Clear Sky Science · en
Highly sensitive profiling somatic mutations of thyroid cancer by nucleotide-enrichment-based MALDI-TOF MS assay
Why this matters for people with thyroid nodules
Many people discover a lump in their thyroid gland and face a stressful wait to learn whether it is harmless or cancerous, and how serious it might be. Doctors increasingly rely on genetic tests to read tiny changes in tumor DNA that can guide diagnosis, surgery, and follow-up care. This study introduces a faster, more sensitive, and more affordable laboratory test that can detect many important thyroid cancer mutations at once, even when only a few cancer cells are present in a sample.
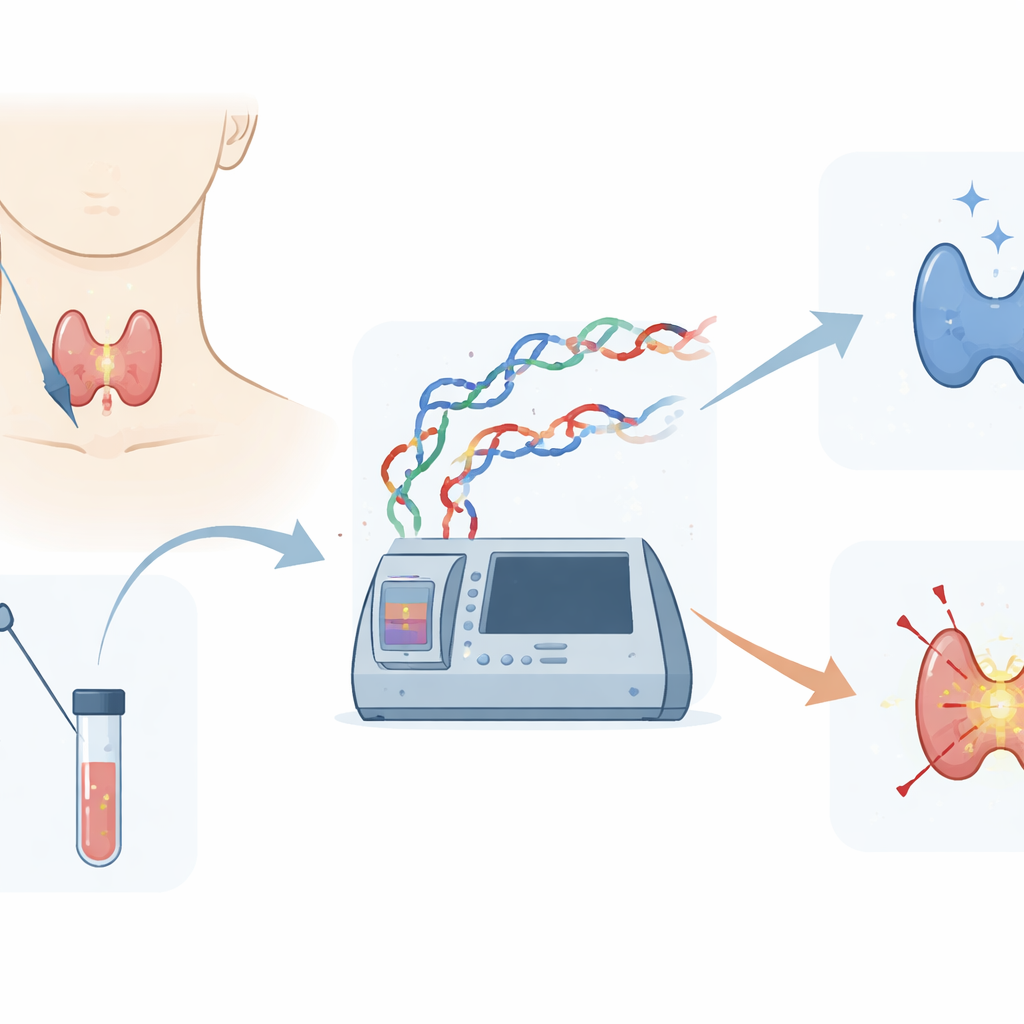
A new way to read cancer DNA
The researchers developed a method called NE-MS, which combines a chemistry trick called nucleotide enrichment with a mass-spectrometry platform that can measure the weights of DNA fragments. In thyroid care, doctors often obtain only a small number of cells using a fine-needle aspiration (FNA) biopsy, making it hard to detect rare cancer mutations with existing tools. NE-MS is designed to work well with these tiny, sometimes poor-quality samples and to test for 26 well-known mutations in genes such as BRAF, RAS, TERT, PIK3CA, and RET that are recommended in major thyroid cancer guidelines.
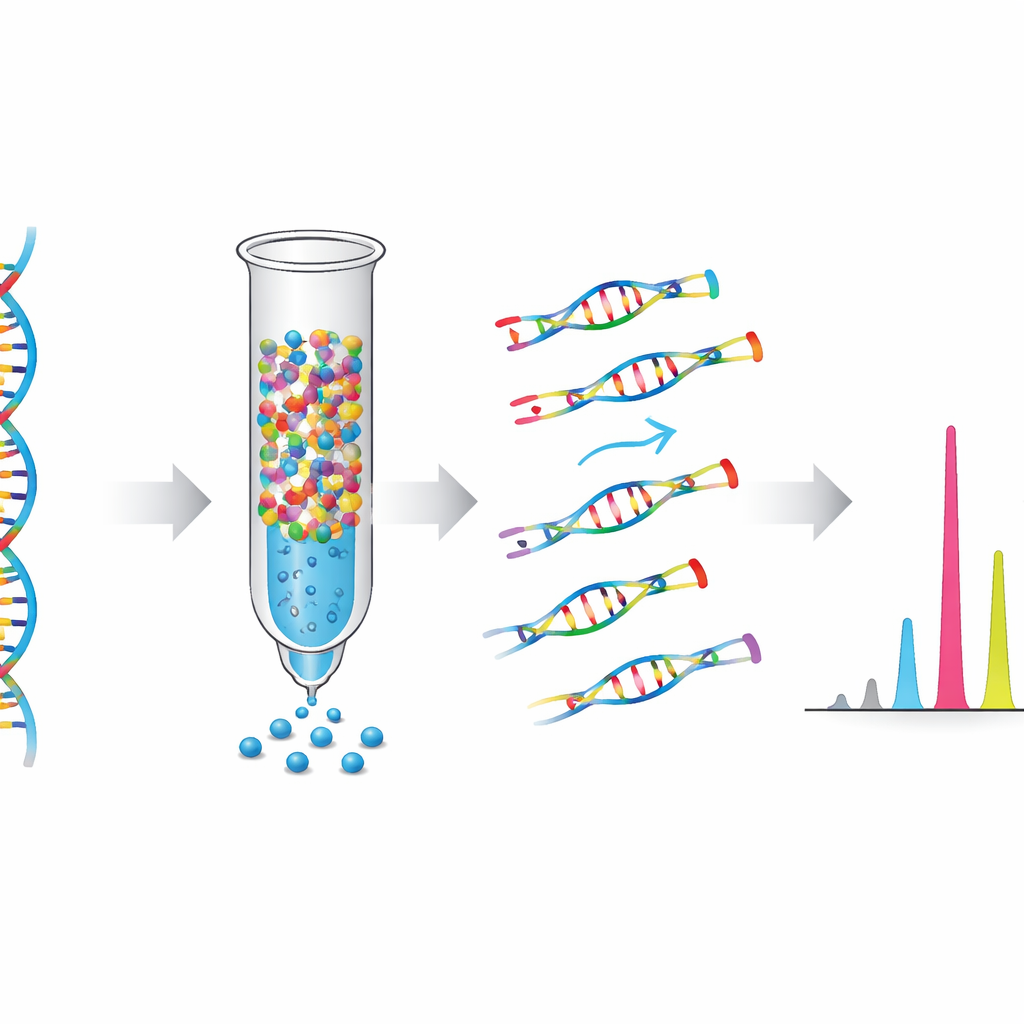
How the test boosts weak signals
Standard mass-spectrometry tests extend both normal and mutant DNA during a key reaction step, so the strong signal from normal DNA can drown out the faint signal from cancer cells. NE-MS turns this problem on its head. During the single-base extension step, the test mixture is deliberately stripped of the building block that would match the normal, or wild-type, DNA at each target position. As a result, only DNA carrying a mutation can be extended and detected, while the normal DNA is largely ignored. The team also built an automated scoring system, based on a robust Z-score, to distinguish true mutation signals from background noise without relying on manual interpretation by a technician.
Proving sensitivity and reliability
To test how well NE-MS works, the scientists used reference DNA samples containing known thyroid cancer mutations at gradually decreasing levels, down to less than one mutant molecule in 300. Compared with the regular mass-spectrometry method, NE-MS lowered the detection limit up to eightfold for key mutations such as NRAS Q61K and TERT C228T, and could reliably detect some changes that the older method essentially missed. In follow-up experiments using real patient samples, NE-MS results for the common BRAF V600E mutation matched both a highly sensitive droplet digital PCR test and next-generation sequencing, achieving 100% agreement. When the method was applied to 466 FNA samples, it showed excellent diagnostic performance, with an area under the ROC curve of 0.99 for identifying BRAF mutations.
Linking mutation patterns to patient outlook
Beyond diagnosis, the team explored how mutation patterns relate to how thyroid cancers behave. They applied NE-MS to more than 1,000 surgically removed thyroid tumor samples preserved in paraffin blocks. Most cancers carried BRAF mutations alone, but a smaller group of patients had multiple mutations, such as combinations of BRAF with TERT or PIK3CA. These patients were more likely to be older, male, have larger tumors, distant metastases, higher tumor stage, and to receive radioactive iodine therapy, all signs of a more aggressive disease course. The test also picked up rare RET mutations linked to a particularly serious form of thyroid cancer, highlighting patients who might benefit from targeted drugs.
What this could mean for care
In summary, this study presents NE-MS as a practical, high-throughput tool that can sensitively profile many thyroid cancer mutations in a single run, using both needle biopsies and surgical samples. It delivers results in about six hours, at roughly one-third the cost of broad next-generation sequencing panels, with a simpler workflow that suits busy clinical laboratories. For patients, such a test could mean clearer answers from small biopsies, better distinction between low-risk and high-risk thyroid cancers, and more personalized treatment decisions. Because the underlying chemistry is not limited to thyroid genes, the same approach could be expanded to guide care in many other types of cancer.
Citation: Bai, H., Li, Y., Li, J. et al. Highly sensitive profiling somatic mutations of thyroid cancer by nucleotide-enrichment-based MALDI-TOF MS assay. Sci Rep 16, 8080 (2026). https://doi.org/10.1038/s41598-026-39755-2
Keywords: thyroid cancer, somatic mutations, molecular diagnostics, fine-needle aspiration, mass spectrometry