Clear Sky Science · en
High-capacity removal of crystal violet using ZIF-8/graphene quantum dot composite with RSM optimization and explainable machine learning
Why This Matters for Everyday Water Safety
Synthetic dyes give our clothes, plastics, and lab tools their vivid colors, but once they end up in rivers or groundwater, they can pose serious risks to people and aquatic life. One such dye, Crystal Violet, is toxic and can linger in the environment for a long time. This study explores a new material that can pull astonishing amounts of this dye out of water and shows how data-driven tools can help fine-tune the cleanup process.
A New Sponge for Toxic Color
The researchers focused on Crystal Violet, a bright purple dye widely used in textiles, inks, and biology labs, but known for being harmful and difficult to remove from water. They built on two advanced materials: ZIF-8, a highly porous crystal made from zinc and organic linkers, and graphene quantum dots, tiny carbon fragments with a large reactive surface. By combining them into a single composite called Z8GD, they hoped to create a sort of “super sponge” that could trap dye molecules more effectively than either material alone.
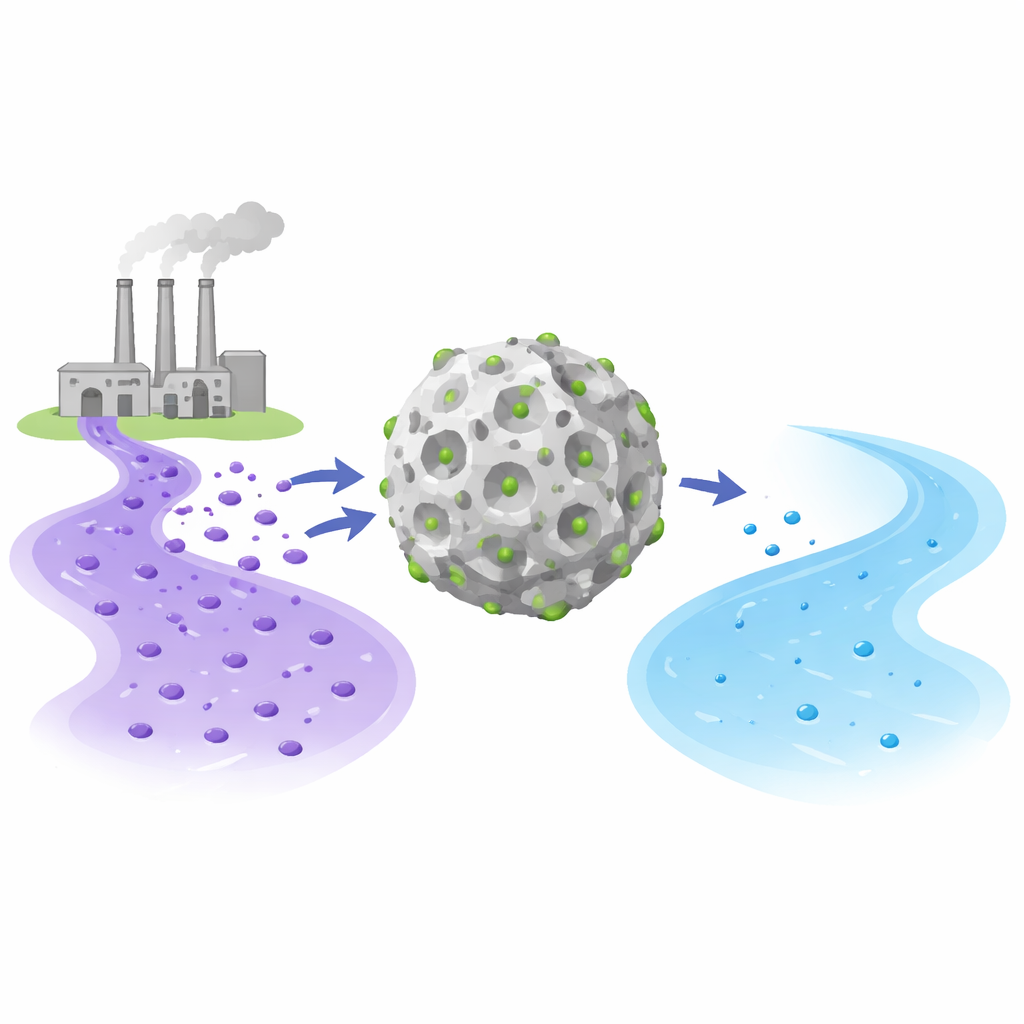
How the Clean-Up Conditions Shape Performance
To test their new composite, the team ran a series of batch experiments in flasks of dye-contaminated water. They systematically varied three practical knobs an engineer can turn: how much of the material to add, how strong the dye solution was at the start, and how long the mixture was shaken. Using a statistical technique called response surface methodology, they created a predictive map of how these factors influence dye capture. They found that using a smaller amount of the material actually gave a higher uptake per gram, stronger starting dye solutions drove more dye onto the surface, and longer shaking times dramatically boosted the amount removed. Across the tested conditions, the material’s performance swung from modest to extremely high, showing both great promise and strong sensitivity to how it is used.
What Happens at the Tiny Scale
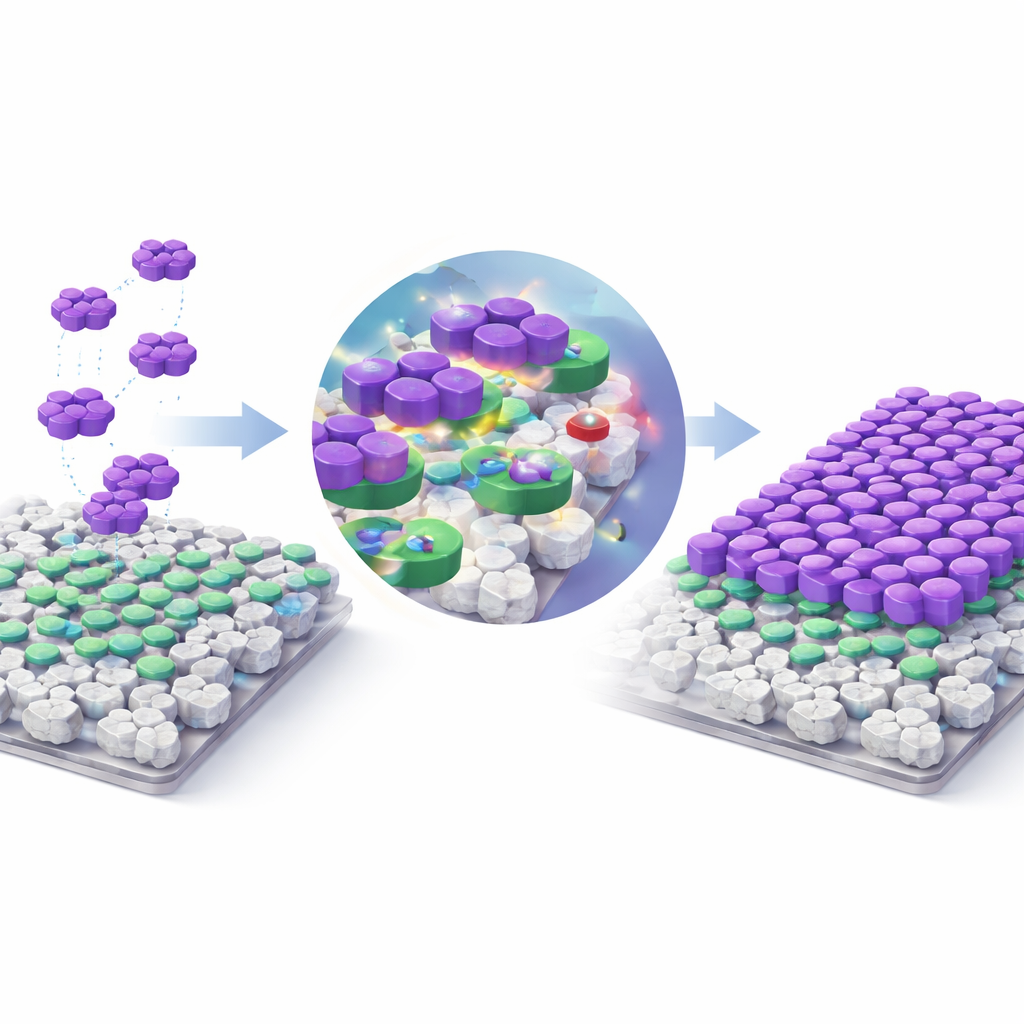
To understand why Z8GD works so well, the researchers probed it before and after dye removal using X-ray diffraction and infrared spectroscopy, techniques that reveal structural and chemical changes. The core crystal framework stayed intact, meaning the material behaved like a reusable scaffold rather than dissolving or falling apart. New signals in the spectra pointed to several cooperative interactions: flat dye molecules stacking against the carbon-rich surfaces, hydrogen bonds forming between dye groups and surface oxygen atoms, and attractive forces between positively charged dye and negatively charged sites on the composite. Together, these effects pack the dye densely onto the outer surfaces and pores of the material, leading to an exceptionally high experimental capacity of about 7,000 milligrams of dye per gram of adsorbent—far higher than many other reported materials.
Letting Data Science Guide the Process
Instead of relying only on trial and error, the authors combined their laboratory results into a single dataset and trained several machine-learning models to predict how much dye would be captured under new conditions. A hybrid model that merged support vector regression with a boosting algorithm proved the most accurate. To avoid having a “black box” predictor, they used an explainability tool known as SHAP to see which inputs mattered most. This analysis confirmed that contact time and the starting dye concentration were the main drivers of performance, while adding too much material could actually reduce how efficiently each gram was used, likely because particles clump and block each other’s active sites.
What This Means for Future Water Treatment
In plain terms, the study shows that the Z8GD composite is an unusually powerful filter for a dangerous purple dye, able to lock away huge amounts without breaking down. It also demonstrates that combining careful experiments with modern machine learning can reveal the best operating conditions and explain why they work, not just that they work. While real-world wastewater is more complex than the test solutions used here, and long-term reuse still needs to be proven, this approach points toward smarter, more efficient design of next-generation materials and processes to keep our water cleaner and safer.
Citation: Hussaini, M., Onaizi, S.A. & Vohra, M.S. High-capacity removal of crystal violet using ZIF-8/graphene quantum dot composite with RSM optimization and explainable machine learning. Sci Rep 16, 9035 (2026). https://doi.org/10.1038/s41598-026-39933-2
Keywords: water pollution, dye removal, adsorbent materials, graphene quantum dots, machine learning in environmental engineering