Clear Sky Science · en
Intravenous transplantation of multi-lineage differentiating stress enduring cell promotes functional recovery after traumatic brain injury in mice
Why Healing the Injured Brain Matters
Traumatic brain injury, or TBI, can happen in an instant—from a car crash, a fall, or a sports collision—and leave people with lasting problems in movement, thinking, and daily life. Current treatments focus mainly on limiting the immediate damage, but they do little to repair the brain itself. This study explores a new kind of repair cell, called a Muse cell, that can be given through a simple vein injection and may help the injured brain rebuild its own circuitry, at least in mice.
A Special Kind of Repair Cell
Muse cells are a rare subset of the body’s own stem-like cells found in bone marrow, blood, and connective tissues. Unlike many experimental cell types, they do not form tumors and can survive harsh conditions such as poor blood flow and inflammation—exactly the environment found in a freshly injured brain. They are drawn to chemical distress signals released by damaged tissue, allowing them to travel through the bloodstream and home in on sites of injury. Once there, they can swallow up debris from dying cells and use that information to turn into the kinds of cells the tissue needs, including nerve cells and their supporting partners.
Testing Muse Cells in a Mouse Brain Injury Model
To see whether Muse cells could help a damaged brain recover, the researchers used a well-controlled mouse model of TBI. They created a localized injury in the outer layer of the brain by briefly touching the skull with a copper probe chilled in liquid nitrogen, producing a reproducible patch of damaged cortex. One week later—after the immediate shock of the injury had passed—mice were randomly assigned to receive one of four treatments through a vein in the tail: a modest dose of human Muse cells, a low dose of standard mesenchymal stromal cells (a more familiar type of therapeutic cell), a much higher dose of these standard cells, or a simple salt solution as a control.
Watching Movement Return
The team tracked how well the mice regained movement over nearly three months. They used a rotating rod test, which measures how long a mouse can keep its balance on a spinning cylinder, and a cylinder test that captures how evenly the animal uses its left and right forelimbs. All animals showed marked motor problems shortly after injury. Those given Muse cells, however, began to improve about a month after treatment and continued to get better through the end of the study. Their performance on both movement tests clearly surpassed that of mice given the low dose of standard cells or the salt solution, and even beat the high-dose standard cell group, which showed only mild and delayed benefit.
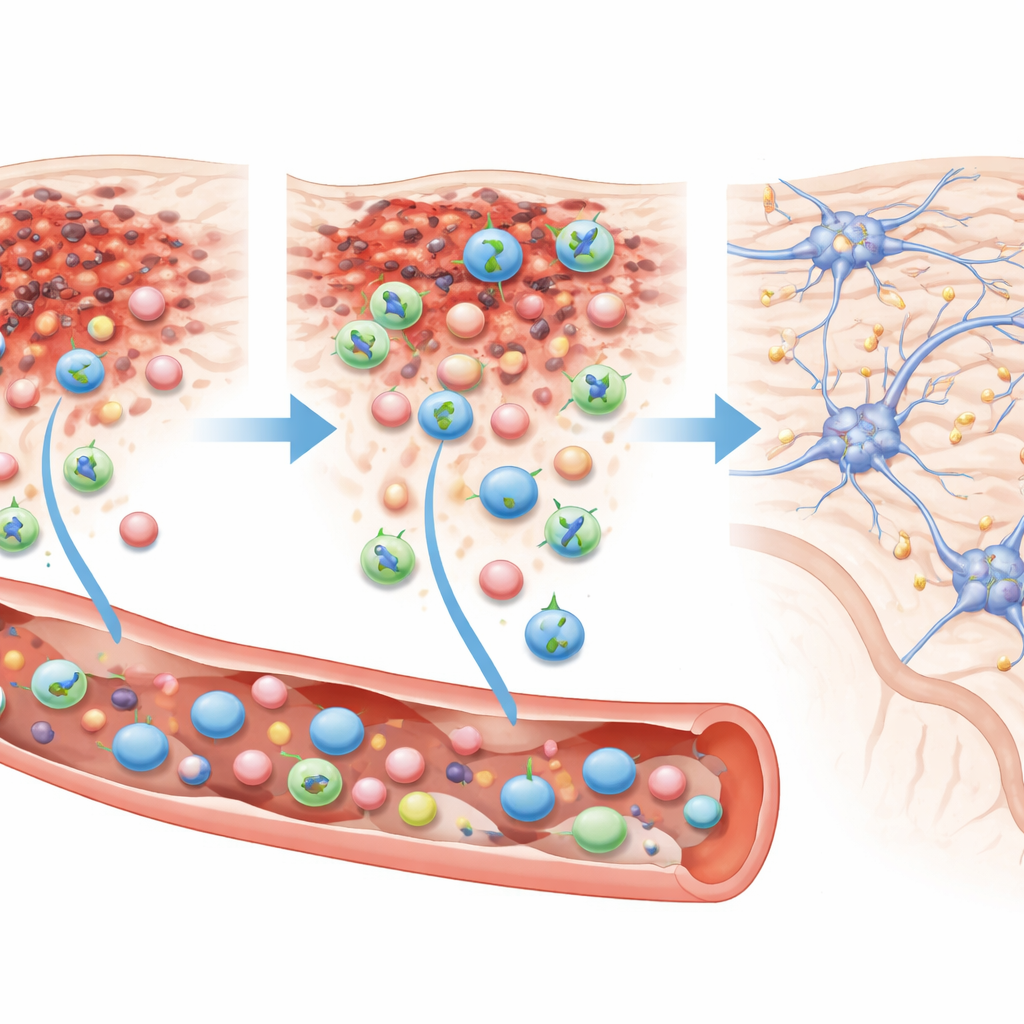
How the Cells Find and Fix the Damage
When the researchers examined the brains under the microscope 84 days after injury, they found many human cells clustered around the damaged area in Muse-treated mice, identified by human-specific markers. In contrast, mice that received standard cells had only a scattering of human cells, even at the higher dose. In the Muse group, roughly 60 percent of these engrafted cells carried a marker typical of mature neurons, and about 20 percent carried a marker of oligodendrocytes, the cells that wrap nerve fibers with insulating layers needed for fast signal transmission. None showed a common astrocyte marker, suggesting a focused shift toward rebuilding nerve pathways and their support structure.
What This Could Mean for Future Treatment
The findings indicate that Muse cells, when given intravenously after a brain injury, can home to the damaged area, become key brain cell types, and support long-lasting recovery of movement in mice. Because these cells naturally resist stress and seem to avoid provoking strong immune reactions, they might one day be used across donors without intensive immune-suppressing drugs. However, questions remain about the best dose, timing, and delivery route, and the current work used a specific type of focal injury in animals, not the full complexity of human TBI. Even so, the study offers a hopeful glimpse of a future in which a simple infusion of repair-ready cells could help the injured brain rebuild rather than merely endure.
Citation: Shiraishi, K., Yamamoto, S., Kushida, Y. et al. Intravenous transplantation of multi-lineage differentiating stress enduring cell promotes functional recovery after traumatic brain injury in mice. Sci Rep 16, 9458 (2026). https://doi.org/10.1038/s41598-026-39760-5
Keywords: traumatic brain injury, stem cell therapy, Muse cells, neuroregeneration, motor recovery