Clear Sky Science · en
The antibacterial effectiveness of fourth-generation poly-amidoamine dendrimers-loaded with amoxicillin in combating methicillin-resistant Staphylococcus aureus
Why these tiny particles matter
Drug-resistant infections are one of the biggest threats to modern medicine, making once-routine illnesses harder and more dangerous to treat. Among the worst offenders is methicillin-resistant Staphylococcus aureus (MRSA), a bacterium that can turn simple skin infections or hospital procedures into life-threatening events. This study explores a clever way to revive the usefulness of an everyday antibiotic, amoxicillin, by packaging it inside specially designed nano-sized carriers called dendrimers. The work suggests that smart drug packaging could give old antibiotics new life against some of our most stubborn germs. 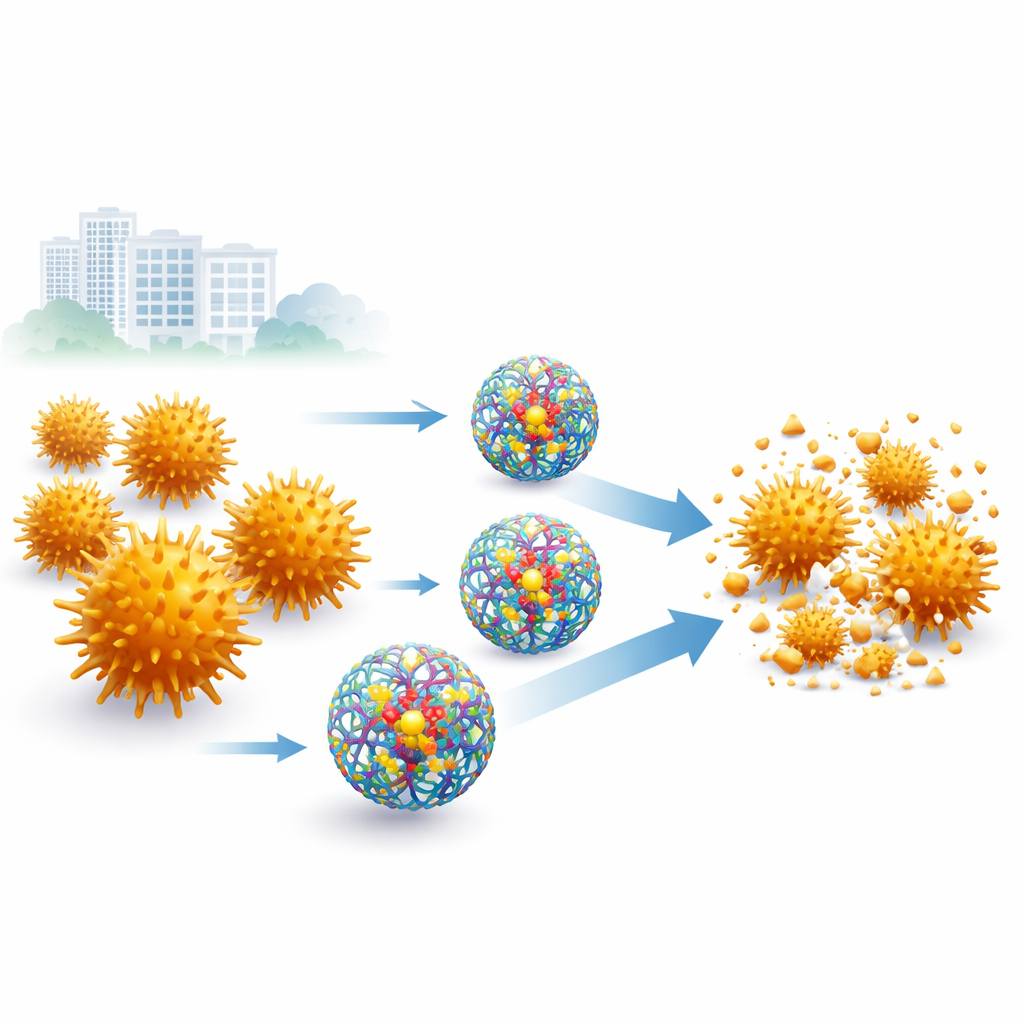
A stubborn germ that won’t go away
MRSA is a form of Staphylococcus aureus that has learned to withstand many commonly used antibiotics, including the widely prescribed β-lactam group to which amoxicillin belongs. It causes a range of illnesses, from skin and soft tissue infections to bone, heart, and device-related infections, and is linked with high rates of complications and death, especially in older adults. MRSA’s success comes from both its genetic resistance to drugs and its ability to produce toxins and build slimy protective layers called biofilms, which shield bacterial communities and make them up to a thousand times harder to kill. Because developing new antibiotics is slow and expensive, researchers are looking for smarter ways to deliver existing drugs more effectively.
A nano-sized delivery vehicle
The team focused on dendrimers, tree-like, highly branched molecules that can be engineered at the nanoscale. They used a fourth-generation poly-amidoamine (PAMAM G4) dendrimer that is water-soluble and can carry other molecules in its interior. By mixing this dendrimer with amoxicillin in a one-to-one ratio, they formed nanoparticles in which the antibiotic is encapsulated rather than left exposed. Detailed laboratory tests showed that the resulting particles were about 219 nanometers across—far smaller than most cells—with a uniform size distribution, a stable surface charge, and a high drug-loading efficiency of about 90%. Electron microscopy revealed that both empty and drug-filled dendrimers formed nearly spherical particles, confirming that the formulation was well structured. 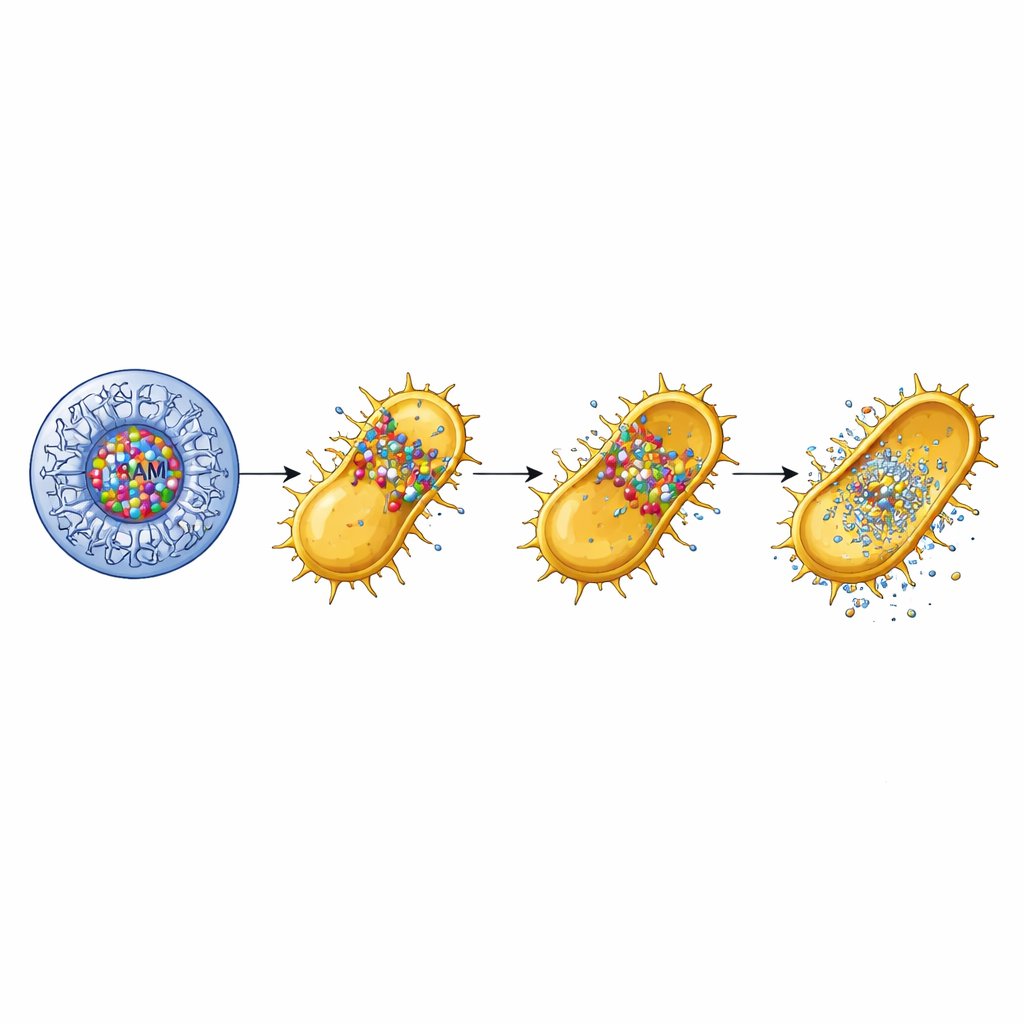
Slow release and stronger punch
To understand how this packaging changed the behavior of amoxicillin, the researchers studied how the drug leaked out of the dendrimers in a salt solution over eight hours. Compared with free amoxicillin, which released only about a third of its content in that time, the dendrimer-based formulation released over 80% in a gradual, sustained way. This meant that the antibiotic could stay available longer instead of washing away quickly. When the team tested the formulation against MRSA in growth experiments, the dendrimer-loaded amoxicillin stopped bacterial growth at relatively low concentrations, while free amoxicillin alone barely slowed it down and the empty dendrimer had only modest effects. Standard plate tests that measure clear zones where bacteria cannot grow showed much larger inhibition areas for the combined nanoparticles than for either ingredient alone, signaling a strong boost in antibacterial power.
Disarming the bacteria’s weapons
Beyond simply killing bacteria, the researchers examined whether the nanoparticles could also blunt MRSA’s virulence—its ability to cause harm. MRSA produces toxins that punch holes in red blood cells, a process called hemolysis, which helps it invade tissues and spread. The study found that neither amoxicillin alone nor the empty dendrimer could block this activity. However, when amoxicillin was encapsulated in the G4 dendrimer, hemolysis was completely prevented across all tested doses. The team also looked at biofilms, the sticky bacterial communities that cling to surfaces and resist treatment. The G4-amoxicillin nanoparticles reduced biofilm formation by about 70%, compared with only 20% for the empty dendrimer and virtually no effect from free amoxicillin. These results suggest that the nano-formulation not only kills MRSA more efficiently but also strips away important tools the bacterium uses to persist and damage the host.
What this could mean for future treatments
Taken together, the findings indicate that packaging amoxicillin inside PAMAM G4 dendrimers turns a largely ineffective drug against MRSA into a potent antibacterial and anti-virulence agent. The nanoparticles deliver the antibiotic in a sustained manner, help it reach and act on the bacteria more effectively, and reduce dangerous behaviors like toxin release and biofilm formation. While this work was done in the laboratory and further studies on stability, dosing, and safety in animals are still needed, it points toward a promising strategy: using smart nanocarriers to recycle familiar antibiotics for modern drug-resistant infections, potentially buying valuable time in the fight against MRSA and related superbugs.
Citation: Alenazi, N., Alhabardi, S.A., Binsuwaidan, R. et al. The antibacterial effectiveness of fourth-generation poly-amidoamine dendrimers-loaded with amoxicillin in combating methicillin-resistant Staphylococcus aureus. Sci Rep 16, 9242 (2026). https://doi.org/10.1038/s41598-026-39752-5
Keywords: MRSA, antibiotic resistance, nanoparticles, dendrimers, amoxicillin