Clear Sky Science · en
RBP4 interferes with tongue squamous cell carcinoma progression by inhibiting the PI3K/AKT signaling pathway and promoting macrophage M1-type polarization
Why Tongue Cancer Needs New Ideas
Tongue cancer can rob people of the ability to speak, eat, and taste, and even with surgery, chemotherapy, and radiation, long-term survival often remains disappointing. This study explores an unexpected helper in our blood, a vitamin A–carrying protein called RBP4, and shows how it can both slow the growth of tongue tumors and rally the body’s own immune defenders. By uncovering how this molecule works inside cancer cells and in the surrounding immune cells, the research points toward fresh strategies that may one day make treatments more effective and less damaging.
A Hidden Protein in Tongue Tumors
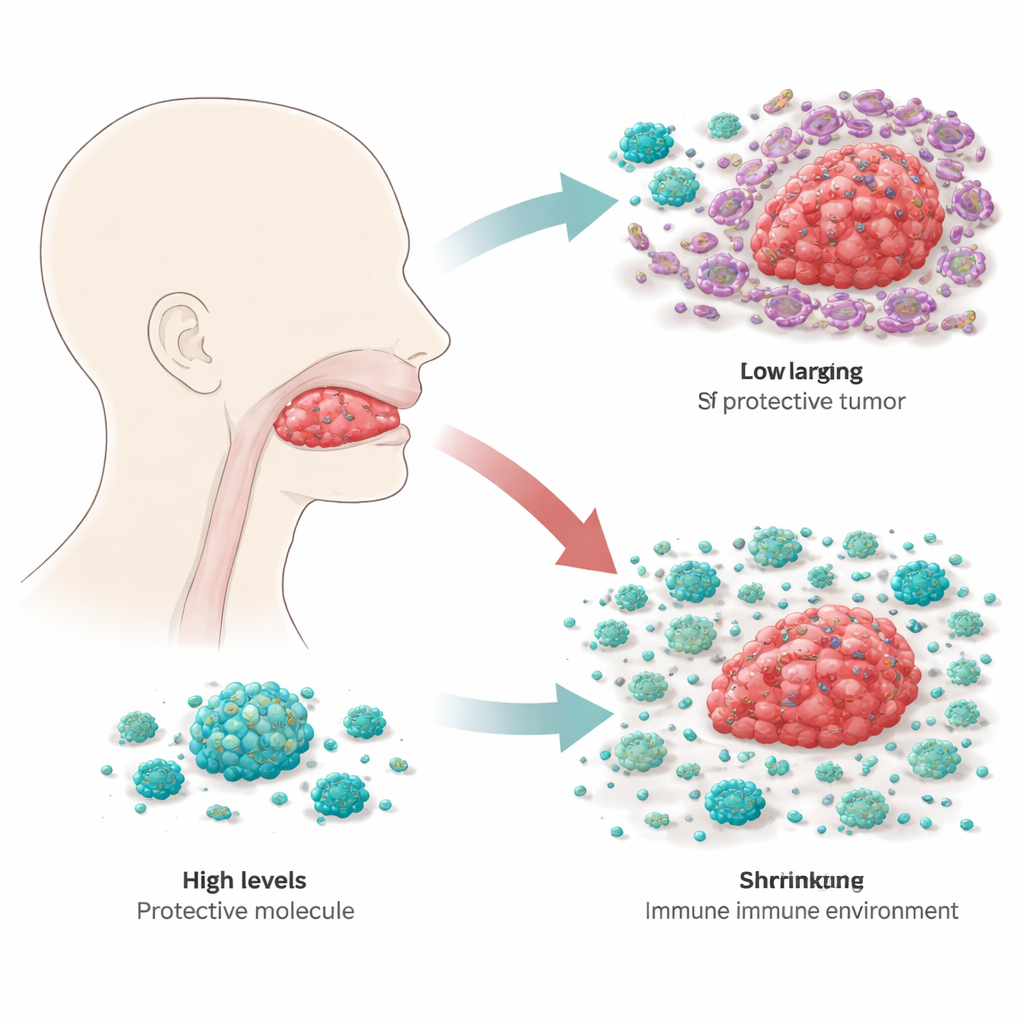
The researchers began by sifting through large cancer gene databases to see which immune-related genes might predict how patients with tongue squamous cell carcinoma fare over time. Among hundreds of candidates, RBP4, a protein best known for transporting vitamin A in the bloodstream, emerged as especially interesting. When the team compared tumor samples with nearby healthy tongue tissue, they found that RBP4 levels were consistently much lower in the cancer. This pattern was confirmed across several independent datasets and in tissue samples from 20 patients, suggesting that loss of RBP4 is a common feature of this disease.
Slowing Tumor Growth From the Inside
To find out what RBP4 actually does in tongue cancer cells, the scientists engineered tumor cell lines to either produce extra RBP4 or make much less of it. In dishes, cells with higher RBP4 grew more slowly, while those with reduced RBP4 multiplied faster and were better at moving and invading through artificial membranes, behaviors linked to spread and relapse. When these modified cells were implanted into mice, tumors with extra RBP4 stayed smaller, whereas tumors with reduced RBP4 grew more aggressively. By tracking key growth-control switches inside the cells, the team showed that RBP4 dampens a major growth pathway known as PI3K–AKT–mTOR, which many cancers rely on as an internal gas pedal. With RBP4 high, this pathway is quieter, and tumor cells are less able to adopt the flexible, mobile state that helps them migrate.
Turning Immune Cells Into Tumor Fighters
Cancer does not grow in isolation; it lives within a neighborhood of immune cells, blood vessels, and support cells called the tumor microenvironment. Here, macrophages—large immune cells that can either attack or assist tumors—play a central role. The investigators noticed that tumors with more RBP4 also tended to contain more of the “M1” type of macrophage, which is associated with inflammation and tumor killing, rather than the “M2” type, which often supports tumor growth. In cell culture, when macrophages were exposed to either purified RBP4 or tongue cancer cells engineered to secrete more RBP4, they shifted toward this M1, tumor-fighting state. When RBP4 was reduced in the cancer cells, the macrophages tended to slide toward the more tumor-friendly profile instead.
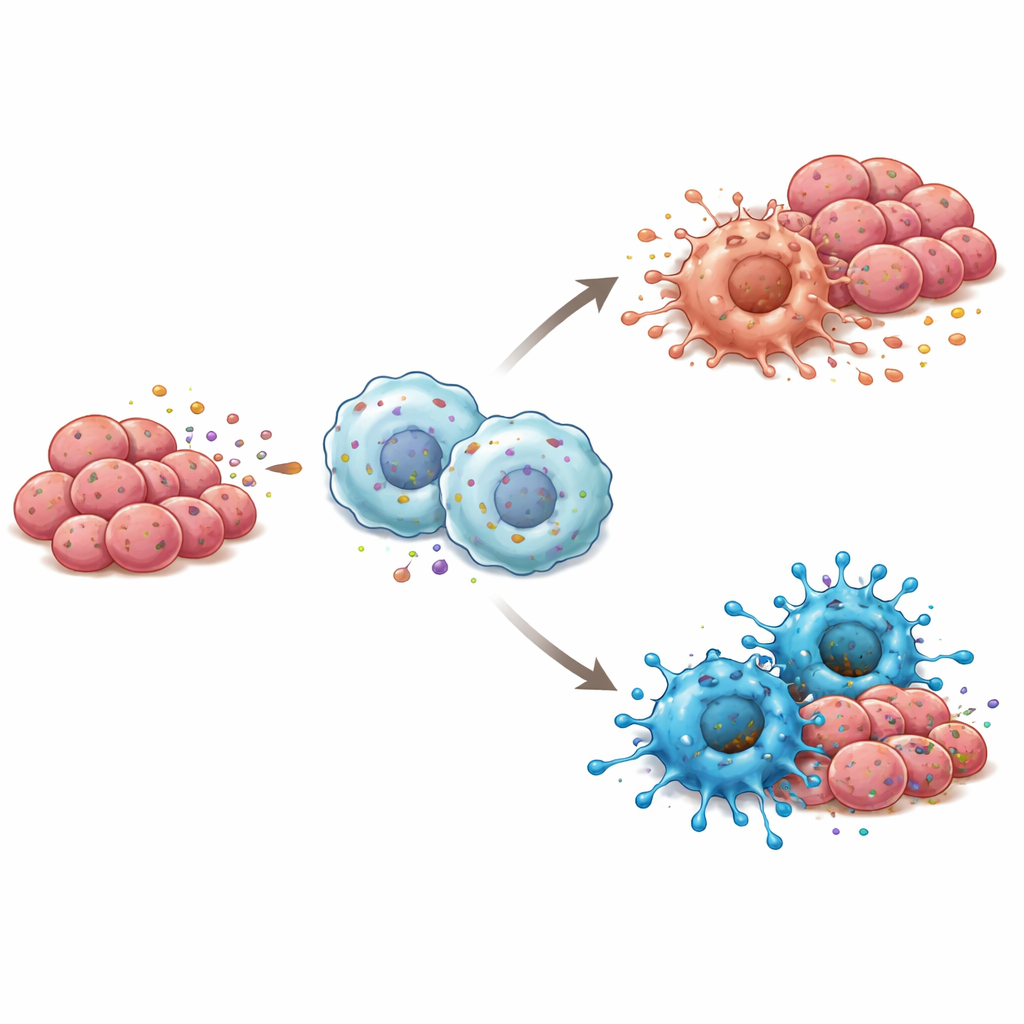
Signaling Pathways Behind the Immune Shift
To understand how RBP4 nudges macrophages into a more hostile stance toward tumors, the researchers examined which internal alarm systems were turned on in these immune cells. They found that RBP4 stimulated a sensor called TLR4 on the macrophage surface and, in turn, activated NF-κB, a master switch that drives inflammatory responses. Blocking TLR4 with a chemical inhibitor shut down this chain reaction and prevented macrophages from adopting the M1 state, even in the presence of RBP4. In mice, tumors formed from tongue cancer cells rich in RBP4 and mixed with macrophages grew more slowly and contained more M1-type cells, along with stronger TLR4 and NF-κB activity, reinforcing the idea that this pathway is key to RBP4’s immune effects.
What This Could Mean for Future Care
Taken together, the work portrays RBP4 as a double-acting brake on tongue cancer: inside tumor cells, it turns down an overactive growth circuit, and in the tumor’s neighborhood, it helps convert macrophages into more vigilant defenders. Although many questions remain—such as exactly how RBP4 interacts with its targets and how best to boost it safely—the study suggests that restoring or mimicking RBP4’s actions could both slow tumor expansion and strengthen the body’s own immune pressure on the cancer. For patients, that raises the prospect of future therapies that do not just cut tumors out, but also reshape their internal wiring and local “ecosystem” to keep them from coming back.
Citation: Yan, Y., Miao, N. & Wang, X. RBP4 interferes with tongue squamous cell carcinoma progression by inhibiting the PI3K/AKT signaling pathway and promoting macrophage M1-type polarization. Sci Rep 16, 9375 (2026). https://doi.org/10.1038/s41598-026-39915-4
Keywords: tongue cancer, RBP4, tumor microenvironment, macrophages, PI3K AKT pathway