched the more direct readouts of larger, healthier-looking muscle fibers in the cultures.
What This Could Mean for People
In simple terms, the study suggests that a carefully prepared calcium powder from tuna bones can help muscle cells resist a strong atrophy-inducing drug by calming inflammatory signals, restoring protein balance, and turning down a small RNA that pushes muscles toward wasting. While the work was done in isolated cells rather than in people, and more animal and human trials are needed, it points to skipjack tuna biocalcium as a promising candidate for a functional food supplement. If its effects translate beyond the lab, this upcycled marine ingredient could one day help older adults preserve muscle mass and strength while also reducing seafood processing waste.
Citation: Jantarawong, S., Senphan, T., Youngruk, C. et al. Skipjack tuna bone derived biocalcium ameliorates C2C12 myotube atrophy through microRNA29b regulation. Sci Rep 16, 8429 (2026). https://doi.org/10.1038/s41598-026-39977-4
Keywords: sarcopenia, muscle atrophy, biocalcium, microRNA-29b, marine nutraceuticals