Clear Sky Science · en
Functional transition of CA2 pyramidal neurons along the proximodistal axis determines resonance frequency preference
How Brain Waves Shape Memory and Social Behavior
The hippocampus is a deep brain structure crucial for forming memories, navigating spaces, and guiding social behavior. Within it lies a narrow but influential zone called CA2, long overshadowed by its better-known neighbors, CA1 and CA3. This study asks a deceptively simple question with big implications: do CA2 nerve cells along this tiny strip of tissue "listen" best to different brain wave rhythms, and could that help explain how the hippocampus coordinates complex thoughts and behaviors? 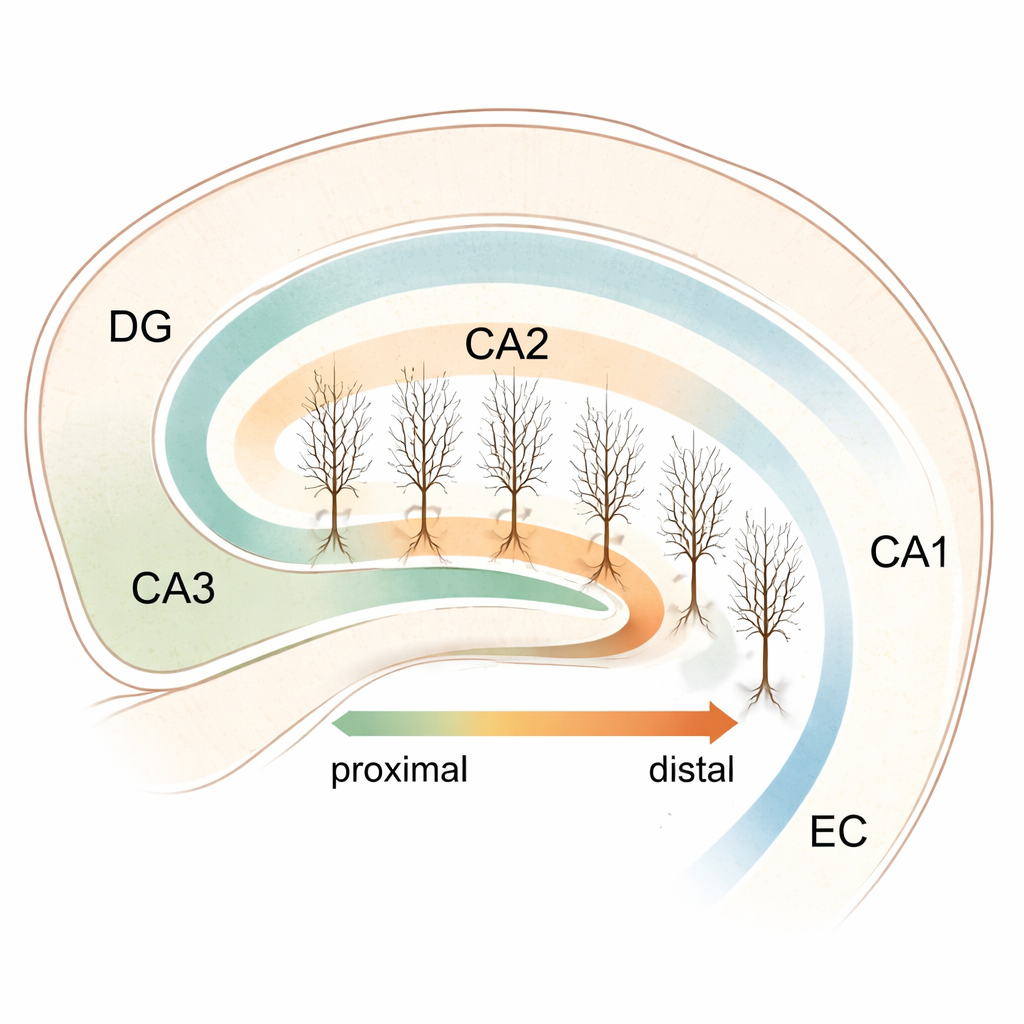
A Hidden Region in the Memory Circuit
CA2 sits between two major hippocampal hubs—CA3, which helps generate fast network bursts, and CA1, which is tightly linked to the slower, rhythmic waves that occur during movement and memory processing. CA2 has been tied to social memory, aggression control, and spatial orientation, yet its internal organization has remained murky. To probe this, the researchers grew thin slices of mouse hippocampus together with the entorhinal cortex, a major input area, in long-term culture. This approach preserves much of the native wiring while avoiding damage from cutting fresh brain tissue, allowing them to study individual CA2 cells under stable conditions.
Same Shape, Different Internal Settings
Using a molecular marker called PCP4, the team precisely outlined the CA2 region and assigned each pyramidal neuron a "proximodistal" position: near the CA3 border (proximal) or closer to CA1 (distal), with values in between forming a continuous axis. They then filled and reconstructed individual cells in 3D to compare their branching patterns. Despite earlier hints from other studies that CA2 structure might vary, they found no strong link between a cell’s position and its overall dendritic shape—numbers of branches, total length, and branching points remained broadly similar across the strip. In parallel, they measured spontaneous excitatory signals arriving at these neurons and again saw no clear gradient: the basic size and frequency of incoming synaptic events were relatively uniform from one end of CA2 to the other. This suggested that if CA2 is functionally partitioned, the key differences might lie in internal electrical settings rather than in raw wiring or input strength.
Gradual Shift in Electrical Excitability
When the researchers injected currents directly into CA2 neurons and monitored how their membranes responded, clear positional trends emerged. Cells near CA3 had higher input resistance, meaning small currents produced larger voltage changes, and they were less prone to a "sag"—a characteristic rebound seen when the membrane is briefly driven more negative. Moving toward CA1, input resistance dropped, while the sag and related rebound became more prominent. Action potentials also changed systematically: distal cells needed less current to fire, produced spikes more readily at the same input level, and showed subtle shifts in spike shape. In other words, CA2 neurons share broadly similar anatomy but operate with gradually tuned electrical settings along the proximodistal axis, making some more excitable and dynamically responsive than others.
Tuning In to Different Brain Wave Bands
One of the most striking findings involved how these cells respond to rhythmic inputs at different frequencies—a property known as subthreshold resonance. By driving the membrane with gentle sinusoidal currents sweeping from slower to faster cycles, the team could see at which frequency each cell’s voltage oscillation was amplified the most. Proximal CA2 neurons showed little or no preference; they behaved like generalists. Distal neurons, however, increasingly exhibited clear resonance peaks that shifted from the very slow delta range toward the lower theta band, around a few cycles per second. Because theta rhythms dominate during exploration, navigation, and memory encoding, this gradient suggests that distal CA2 cells are naturally tuned to lock onto these behavioral brain waves, likely through graded activity of ion channels that also produce the sag response. 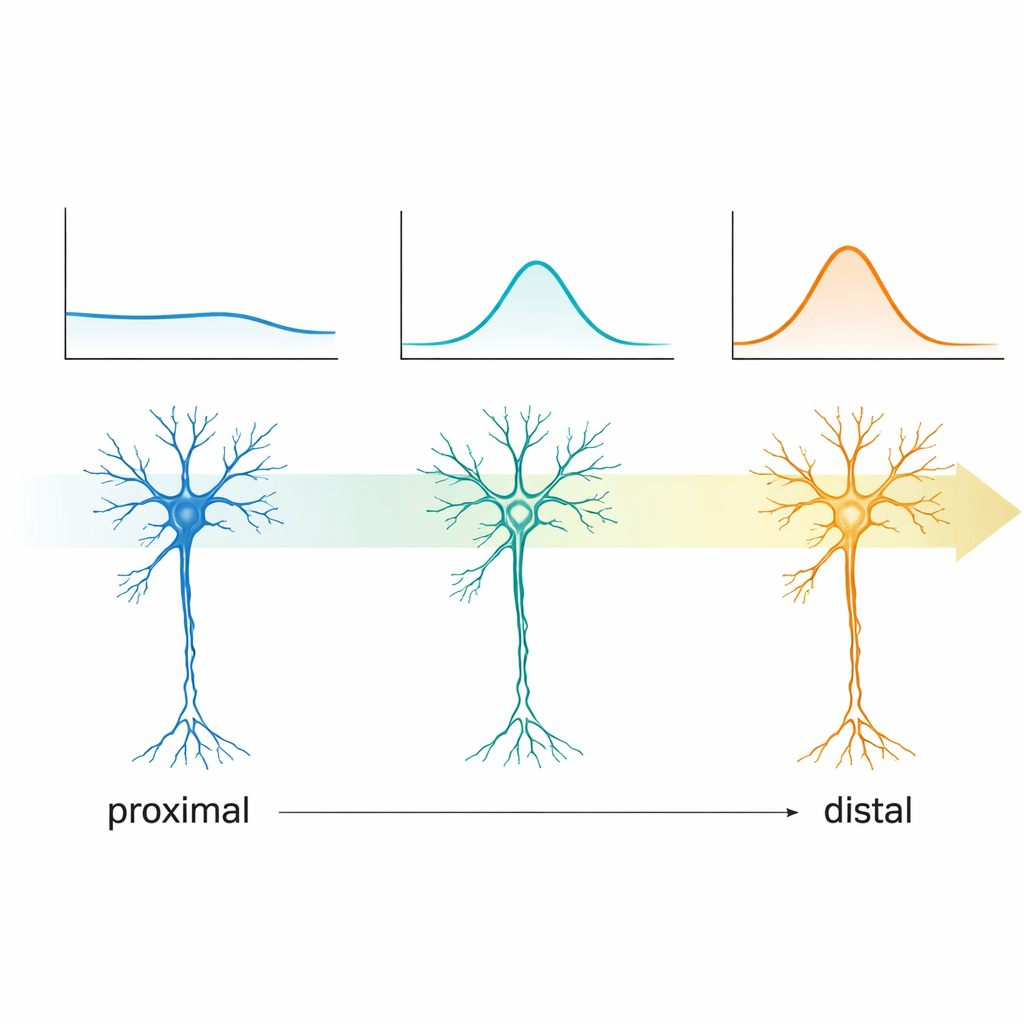
A Subtle Gradient with Big Network Consequences
Taken together, the work reveals CA2 not as a uniform strip of identical neurons, but as a smooth transition zone between two distinct operating modes in the hippocampus. Near CA3, CA2 cells lack strong resonance and may be better suited to participate in brief, fast events such as sharp-wave ripples, which help replay memories during rest and sleep. Toward CA1, cells become more responsive to theta rhythms and may preferentially couple to entorhinal inputs that carry information about position, context, and ongoing experience. For the non-specialist, the message is that even over tiny distances in the brain, neurons can be finely tuned to different "stations" on the dial of brain waves, allowing a small region like CA2 to flexibly route and shape information that underpins memory, navigation, and social behavior.
Citation: Kruse, P., Eichler, A., Brockmeyer, K. et al. Functional transition of CA2 pyramidal neurons along the proximodistal axis determines resonance frequency preference. Sci Rep 16, 7172 (2026). https://doi.org/10.1038/s41598-026-39754-3
Keywords: hippocampus, CA2 neurons, brain oscillations, theta rhythm, memory circuits