Clear Sky Science · en
Chronic inflammation promotes gastric cancer progression via ADAM10-mediated cleavage of CX3CL1
Why long-lasting stomach irritation matters
Many people live for years with ongoing stomach irritation caused by infection, diet, or other health problems. This constant low-level irritation, known as chronic inflammation, is a silent risk factor for stomach (gastric) cancer, one of the deadliest cancers worldwide. The study summarized here asks a focused question: how does chronic inflammation push early stomach tumors to grow faster and spread? By tracing one specific chemical signal and the enzyme that activates it, the researchers uncover a potential weak point that future treatments might target.
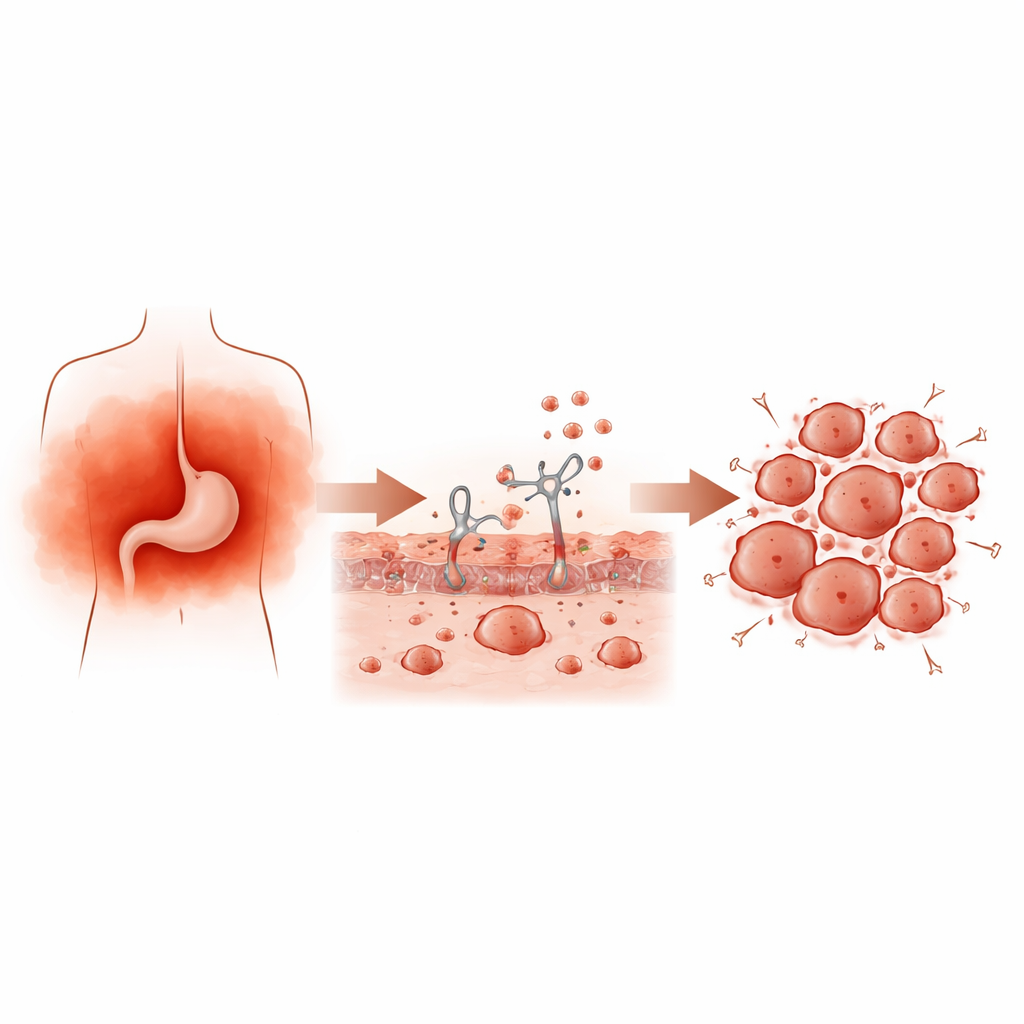
From irritation to faster-growing tumors
To mimic long-lasting inflammation, the team repeatedly exposed mice to substances that trigger immune reactions, then implanted gastric cancer cells under the skin. Compared with control animals, mice with chronic inflammation developed larger, heavier tumors packed with dividing cells and immune-cell infiltrates. In cell-culture experiments, blood serum taken from rats with different levels of inflammation made human gastric cancer cells grow and form colonies more vigorously. Together, these results show that inflammatory signals circulating in the body can directly accelerate tumor growth.
Hunting for the key troublemaking signal
The next step was to work out which inflammatory messenger molecules were doing the most harm. Using a broad protein array, the researchers compared many cytokines in tumors from inflamed and non-inflamed mice and found nine that were clearly increased. By cross-checking public patient datasets, they zeroed in on one standout candidate: CX3CL1, a chemokine that helps guide immune cells. CX3CL1 levels were higher in gastric tumors than in normal stomach tissue, were especially elevated in more advanced tumors, and were linked to poorer survival in patients. Measurements in mouse tumors confirmed that chronic inflammation raised CX3CL1 levels locally, suggesting that this single signal might help explain the link between inflammation and worse outcomes.
Which cells send and receive the signal?
Using single-cell RNA sequencing, the investigators mapped gene activity cell by cell inside tumors. They discovered that CX3CL1 was produced mainly by the cancer cells themselves rather than by surrounding immune or support cells. Its partner receptor, CX3CR1, was found on several cell types, but under chronic inflammation it became more prominent on tumor cells. A specific tumor subpopulation expanded in inflamed tumors showed high CX3CL1 expression and signs of boosted protein production and growth. In lab tests, adding purified CX3CL1 directly to gastric cancer cells increased their survival, colony formation, and movement. Blocking CX3CR1 dampened these effects, indicating that cancer cells were both sending and responding to this growth-promoting signal.
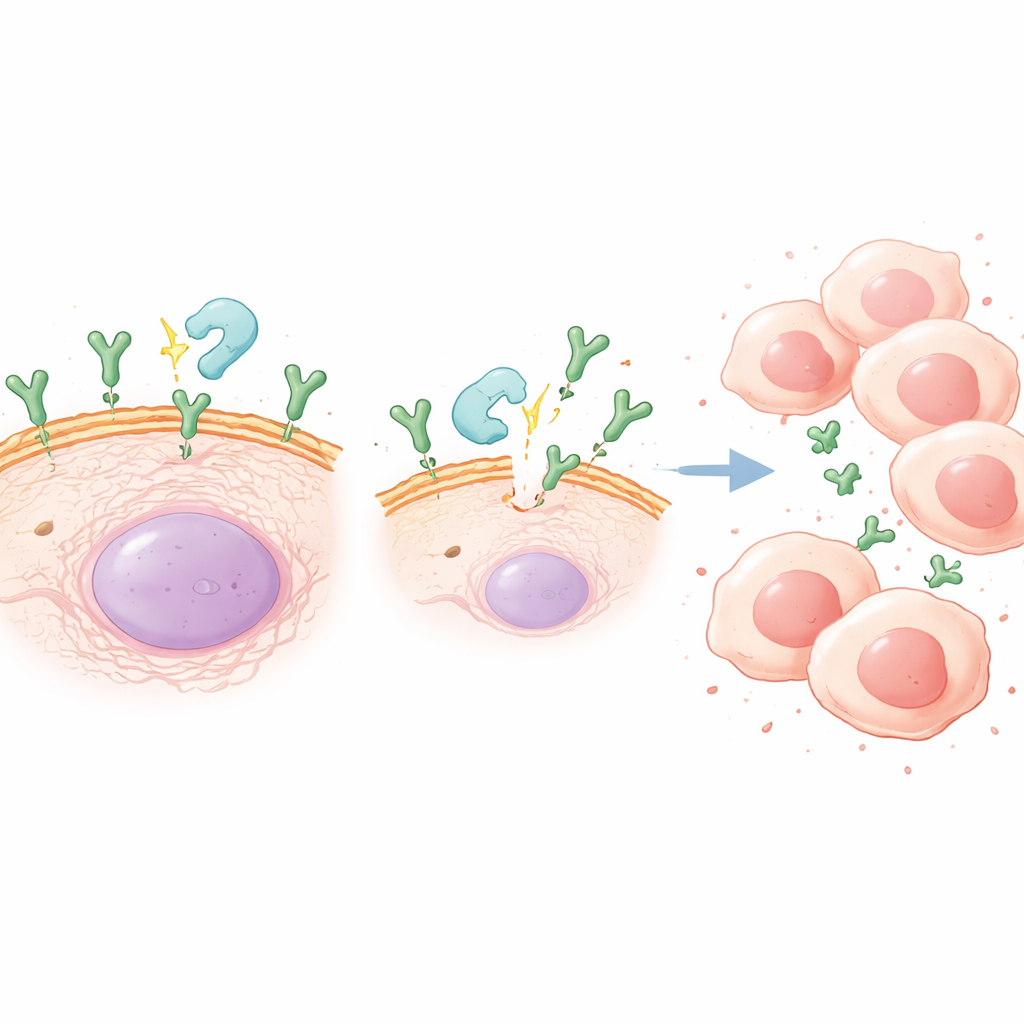
The enzyme that turns up the danger
CX3CL1 exists as a form anchored in the cell membrane and as a free-floating soluble form that can spread through tissue. The soluble form is created when an enzyme called ADAM10 slices the membrane-bound version. The study found that chronic inflammation boosted ADAM10 levels in mouse tumors and in gastric cancer cells exposed to an inflammatory trigger in the lab. When the researchers treated cells with an ADAM10 inhibitor, the amount of soluble CX3CL1 in the surrounding fluid dropped, and the inflammation-driven increases in cancer cell growth and movement were largely reversed. In mice with chronic inflammation, blocking ADAM10 slowed tumor progression and reduced CX3CL1 levels in tumors, strengthening the case that this enzyme-driven "shedding" step is critical.
What this means for patients with chronic stomach problems
Taken together, the work outlines a simple chain of events: ongoing inflammation in and around the stomach switches on ADAM10 in gastric cancer cells; ADAM10 then cuts loose more soluble CX3CL1, which in turn feeds back onto cancer cells to help them multiply and migrate. For a layperson, this is like an irritated environment teaching tumor cells to both shout and listen to a growth-promoting message. Because both ADAM10 and CX3CL1 are identifiable molecules with existing drug interest, this newly described "ADAM10/CX3CL1 axis" offers a tangible target for therapies aimed at slowing or preventing inflammation-driven gastric cancer progression.
Citation: Song, Z., Liu, Y., Wang, Z. et al. Chronic inflammation promotes gastric cancer progression via ADAM10-mediated cleavage of CX3CL1. Sci Rep 16, 9169 (2026). https://doi.org/10.1038/s41598-026-39743-6
Keywords: gastric cancer, chronic inflammation, chemokines, tumor microenvironment, ADAM10