Clear Sky Science · en
Impact of G-quadruplex RNA oxidation on its conformational dynamics and interaction with ALS-associated TDP-43
Why This Matters for Nerve Health
Amyotrophic lateral sclerosis (ALS) is a fatal disease in which the nerve cells that control movement slowly die, yet the root causes of this selective damage remain mysterious. This study asks a simple but important question: as we age and our cells experience more “rust-like” chemical damage, does this wear and tear on RNA—the working copy of our genes—disrupt vital signals that keep motor neurons alive? By zooming in on a special folded RNA shape and a key ALS-linked protein called TDP-43, the work uncovers how tiny chemical scars may help set the stage for disease. 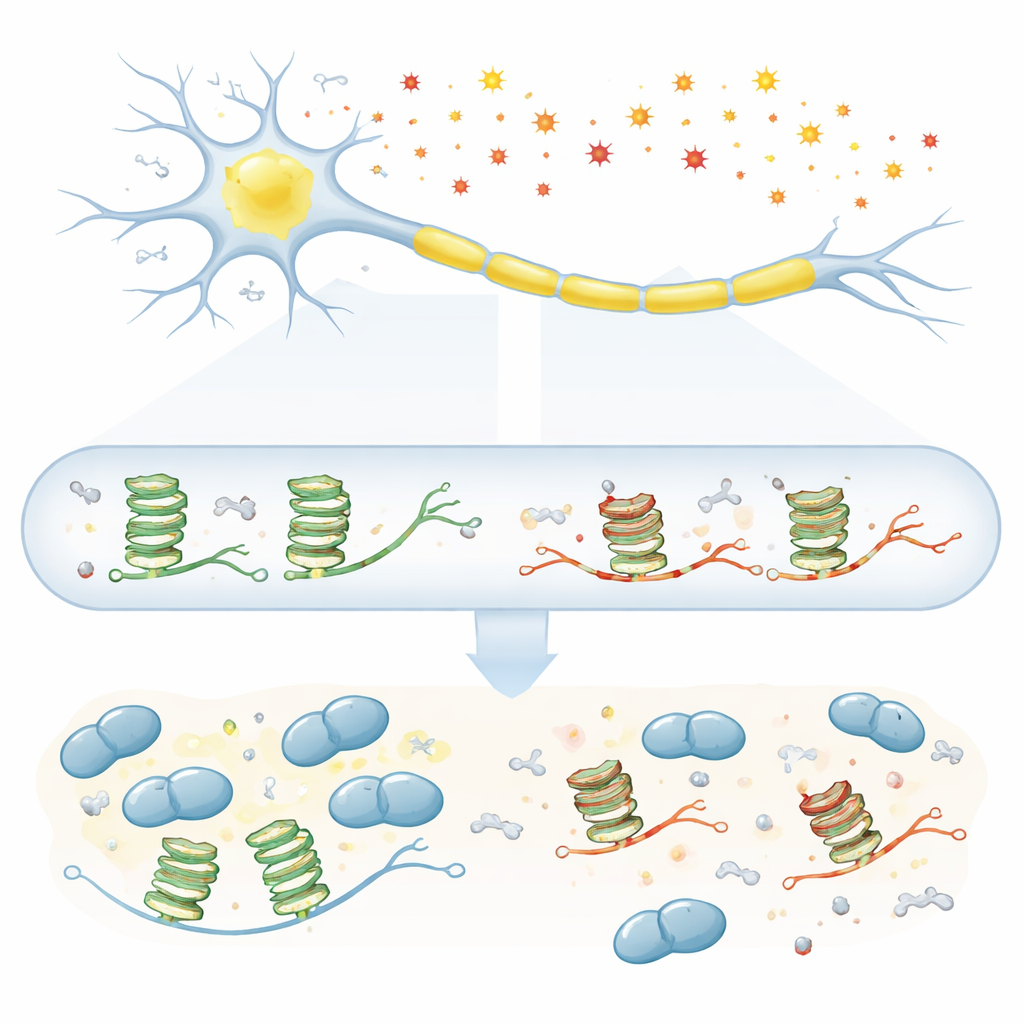
A Fragile RNA Knot at the Heart of Motor Neurons
Motor neurons rely on long-distance shipping of RNA messages down their axons so that proteins can be made on site, near the connections with muscle. Many of these messages carry a special structural “knot” called a G-quadruplex, built from stretches of the base guanine. Proteins such as TDP-43 recognize this knot and hitch the RNA onto transport granules that move along the axon. The problem is that guanine is also the easiest base to oxidize when cells are under stress from reactive oxygen species, which accumulate with aging. Earlier clues suggested that G-quadruplexes and TDP-43 are central in ALS, but it was unclear exactly how oxidation of these RNA knots might interfere with their partnership.
Watching Oxidative Stress Distort RNA Shapes
The researcher first exposed human neuroblastoma cells to hydrogen peroxide, a common oxidative agent, and then examined the state of G-quadruplex structures. Signals from a G-quadruplex-sensitive probe dropped as oxidative stress increased, indicating that these compact RNA knots were losing their normal shape. To pin down the chemistry, the team then built synthetic RNAs that form G-quadruplexes and deliberately mixed in varying amounts of 8-oxoguanine, a well-known oxidized version of guanine found in aging neurons and ALS tissue. Using a suite of biophysical tools, they showed that even modest levels of this modification loosen the orderly stacking of the G-quadruplex, although the exact outcome depends on the surrounding RNA sequence. 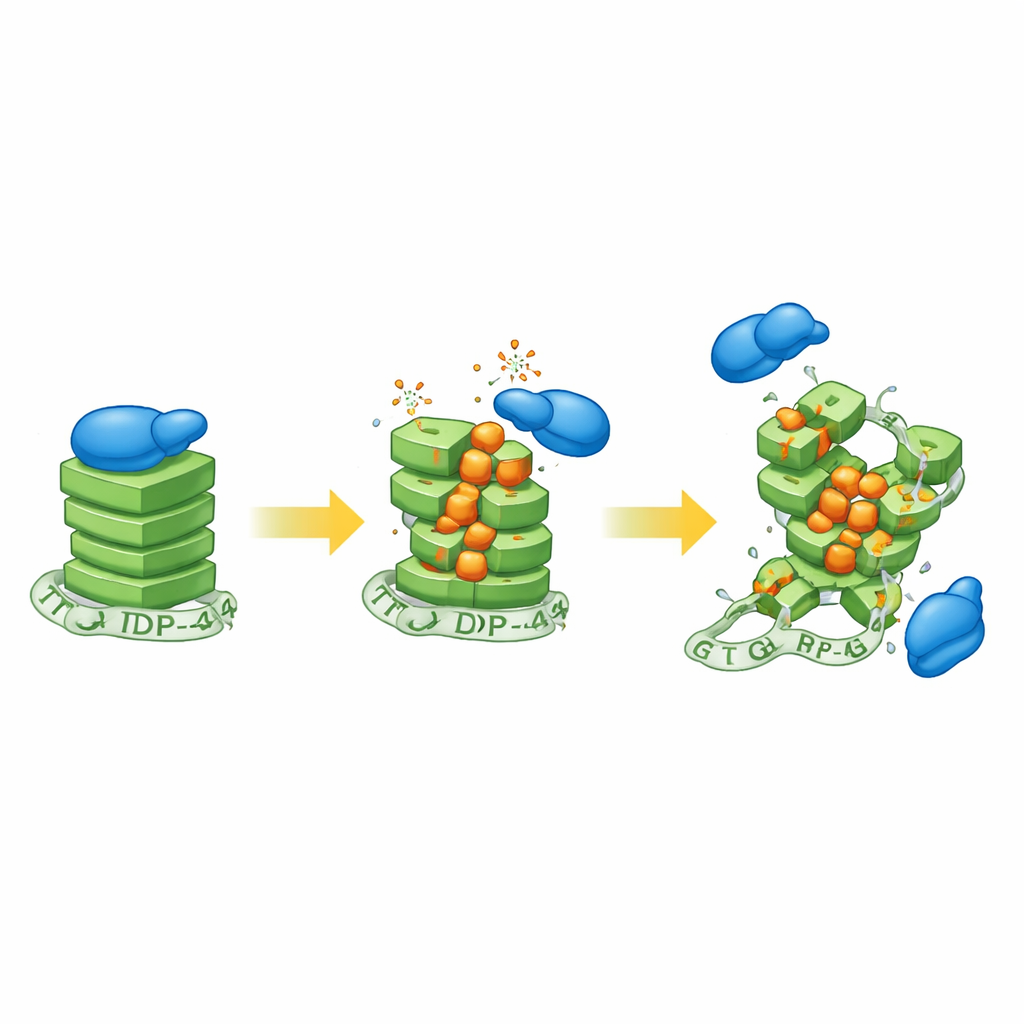
New Faults: Mismatched Bases and Mispaired Strands
Closer analysis revealed two distinct kinds of structural trouble. In one G-quadruplex derived from the PSD-95 message, oxidized guanines tended to pair inappropriately with nearby adenines, forming “mismatches” that subtly distort the fold. These mismatches appeared most readily at intermediate oxidation levels, when the RNA could still transiently assemble into a G-quadruplex that brought the wrong partners into contact. In another G-quadruplex from the CaMKIIα message, heavy oxidation destabilized the usual intramolecular knot so strongly that the remaining intact guanines sought partners on other RNA strands, promoting abnormal intermolecular G-quadruplexes. Such miswired structures could tangle different RNAs together and interfere with normal RNA traffic inside neurons.
When Damaged RNA Loses Its Protein Partner
The study then asked how these distorted knots interact with TDP-43. Using gel-based binding assays, the researcher showed that TDP-43 strongly prefers intact, parallel G-quadruplexes and increasingly fails to bind as more oxidized bases are introduced. The CaMKIIα structure, already tightly packed, proved especially sensitive: even low oxidation almost abolished TDP-43 binding. A second protein, FUS—also linked to ALS—showed a similar but slightly less severe loss of affinity, suggesting that oxidation broadly weakens the grip of G-quadruplex-binding proteins. Intriguingly, when TDP-43 was mixed with mildly oxidized RNA, the evidence pointed to the protein engaging an unstable intermediate form of the G-quadruplex rather than a fully folded knot, hinting at a dynamic struggle between stabilization and breakdown.
Extra Vulnerability in ALS-Linked Mutant Proteins
The work further examined ten TDP-43 variants found in ALS patients, most carrying changes in a flexible, glycine-rich tail that fine-tunes RNA recognition. All of these mutants already bound normal G-quadruplexes more weakly than the wild-type protein. When oxidized G-quadruplex RNAs were introduced as competitors, the mutants were even less able than the normal protein to recognize and bind the damaged structures. Some variants located in particularly disorder-prone regions were the most affected. This suggests a double hit in aging individuals who carry such mutations: not only is their TDP-43 intrinsically less effective, but age-related RNA oxidation further erodes its ability to shepherd crucial messages along motor neuron axons.
What This Means for Understanding ALS
Put simply, this study shows that age-driven chemical damage reshapes delicate RNA knots that guide message transport in motor neurons, and these misshapen knots no longer cooperate well with TDP-43 or related proteins. The result is a higher chance of misdirected or stalled RNA cargo, exactly in the long, vulnerable axons that keep muscles under control. For people who also inherit TDP-43 mutations, this oxidation-induced weakening of RNA–protein interactions may push already stressed neurons closer to failure. By illuminating this subtle but potent chain of events—from oxidative stress to altered RNA architecture to disrupted transport—the work highlights G-quadruplex RNA oxidation as a promising molecular link between aging and ALS, and a potential target for future protective therapies.
Citation: Ishiguro, A. Impact of G-quadruplex RNA oxidation on its conformational dynamics and interaction with ALS-associated TDP-43. Sci Rep 16, 8802 (2026). https://doi.org/10.1038/s41598-026-39767-y
Keywords: amyotrophic lateral sclerosis, RNA oxidation, G-quadruplex, TDP-43, motor neuron degeneration