Clear Sky Science · en
Structural investigations of sandwich coating system containing self-healing core–shell nanofibers resistant to corrosive environment
Why Metals Need Help to Stay Rust-Free
Bridges, ships, pipelines, and storage tanks all rely on thin paint-like coatings to keep steel from rusting. But once those coatings are scratched or cracked, salty water and oxygen can sneak through, starting corrosion that is costly and sometimes dangerous. This study explores a new kind of “smart” protective coating that can sense damage and automatically heal itself, helping metal structures last longer with less maintenance.

A Sandwich Shield for Steel
The researchers designed a three-layer "sandwich" coating for mild steel. The top and bottom layers are based on a common epoxy paint that sticks well to metal. Into this paint they mixed ultra-thin sheets of modified graphene oxide, which act like overlapping plates to make it harder for water and ions to weave their way down to the steel. Between these two layers they added a thin middle layer made of special fibers. Each fiber has a soft liquid core and a solid shell, forming countless tiny reservoirs of healing material hidden inside the coating.
Tiny Fibers That Store Healing Liquid
To create these fibers, the team used a technique called coaxial electrospinning, which stretches two liquids into long core–shell threads. The shell is made of polyvinyl alcohol, a water-loving polymer, while the core holds a silicone-based liquid (PDMS) that can flow into damaged areas and form a protective film. By changing how concentrated the shell solution was (7, 10, or 15 percent), they controlled the thickness of the fibers and how much healing liquid each one could carry. Microscopy images confirmed that the fibers had a clean core–shell structure and that higher shell concentration produced thicker, more evenly distributed fibers loaded with more healing agent.
How the Self-Healing Coating Works
When the coated steel is placed in a salty solution, water and corrosive ions slowly try to pass through the top epoxy–graphene layer. If they reach the middle fiber layer, the water begins to dissolve the outer shell of the fibers. This releases the silicone liquid inside, which seeps into cracks and pores and spreads along the damaged paths. At the same time, silane groups in the system react with water and the surrounding epoxy to form new siloxane bonds, tightening the polymer network and creating a dense, water-resistant barrier that blocks further attack.
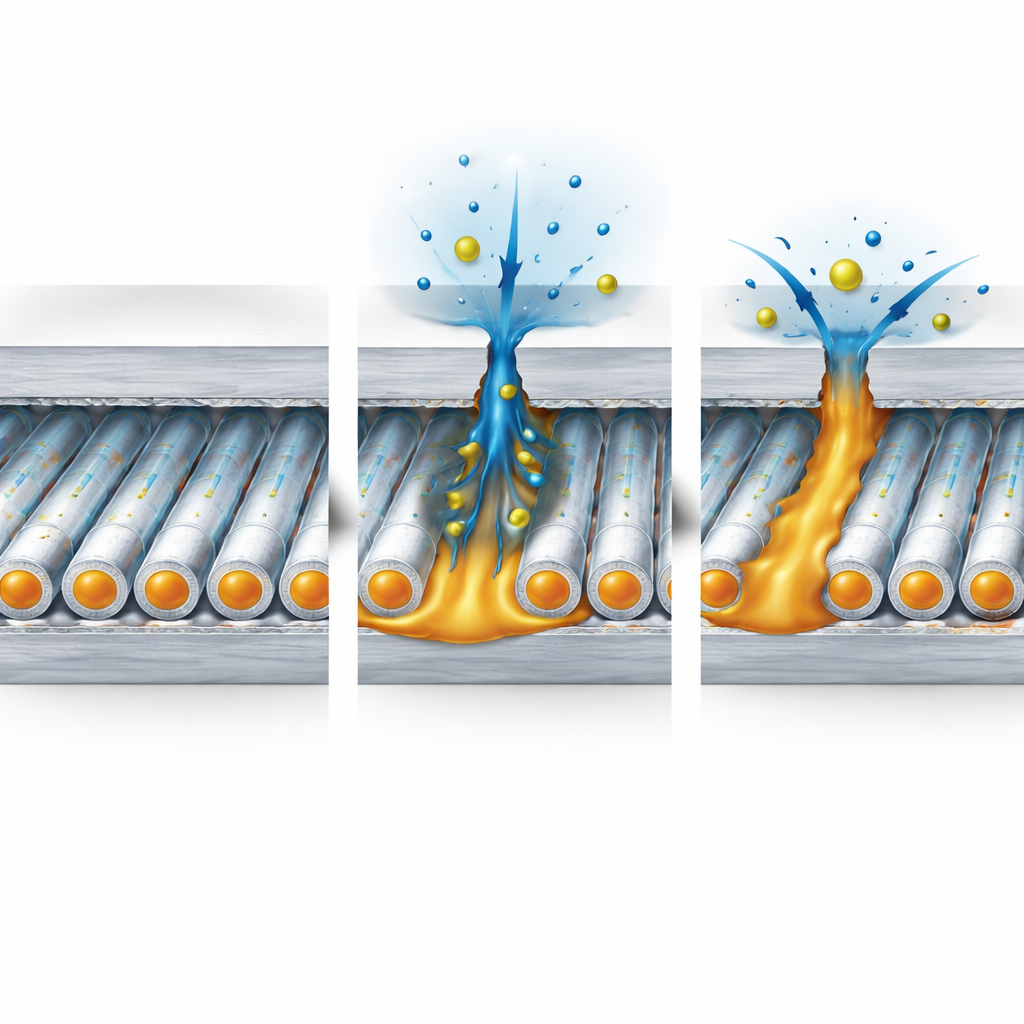
Putting the Smart Coating to the Test
To see how well the coatings worked, the authors carried out long-term corrosion tests in salt solutions and in a salt spray chamber, both on intact panels and on panels deliberately scratched down to the metal. They used electrochemical measurements to track how easily current could pass through the coating—a strong indicator of how well it was still protecting the steel. Coatings with more robust fibers (made with 15 percent shell solution) showed the highest resistance and kept that protection over nearly five months of immersion. Even when scratched, these coatings could regain much of their barrier strength within about a day, as the released liquid filled in the cut and slowed further rusting. Microscopic images of the scratch region after 480 hours of salt spray showed almost complete closure and very few corrosion products for the best-performing formulation.
Why the Fiber Design Matters
The comparison between the three fiber formulations revealed a clear pattern. Thinner fibers with less healing liquid (7 percent shell) offered only modest repair, and rust spread more quickly from the scratch. Intermediate fibers (10 percent shell) improved the situation but still allowed more damage over time. The thickest and most densely packed network (15 percent shell) provided the most healing liquid and the most continuous coverage, leading to the slowest corrosion, the smallest change in electrical behavior, and the cleanest scratch area in both imaging and chemical analysis. This shows that not just the presence, but also the amount and distribution of healing reservoirs, strongly controls how well the coating can repair itself.
What This Means for Real-World Structures
For non-specialists, the main message is that it is now possible to build protective paints that do more than just sit on the surface: they can actively respond when damaged. By combining barrier-forming graphene-filled epoxy with a hidden layer of liquid-filled fibers, this work demonstrates a coating that can close scratches and maintain high corrosion resistance for extended periods in harsh salty environments. While questions remain about long-term durability and large-scale manufacturing, such self-healing sandwich coatings could one day help keep ships, bridges, and industrial plants safer and in service longer, with fewer costly repairs.
Citation: Madani, S.M., Sangpour, P., Vaezi, M.R. et al. Structural investigations of sandwich coating system containing self-healing core–shell nanofibers resistant to corrosive environment. Sci Rep 16, 9361 (2026). https://doi.org/10.1038/s41598-026-39735-6
Keywords: self-healing coatings, corrosion protection, graphene oxide epoxy, core–shell nanofibers, smart materials