Clear Sky Science · en
Bacterial colonization of tumors drives immune activation and checkpoint blockade efficacy
Why Tiny Tenants in Tumors Matter
Cancer treatment has been transformed by immunotherapies that unleash the body’s own immune cells against tumors. Yet many patients still see little benefit, and it has been hard to predict who will respond. This study explores an unexpected player that may tip the scales: bacteria living inside tumors themselves. By examining mouse cancer models, the researchers show that these microscopic tenants can change how the immune system behaves in and around a tumor, and in turn help determine whether a widely used immunotherapy called anti–PD-1 works well or falls flat.
Hidden Bacteria Inside Tumors
For years, most attention has focused on the trillions of microbes in the gut and how they shape overall immunity. More recently, scientists have discovered that many human tumors also harbor small but active bacterial communities. The team behind this study asked two basic questions: do commonly used mouse tumor models also contain bacteria, and if so, do those bacteria actually influence how tumors grow and respond to treatment? They compared two models: MCA-205, a type of sarcoma, and MOC1, a head and neck cancer model. Using DNA-based tests, sequencing, and culture methods, they found that MCA-205 tumors consistently carried living bacteria, while MOC1 tumors did not, even when placed in the bacteria-rich mouth. In MCA-205, bacterial levels tended to rise as the tumors enlarged, hinting that the growing tumor provided a favorable niche for microbes.
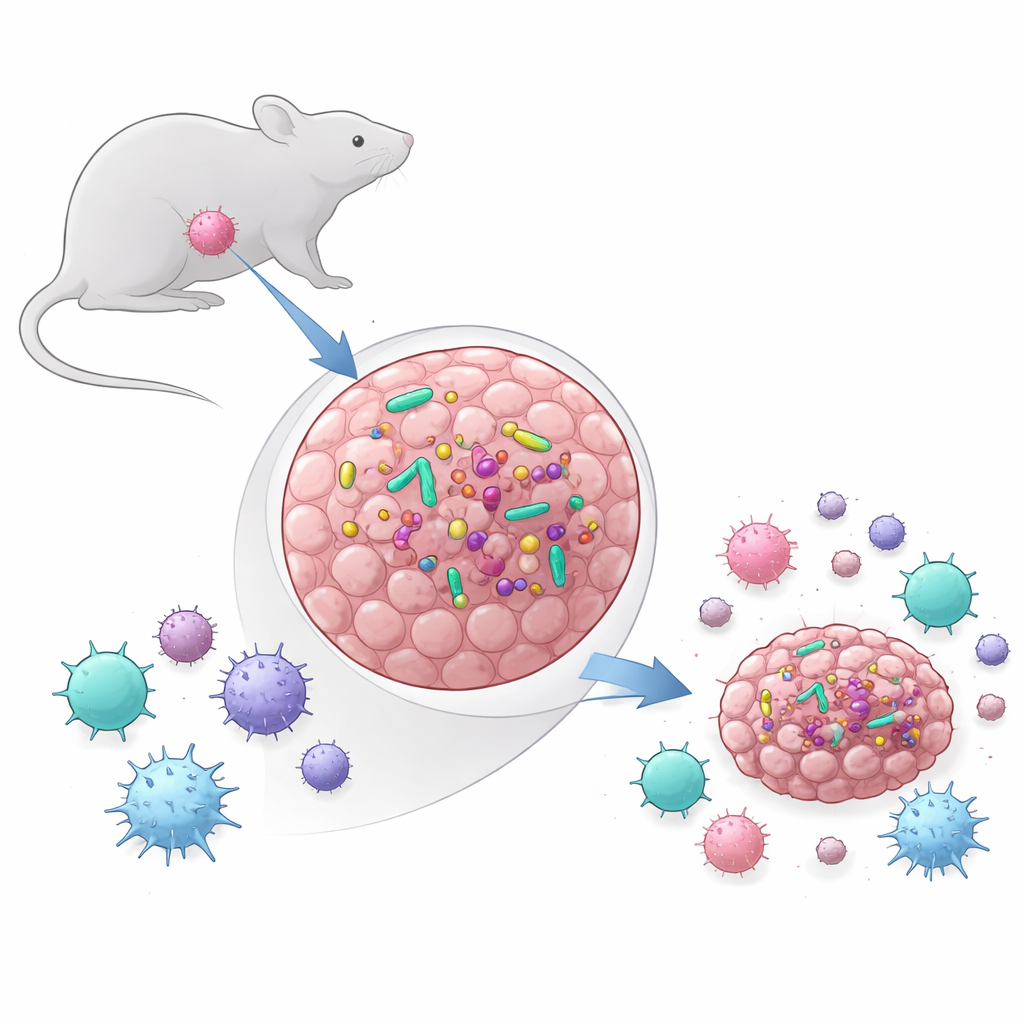
Tracing Where the Bugs Come From
Finding bacteria inside tumors raised another question: are these invaders coming from the skin, the mouth, or the gut? By comparing the bacterial DNA in tumors with samples from stool, skin swabs, and oral swabs, the researchers found that the intratumoral community was simpler and less diverse than the rich ecosystems in the gut and mouth. Most of the bacterial types in tumors overlapped with those seen in the gastrointestinal tract, while relatively few matched the skin. This suggests that bacteria may leak from the gut or oral cavity into the bloodstream and then seed the tumor, where only certain species manage to persist. The mix of bacterial groups mirrored those reported in several human cancers, reinforcing the idea that these mouse tumors can model real-world tumor–microbe interactions.
How Tumor Bacteria Shape Immunotherapy Success
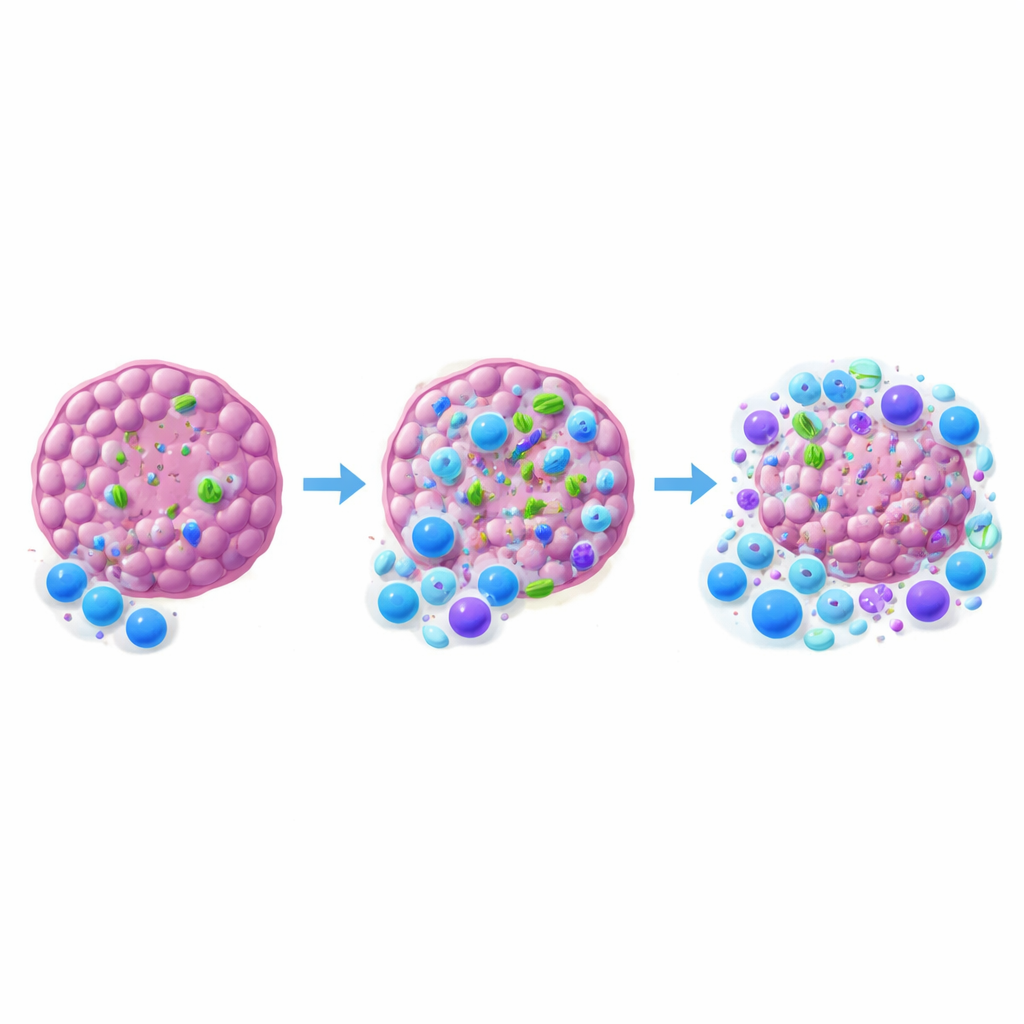
The central finding of the study is that tumor-dwelling bacteria can make or break the success of anti–PD-1 therapy, a checkpoint blocker designed to reinvigorate exhausted killer T cells. When the team used antibiotics that penetrated the tumor and wiped out local bacteria while largely sparing gut colonization, MCA-205 tumors became much less responsive to anti–PD-1. Responses dropped even more when the gut microbiota were broadly disrupted. By contrast, in MOC1 tumors that lacked detectable bacteria from the outset, antibiotics had little effect on either bacterial load or treatment outcome. In MCA-205, tumors that shrank in response to anti–PD-1 tended to contain more bacteria than those that resisted therapy, linking high intratumoral bacterial burden with better control of growth.
Inside the Immune Battlefront
To understand how bacteria tilt the immune balance, the researchers examined immune cells within and around the tumors. When intratumoral bacteria were removed with tumor-penetrating antibiotics, fewer killer CD8 T cells infiltrated the tumor, and the ratio of these cells to regulatory T cells shifted in favor of suppression. Dendritic cells, the scouts that pick up tumor material and activate T cells, were also fewer in number and showed lower signs of activation when bacteria were absent. Interestingly, simply adding an extra strain of harmless E. coli into the tumor, or disabling a major microbe-sensing pathway (MyD88) in key immune cells, did not boost therapy. This implies that not just any bacterial presence or simple alarm signaling will do; specific communities and more nuanced communication with the immune system are likely required.
What This Means for Future Cancer Care
To a non-specialist, the takeaway is that bacteria living inside tumors are not mere bystanders. In at least some cancers, they help rally the right immune cells into the tumor and support the action of anti–PD-1 drugs. Other tumors may remain nearly bacteria-free and respond very differently. As scientists design and test new treatments in mice, knowing whether a given tumor model carries bacteria—and which kinds—may be essential for interpreting results and choosing strategies that will translate to patients. In the long run, carefully tuning the tiny communities within tumors themselves could join drugs and immune cells as a new tool for improving cancer immunotherapy.
Citation: Rolig, A.S., Ziglari, T., McGee, G.H. et al. Bacterial colonization of tumors drives immune activation and checkpoint blockade efficacy. Sci Rep 16, 8464 (2026). https://doi.org/10.1038/s41598-026-39758-z
Keywords: intratumoral microbiota, cancer immunotherapy, anti-PD-1, tumor microenvironment, bacteria and tumors