Clear Sky Science · en
Embigin is involved in the regulation of early mouse kidney development
Why tiny kidneys matter
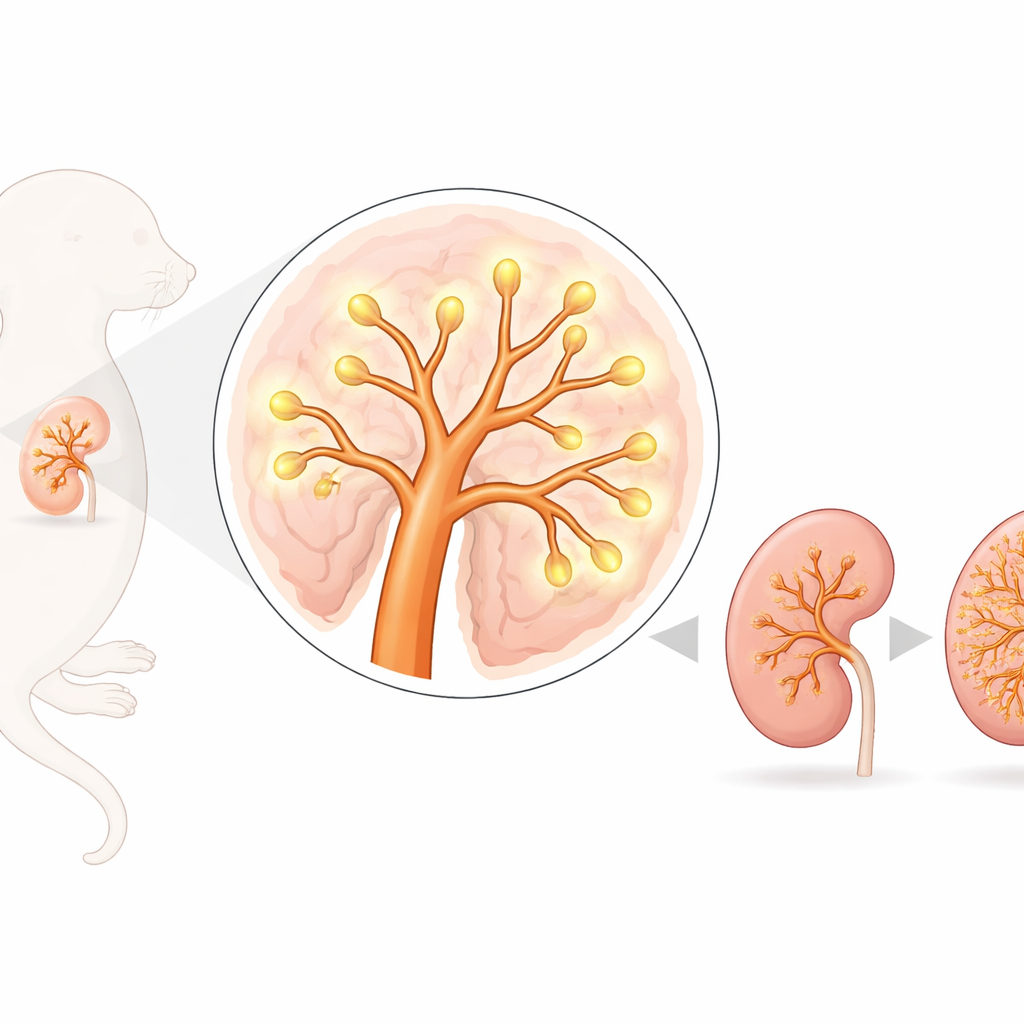
Before a baby is born, its organs go through an intricate building process where cells must divide, move, and connect in exactly the right way. The kidneys, which filter blood and keep our body’s salt and water in balance, are assembled from branching tubes that grow out like the limbs of a tree. This study explores a little-known surface protein called embigin and shows that it quietly helps guide the early shaping of the mouse kidney—especially how those first branches form—offering clues to how organ growth is coordinated and what might go wrong in disease.
A helper at the cell surface
Embigin sits on the outer membrane of cells and is known to assist certain transport proteins that move small metabolic molecules in and out of cells. It can also latch onto components of the tissue scaffold that surrounds cells. Earlier work showed that mice lacking embigin struggle with lung development and often die soon after birth, hinting that this protein is important for building organs. Here, the researchers asked whether embigin plays a similar role in the kidney, where development begins with a simple outgrowth that repeatedly splits to form the future urine-collecting system and triggers the formation of filtering units called nephrons.
Guiding early kidney branching
Using fluorescent microscopy, the team found that embigin is strongly present in the early kidney’s branching tubes—known as the ureteric bud—and in cells that are just starting to turn into nephrons, but not in the most primitive kidney stem cells that cap the growing tips. When they cultured embryonic kidneys from normal and embigin-deficient mice at an early stage, the difference was striking: kidneys lacking embigin produced far fewer branch tips, and the overall kidneys were smaller later in gestation. However, when kidney size was compared with overall body size, the ratios were similar, suggesting that embigin contributes to general growth as well as kidney-specific shaping.
Delayed, not destroyed development
To see what was happening at the molecular level, the researchers measured gene activity in developing kidneys. At a mid-gestation time point, kidneys without embigin showed reduced activity in dozens of genes tied to building nephrons, shaping tubes, controlling blood pressure, and handling body fluids. Several of the most affected genes are crucial for specialist cells called podocytes, which form part of the kidney’s filtration barrier. Yet by a later stage, these gene expression differences had largely disappeared and kidney structure looked normal under the microscope. Adult mice that survived without embigin did not leak protein into their urine and had no obvious structural kidney defects, pointing to a temporary developmental lag rather than permanent damage.
Shared signals across tissues
The team then turned to a mouse epithelial cell line, silencing embigin to see which genes changed. Even though these cells were not from the kidney, loss of embigin again altered many genes involved in organ shaping, tube formation, and kidney-related pathways. Comparing the cell line data with the early kidney data highlighted three genes—Pappa2, Acta2, and Tagln—that were consistently reduced when embigin was missing. These genes have all been linked to kidney development, blood vessel tone, and cell movement, processes that depend on how cells sense and grab onto their surrounding matrix. Reanalysis of single-cell RNA sequencing datasets from very early embryos showed that embigin is already active in the earliest kidney precursor cells, reinforcing the idea that it supports key decisions early in organ formation.
What this means for organ building
Taken together, the findings present embigin as a subtle but important organizer of early kidney growth. It does not appear to be absolutely required for a working kidney—other proteins can eventually compensate—but it helps ensure timely branching of the early tubular tree and proper activation of gene networks that shape nephrons and blood-filtering structures. For a lay reader, the message is that seemingly obscure cell-surface proteins can have broad effects on how organs take shape, and that understanding these hidden helpers may one day improve our insight into congenital kidney problems and guide strategies to grow or repair kidney tissue.
Citation: Talvi, S., Jokinen, J., Rappu, P. et al. Embigin is involved in the regulation of early mouse kidney development. Sci Rep 16, 8403 (2026). https://doi.org/10.1038/s41598-026-39966-7
Keywords: kidney development, embigin, branching morphogenesis, stem cells, organ formation