Clear Sky Science · en
Identification of blood-brain barrier injury-related biomarkers in cerebral infarction using transcriptomic analysis
Why This Matters for Stroke and Brain Health
When someone has a stroke caused by a blocked blood vessel in the brain, doctors can see the damage on scans but still struggle to predict who will get worse swelling, bleeding, or long-term disability. A big part of that damage happens at the brain’s protective gatekeeper, the blood–brain barrier, which normally keeps harmful substances in the blood from leaking into fragile nerve tissue. This study asked a simple but important question: are there telltale molecular signals that reveal when this barrier is breaking down, and could those signals one day guide better diagnosis and treatment for stroke patients?
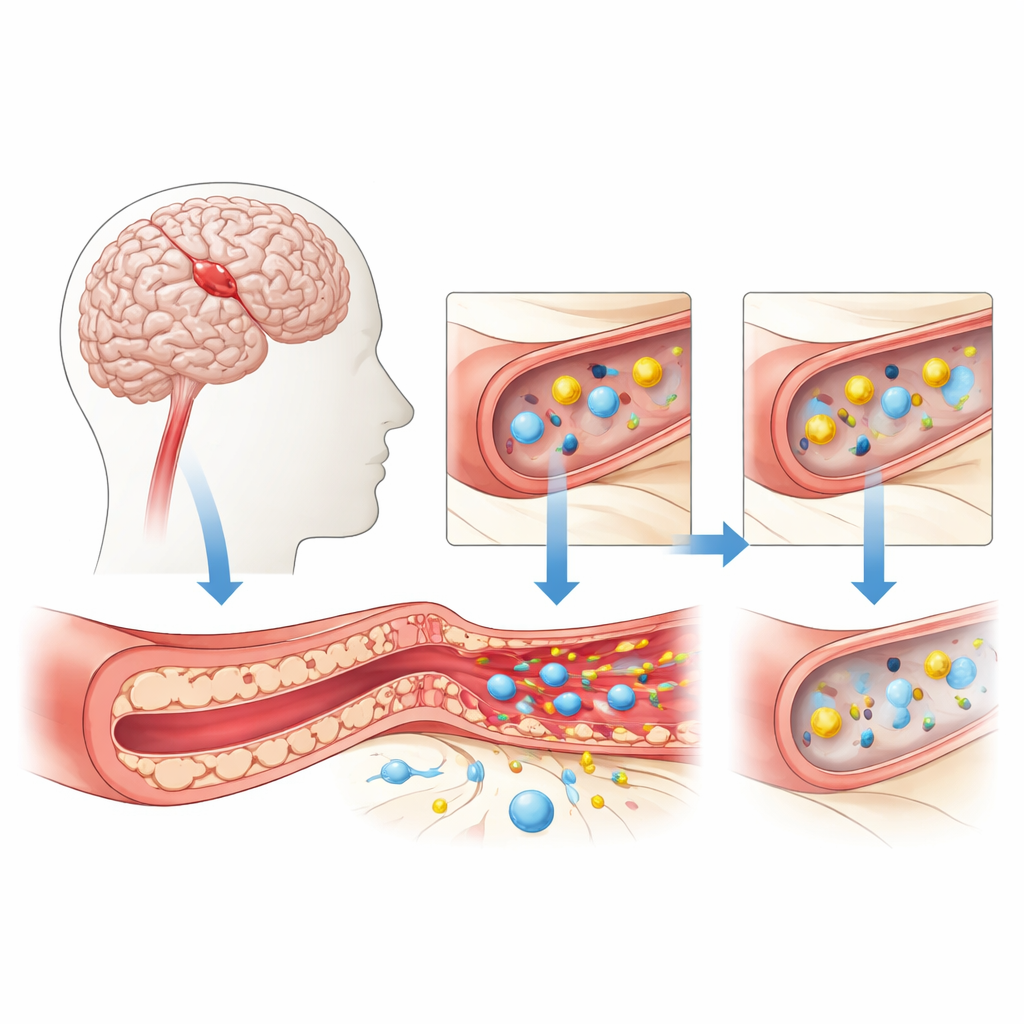
The Brain’s Protective Gate Under Stress
The blood–brain barrier is formed by tightly packed cells lining tiny brain blood vessels. These cells carefully control what can pass from blood to brain, allowing in vital nutrients like oxygen and glucose while blocking toxins and immune cells that could cause harm. During a cerebral infarction, or ischemic stroke, blood flow drops suddenly. Without enough oxygen and sugar, these vessel-lining cells become stressed and begin to lose their tight seal. Gaps open between them, fluid seeps into the surrounding tissue, and chemical messengers of inflammation flood the area, all of which can worsen brain swelling and injury.
Recreating Stroke Damage in the Lab
To probe what happens inside these barrier cells, the researchers used a well-known human cell line that mimics the lining of brain blood vessels. They exposed these cells to a low-oxygen, no-glucose environment—called oxygen–glucose deprivation—to imitate the conditions of a stroke, and then restored normal conditions to mimic treatment and recovery. They measured how well the cells survived, how much they leaked a cell-damage enzyme, and how many inflammatory molecules they released. As expected, the stressful treatment reduced cell health, boosted inflammatory signals such as IL-1β, IL-6, and TNF-α, and increased leakage of a damage marker called LDH. When oxygen and nutrients were restored, many of these harmful changes eased, and the cells regained some of their ability to form new vessel-like tubes in a growth test.
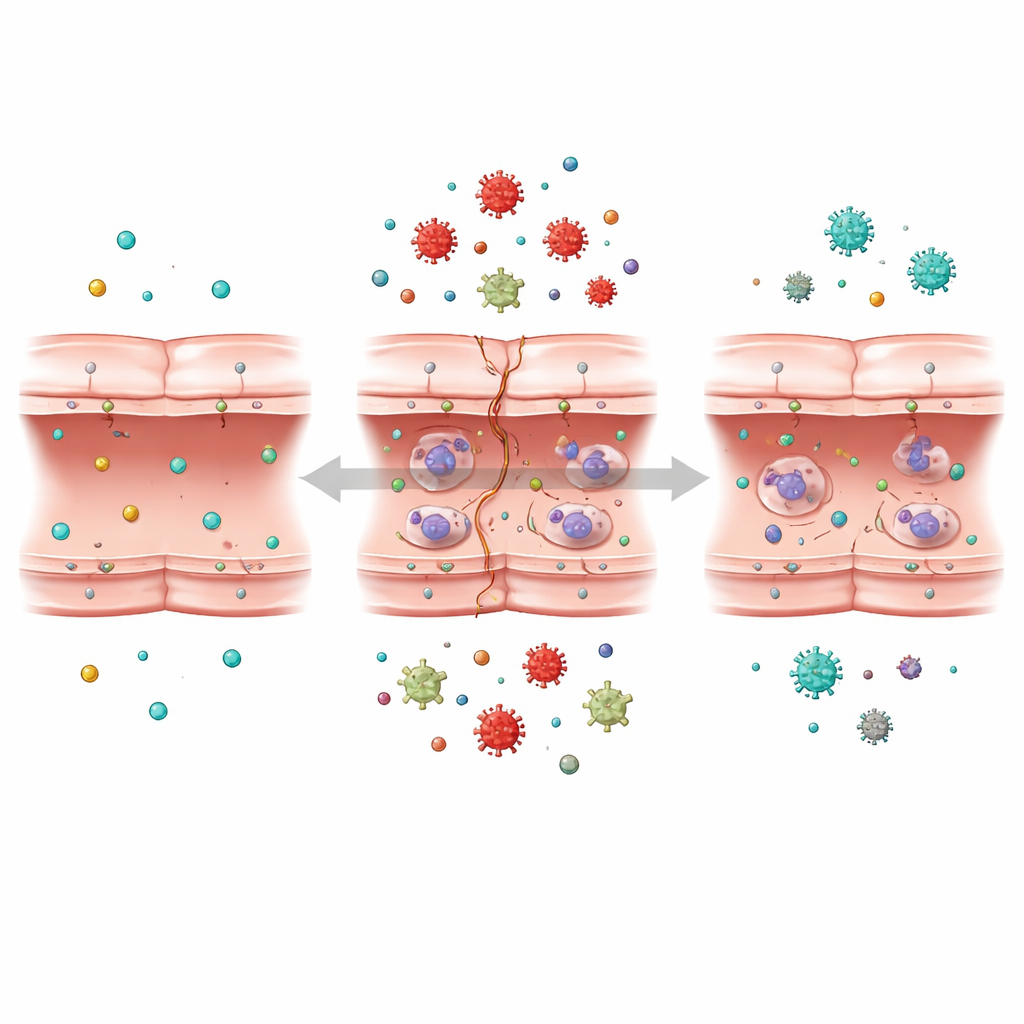
Listening to the Cell’s Molecular Conversation
The team then turned to a powerful technique known as transcriptomics, which reads out the activity levels of thousands of genes at once. By comparing normal cells, oxygen–glucose–deprived cells, and cells allowed to recover, they identified more than a thousand genes whose activity changed in response to stroke-like stress. Using advanced data tools and machine learning, they grouped genes that behaved together and searched for those most strongly tied to barrier injury. They found that many of the most affected genes were linked to three key structures inside the cell: the ribosome, which builds proteins; the endoplasmic reticulum, which helps fold and process them; and mitochondria, the tiny power plants that supply energy.
Ribosomes Emerge as Telltale Signals
From this large gene set, the researchers used a random forest algorithm—a type of decision-tree-based machine learning—to narrow down a short list of especially informative genes. They then mapped how the corresponding proteins interact with each other. This analysis highlighted six core genes, most of them tied to the ribosome, the cell’s protein factory. In the stroke-like condition, these ribosome-related genes were switched on more strongly, while their activity dropped back toward normal when oxygen and glucose were restored. This pattern suggests that changes in the cell’s protein-making machinery may be a sensitive early signal of blood–brain barrier damage and recovery.
What This Could Mean for Future Care
For a layperson, the take-home message is that this study points to a new class of molecular “warning lights” that turn on when the brain’s protective barrier is hurt during a stroke. Instead of focusing only on well-known culprits like enzymes that chew up the vessel wall, the work shows that the cell’s own protein factories respond strongly to injury and calm down again when conditions improve. If future research confirms that these ribosome-related markers can be detected in patient samples such as blood, doctors might one day use them to gauge how badly the barrier is damaged, to time treatments that reduce inflammation and swelling, and to test new drugs aimed at preserving this crucial gatekeeper of brain health.
Citation: Liu, X., He, Y., Zhang, N. et al. Identification of blood-brain barrier injury-related biomarkers in cerebral infarction using transcriptomic analysis. Sci Rep 16, 8119 (2026). https://doi.org/10.1038/s41598-026-39763-2
Keywords: ischemic stroke, blood-brain barrier, biomarkers, ribosomal genes, endothelial cells