Clear Sky Science · en
Novel sterol-spiropyran derivatives: synthesis and photoactivity in Langmuir monolayers
Light as a Gentle Switch
Imagine being able to gently "turn a dial" on the surface of a cell-sized membrane using nothing more than a beam of light. This study introduces a new family of light-sensitive molecules that do exactly that. By attaching a tiny light-activated switch to a cholesterol-like structure, the researchers show how to expand or relax a membrane-like film on water, simply by changing the color of the light. Such control could one day help shape smart drug carriers, soft microscopic machines, or sensors that respond cleanly and reversibly to light.
Building a Light-Responsive Membrane Helper
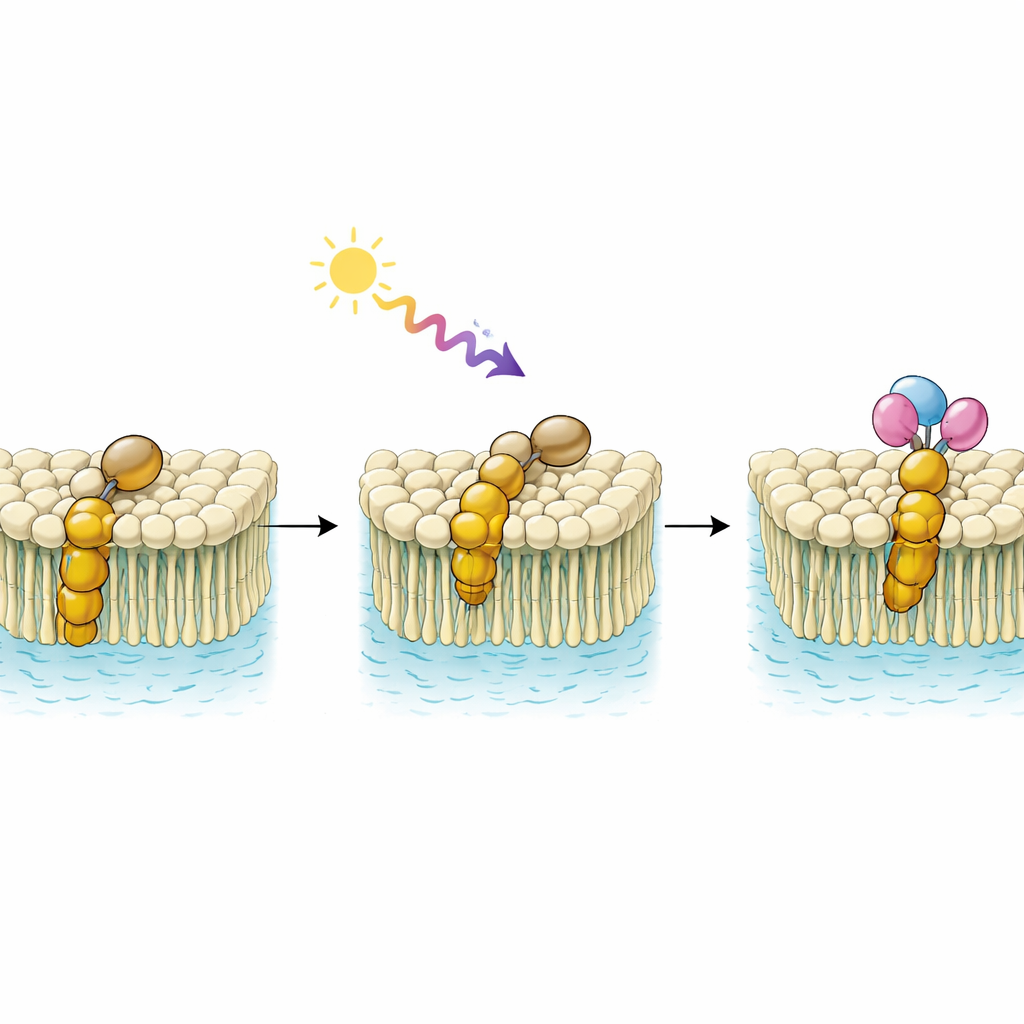
The team designed three new molecules by joining a well-known light-switchable unit, called a spiropyran, to different sterols – the same family of molecules that includes cholesterol. Sterols are nature’s favorite way of tuning the stiffness and organization of cell membranes, because they slot neatly among common membrane lipids. By fusing the spiropyran switch to sterols based on cholenic acid, cholesterol, and ergosterol, the authors created conjugates that should slip easily into membrane-like environments while also changing shape when illuminated.
In solution, these conjugates behave much like other spiropyran systems. Under ultraviolet (UV) light they convert to a flatter, more water-loving form (known as merocyanine); under visible light they switch back to a more compact, water-shy form (spiropyran). The researchers tracked this back-and-forth by recording how the molecules absorb light across the visible spectrum, seeing characteristic broad peaks appear and disappear as the forms interconverted. Although the solubility of the new compounds is limited, mixed methanol–water allowed both versions to be observed clearly, confirming reliable and reversible switching.
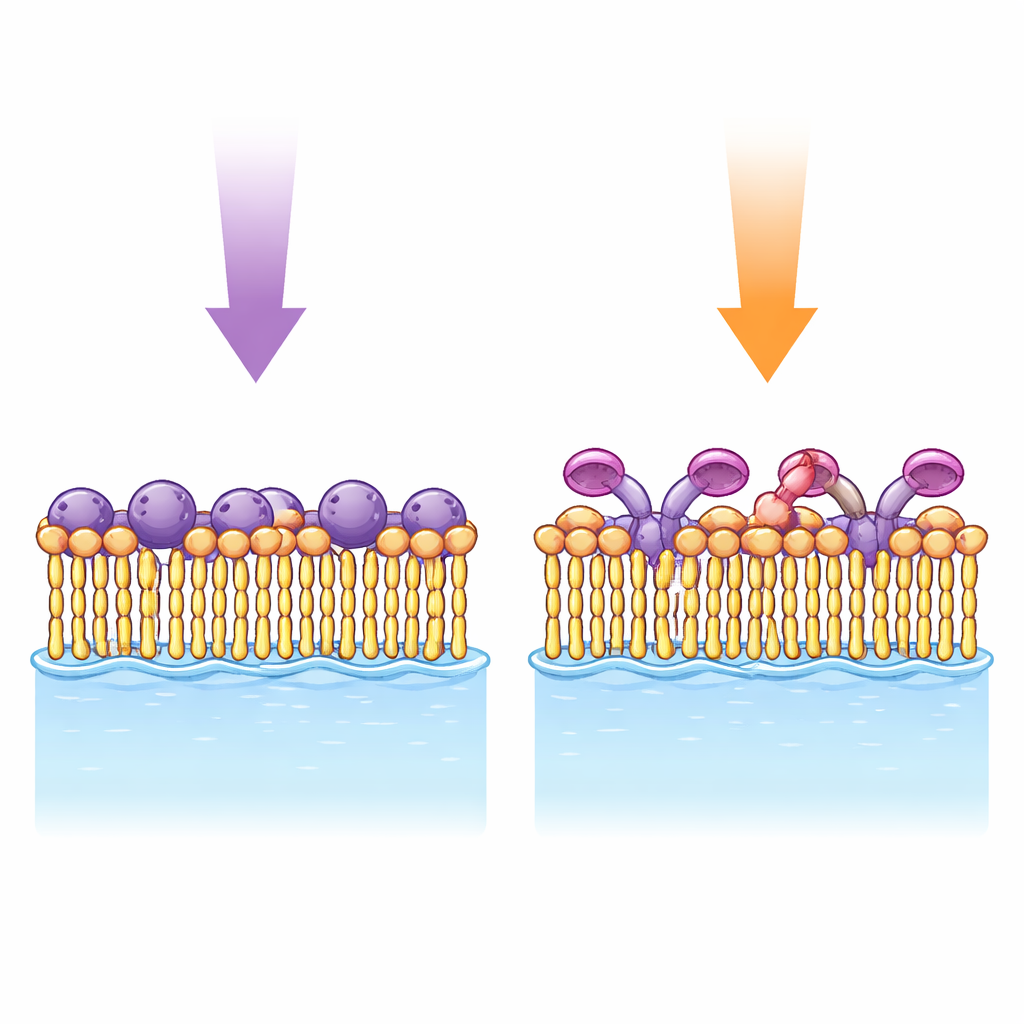
Testing the Switch on a Floating Film
To find out whether these conjugates can really act inside a membrane-like setting, the authors spread thin films – Langmuir monolayers – on the surface of water. These monolayers were made from a charged lipid known to interact strongly with cholesterol, mixed with one of the new spiropyran–sterol molecules. By slowly squeezing the monolayer and measuring the resulting surface pressure, they mapped how tightly packed the molecules were, and how this changed when the light switch was flipped between its two forms. After UV illumination, which favors the more water-loving merocyanine, the monolayer consistently occupied a larger area at the same pressure, indicating that the switched heads were pulling closer to the water and pushing neighboring molecules apart.
How Membrane Stiffness Controls the Response
Beyond showing that the films respond to light, the researchers wanted to understand how quickly they do so and what controls that speed. They fixed the film’s area, illuminated it, and watched the surface pressure relax over time. These measurements revealed that the pressure decayed in a simple, exponential fashion, as if governed by a single, dominant switching process. By comparing films containing cholesterol-based and ergosterol-based conjugates, they could then connect the switching time with how stiff or compressible the monolayer was. Using standard relations between pressure and area, they calculated the monolayer’s compression modulus – a measure of how hard it is to squeeze – and found a clear linear link: stiffer monolayers responded more slowly.
Hidden Structure and Subtle Phase Changes
The detailed pressure–area measurements also hinted at more complex behavior, especially for films containing the ergosterol-based conjugate. For these systems, the relation between stiffness and pressure showed a minimum and a maximum, much like what is seen during a first-order phase transition, where two distinct states coexist. One possible explanation is that, when the conjugate is in its more water-shy form, it is partially expelled from the flat film to form tiny three-dimensional structures as the surface is compressed, producing plateau-like regions in the data. Regardless of the exact microscopic picture, the experiments show that the way the light-switching molecules move and rearrange is tightly linked to the mechanical properties of the surrounding membrane.
From Floating Films to Smart Membranes
In essence, this work demonstrates that purpose-built spiropyran–sterol molecules can nestle into membrane-like films and act as local, reversible light switches, expanding or relaxing the surface in a controlled manner. For a non-specialist, the key message is that we now have a way to couple simple beams of light to subtle mechanical changes in materials that closely mimic cell membranes. Looking ahead, embedding these conjugates into full bilayer membranes or vesicles could allow researchers to tune permeability, tension, and curvature on demand, paving the way for light-steered drug carriers, responsive soft machines, and new tools to probe how real biological membranes change shape and function under dynamic control.
Citation: Negus, T., Perry, A. & Petrov, P.G. Novel sterol-spiropyran derivatives: synthesis and photoactivity in Langmuir monolayers. Sci Rep 16, 9258 (2026). https://doi.org/10.1038/s41598-026-39881-x
Keywords: light-responsive membranes, spiropyran switches, cholesterol-based conjugates, Langmuir monolayers, photochromic materials