Clear Sky Science · en
Multi-omics identification of key targets for the osteogenic differentiation of human bone marrow mesenchymal stromal cells under oxidative stress
Why stressed bones matter
As we age or develop chronic diseases like diabetes and osteoporosis, our bones lose their ability to repair themselves. A big culprit is “oxidative stress” – a buildup of reactive molecules that damage cells. This study asks a practical question with major implications for fractures and bone implants: what exactly goes wrong inside human bone marrow stem cells when they are exposed to oxidative stress, and can we find a molecular switch that helps them keep forming new bone?
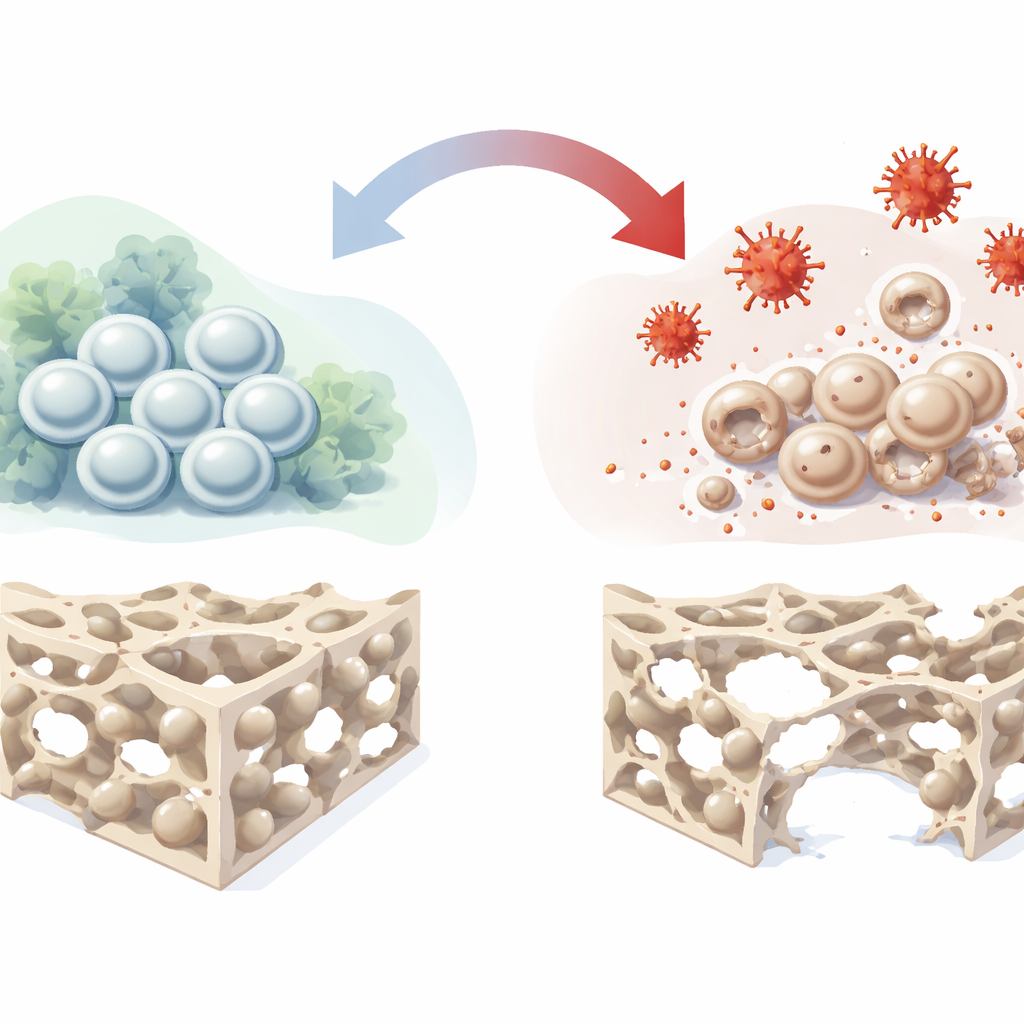
Stem cells that build bone
Deep inside our bones live bone marrow mesenchymal stromal cells, a versatile population that can renew themselves and mature into bone-forming cells, cartilage cells, and fat cells. Because they naturally help mend damaged tissue, they are a leading candidate for next‑generation treatments to repair bone defects and osteonecrosis. However, in real patients these cells often sit in hostile environments marked by poor blood flow, low oxygen, inflammation, and oxidative stress. In such settings their ability to turn into bone cells falters, limiting the success of stem‑cell‑based therapies. The authors set out to recreate this hostile environment in the lab and chart, in detail, how it derails bone formation.
Recreating a harsh environment in the lab
The researchers used hydrogen peroxide, a common source of reactive oxygen species, to impose oxidative stress on human bone marrow stem cells grown in culture. They carefully titrated the dose to find a sweet spot where the cells were stressed but not killed. At or below 400 micromolar, the cells kept their normal spindle-like shape and remained viable, although their internal chemistry clearly shifted: reactive oxygen levels rose, mitochondrial function began to change, and the balance between survival and death‑related proteins tipped toward stress adaptation. At higher doses, the cells lost their shape and died in large numbers. Using the well‑tolerated 400 micromolar dose, the team then triggered the cells to form bone and watched what happened.
How stress blocks bone building
Under oxidative stress, the stem cells’ ability to become bone cells declined in several complementary tests. Early bone‑forming activity, tracked by an enzyme called alkaline phosphatase, dropped off as stress increased. Later, when the cells should have laid down mineral deposits, the culture dishes showed fewer and fainter calcium nodules. Key bone‑related genes and proteins, such as RUNX2 and osteopontin, were also turned down. To look under the hood, the scientists combined two powerful “omics” approaches: RNA sequencing to profile which genes were more or less active, and large‑scale protein analysis to see which proteins actually changed in abundance. Together these datasets revealed hundreds of shifts in cell cycle control, chromosome behavior, metabolism, and the organization of the scaffolding that surrounds cells, painting a picture of stem cells whose internal timing and structural support are thrown off by oxidative stress.
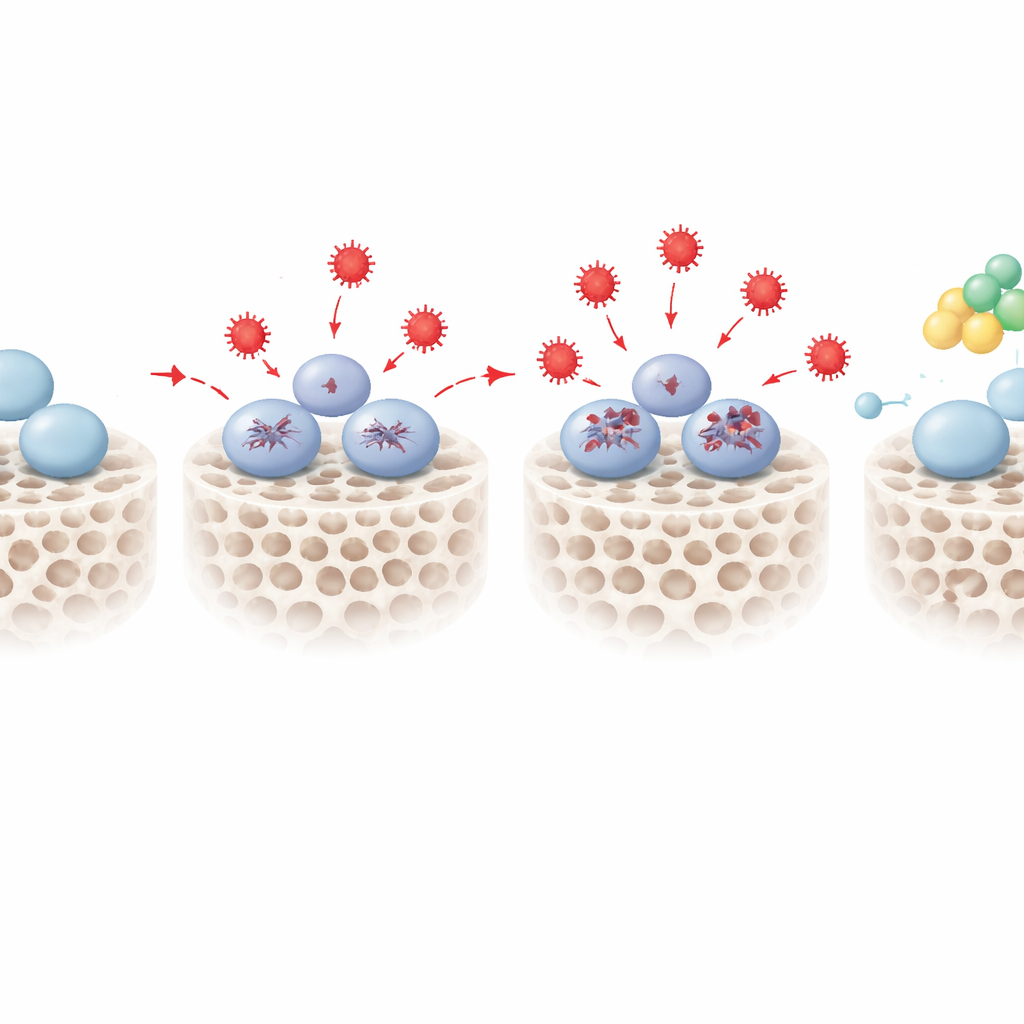
Finding a protective switch called PENK
By overlaying the RNA and protein maps, the team narrowed in on 18 molecules that consistently changed in stressed cells and were linked to both stress responses and bone formation. One stood out: proenkephalin, or PENK, best known as a precursor of natural opioid peptides. Under oxidative stress, PENK levels rose in a dose‑dependent manner. When the scientists artificially dialed PENK down using genetic tools, stressed stem cells became even worse at forming bone, showing weaker early enzyme activity and less mineral deposition. When they boosted PENK, the opposite happened: even under the same oxidative conditions, the cells recovered much of their ability to build mineralized bone matrix. Additional pathway analyses suggested that PENK may work by tuning certain metabolic routes, including sphingolipid metabolism, that connect redox balance to decisions about whether a stem cell keeps its potential or moves toward a bone fate.
What this means for future bone repair
This study demonstrates that oxidative stress alone is enough to cripple the bone‑forming capacity of human bone marrow stem cells, and it identifies PENK as a built‑in protective factor that helps them resist this damage. For non‑specialists, the message is clear: the success of stem‑cell‑based bone therapies will depend not just on the cells themselves, but also on the stressed environments they are placed into and the molecular switches that help them cope. By highlighting PENK as a promising target, the work points toward future drugs or gene‑based approaches that could shore up bone repair in patients whose tissues are bathed in oxidative stress, from elderly individuals to those living with chronic metabolic or inflammatory diseases.
Citation: Dong, W., Zheng, Y., Zhou, Y. et al. Multi-omics identification of key targets for the osteogenic differentiation of human bone marrow mesenchymal stromal cells under oxidative stress. Sci Rep 16, 8215 (2026). https://doi.org/10.1038/s41598-026-39818-4
Keywords: bone regeneration, oxidative stress, mesenchymal stem cells, osteogenic differentiation, PENK