Clear Sky Science · en
Rational design of a cascade CdS/C3N4/COF heterostructure for high-performance Cr(VI) photoreduction
Turning Light into a Tool for Cleaner Water
Chromium pollution is a serious threat to drinking water and aquatic life, especially when this metal is present in its highly toxic hexavalent form, often released from factories such as tanneries and plating plants. This study explores how to harness ordinary visible light to turn dangerous chromium into a safer form, using a carefully engineered nanomaterial that makes better use of light energy than traditional catalysts. The work offers a glimpse of how smart materials design can help tackle both industrial contamination and the need for low-energy water treatment.
Why Toxic Chromium Is So Hard to Remove
In nature, chromium mainly appears in two forms: a relatively benign trivalent state and a hexavalent state that is far more mobile, more soluble, and strongly linked to cancer and organ damage. Once hexavalent chromium dissolves into water, it travels easily through soil and groundwater, making cleanup difficult. Conventional approaches such as filtration, chemical precipitation, or simple adsorption can trap chromium but often create new waste streams and require added chemicals or energy. Photoreduction—a process where light-driven electrons convert hexavalent chromium into its safer trivalent form—has emerged as a promising alternative. However, most light-activated materials struggle because the charge carriers they generate (electrons and holes) tend to cancel each other out before they can do useful chemistry.
Building a Three-Part Light-Activated Cleaner
To overcome these limitations, the researchers built a "cascade" structure that combines three different components, each playing a distinct role. They start with thin sheets of graphitic carbon nitride, a metal-free material that can absorb visible light and supply powerful reducing electrons. Onto this, they add tiny particles of cadmium sulfide, a classic light absorber with good charge mobility. Finally, they weave in a porous covalent organic framework, a rigid, sponge-like organic network whose pores and chemical groups help tune how charges move and where they recombine. Made by relatively simple heating and ultrasonic mixing steps, the resulting CdS/C3N4/COF composite forms an intimately connected network in which all three materials touch and share charges at many small interfaces. 
Guiding Charges Instead of Simply Separating Them
Most advanced catalysts aim to keep electrons and holes apart to prevent them from annihilating each other. This study takes a more subtle route: it accepts that recombination will happen, and instead controls which charges recombine and where. Detailed measurements of crystal structure, light absorption, light emission, and electrochemical behavior reveal that the porous framework acts as an electronic traffic director. Low-energy electrons that are less useful for tough reactions are steered into the framework, where they meet and neutralize holes. At the same time, the higher-energy electrons generated in the carbon nitride sheets are preserved and kept away from these dead-end pathways. This deliberate "charge-preferential recombination" design creates a so‑called cascade S-scheme: an energy landscape where unhelpful charges are quietly removed, leaving the most powerful electrons free to attack hexavalent chromium at the catalyst surface.
How Well the New Material Cleans Water
When tested under visible light in mildly acidic water, the optimized three-part catalyst removed about 92 percent of hexavalent chromium within 90 minutes—far better than any of the individual ingredients or simpler two-part mixtures. Careful control experiments showed that most of the chromium was truly converted, not just stuck to the surface, and that the key players were the electrons delivered directly from carbon nitride to chromium ions. Adjusting conditions such as catalyst amount, pH, and initial chromium level revealed a sweet spot: enough catalyst to capture light without blocking it, and a pH around 3, where chromium is easy to reduce but not so strongly adsorbed that electrons cannot reach it. The material also worked for several cycles, though its activity slowly declined as reaction products and minor structural changes partially blocked active sites. 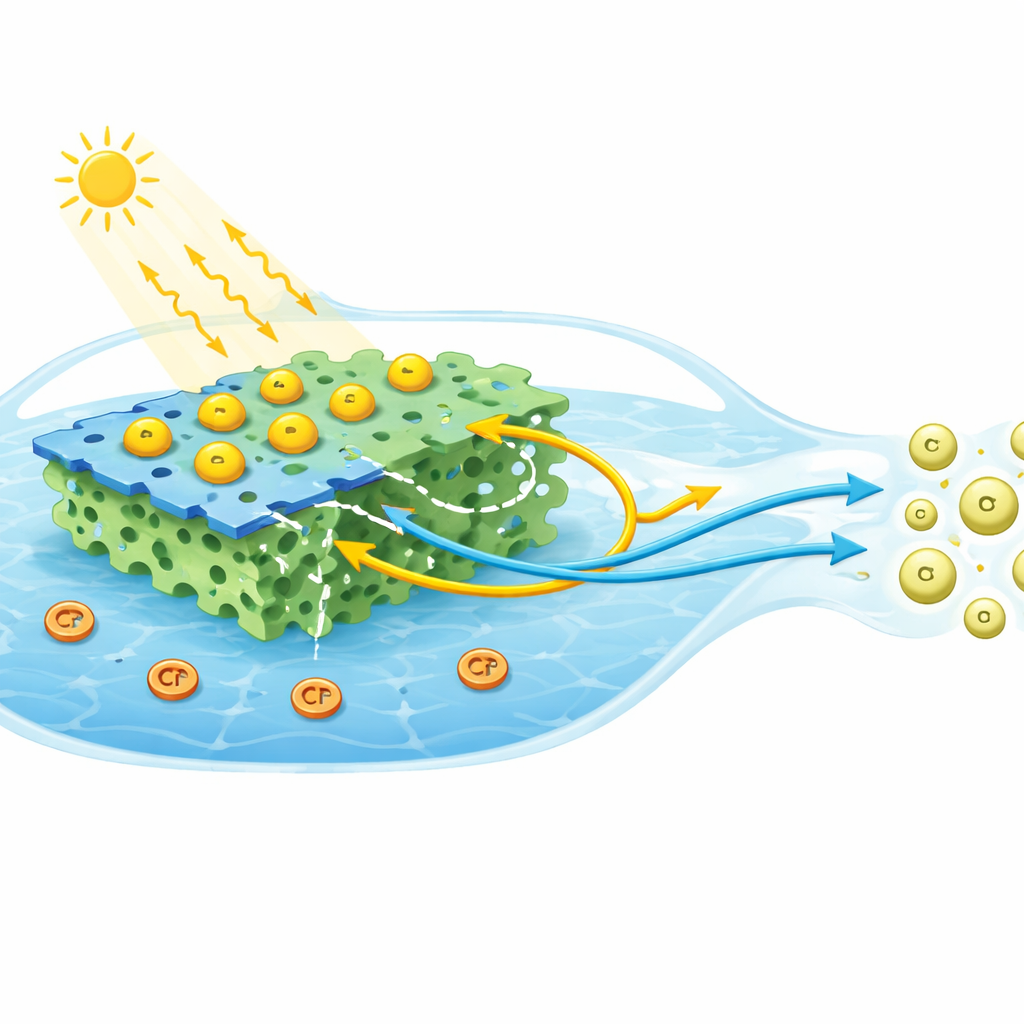
What This Means for Future Water Treatment
For non-specialists, the main message is that the way we arrange and connect materials at the nanoscale can dramatically change what light can do for us. By purposefully allowing less useful charges to cancel out inside a porous framework while protecting the most energetic electrons, the authors turned a familiar combination of substances into a much more effective chromium-cleaning system. Although the current design still relies on acidic conditions and contains cadmium, which raises its own safety concerns, the underlying concept—using programmable organic frameworks as electronic mediators in multi-part structures—could be extended to safer chemistries. This approach points toward future photocatalysts that clean water more completely and efficiently, powered simply by visible light.
Citation: Babaie, H., Sohrabnezhad, S. & Foulady-Dehaghi, R. Rational design of a cascade CdS/C3N4/COF heterostructure for high-performance Cr(VI) photoreduction. Sci Rep 16, 8238 (2026). https://doi.org/10.1038/s41598-026-39799-4
Keywords: chromium pollution, photocatalytic water treatment, graphitic carbon nitride, covalent organic frameworks, visible light catalysts