Clear Sky Science · en
Autism spectrum disorder-associated Sema5A p.Arg676Cys drives Arf6/FE65 signaling and aberrant cell morphogenesis
How a Tiny Change May Disrupt Brain Wiring
Autism spectrum disorder affects how people communicate, interact, and respond to the world around them, yet the biological steps that lead from a DNA change to altered brain function are often mysterious. This study digs into one such genetic change in a brain "guidance" protein called Sema5A and shows, in unusually fine detail, how a single swapped building block in that protein can push young nerve cells to grow overly long, tangled branches. Understanding this microscopic wiring error may eventually help scientists design treatments that nudge brain cells back toward a healthier shape.
Brain Cells That Grow Too Far
During brain development, young nerve cells send out long, thin extensions that search for the right partners, forming the circuits that underlie thought and behavior. This growth must be tightly controlled: too little branching and cells fail to connect; too much and circuits can become noisy or miswired. Earlier work had shown that an autism-linked version of Sema5A, in which one amino acid at position 676 is changed from arginine to cysteine, makes nerve cells in culture grow unusually long processes. The new study set out to uncover which internal switches and helper proteins this mutant Sema5A uses to drive such runaway growth.
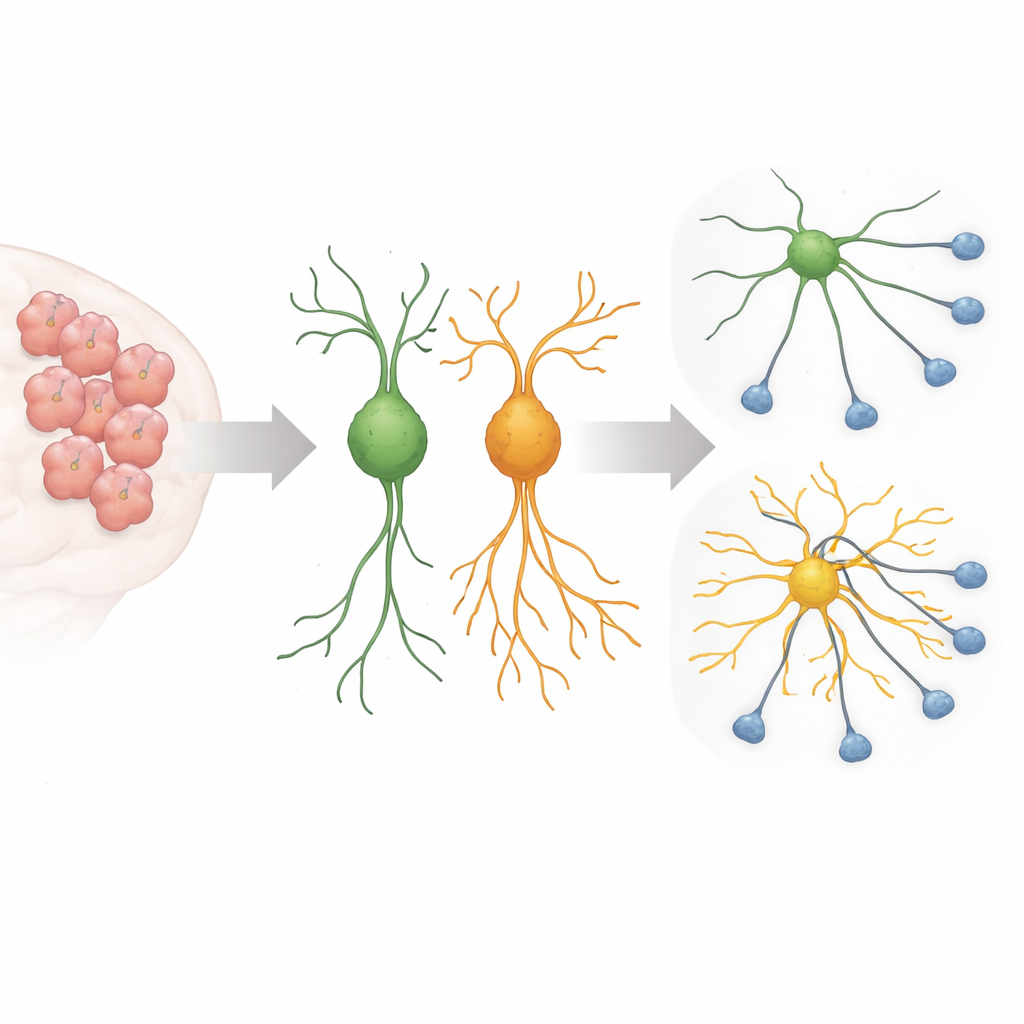
A Misleading Signal Inside the Cell
The researchers focused on a small molecular switch called Arf6 and a scaffold protein called FE65, both known to help shape cell membranes and the inner skeleton that supports cell extensions. Using a CRISPR–Cas13 system to selectively reduce these proteins in mouse nerve-like cells and in primary mouse brain cells, they found that turning down Arf6 or FE65 sharply cut back the excessive outgrowth caused by the mutant Sema5A. The long, spidery processes shrank toward more typical lengths, and markers of neuronal maturation dropped, suggesting that the mutation’s effect depends heavily on this particular signaling route.
Connecting to the Cell’s Shape Engine
Inside nerve cells, a family of molecules acts as a shape "engine" by controlling actin, the main structural filament in cell branches. A key member of this family, Rac1, normally becomes active to help neurites extend, but too much activity can drive uncontrolled growth. The team showed that in cells carrying normal Sema5A, Arf6 is needed for ordinary Rac1 activation during healthy outgrowth, whereas FE65 is not essential. With the mutant Sema5A, however, both Arf6 and FE65 became critical: lowering either one, or flooding the cell with just the FE65 region that binds its partner ELMO2, reduced the abnormally high Rac1 activity back toward normal. This implies that the harmful version of Sema5A taps specifically into an Arf6–FE65–ELMO2–DOCK5 complex to over-stimulate Rac1 and push neurites to grow too far.
A Crowded Hub of Signaling Partners
To see how these pieces fit together, the scientists also looked at ELMO2-based "signalosomes"—clusters of proteins that assemble to relay growth cues. When Arf6 or FE65 was reduced, cells expressing mutant Sema5A formed fewer of these ELMO2 complexes in their bodies and growing tips, consistent with the idea that Arf6 and FE65 help build the machinery that turns the Sema5A mutation into a physical change in cell shape. The work fits into a broader picture in which small molecular switches like Arf6 and Rac1, helped by scaffolds such as FE65, act as central hubs linking many autism-associated genes to the final architecture of brain circuits.
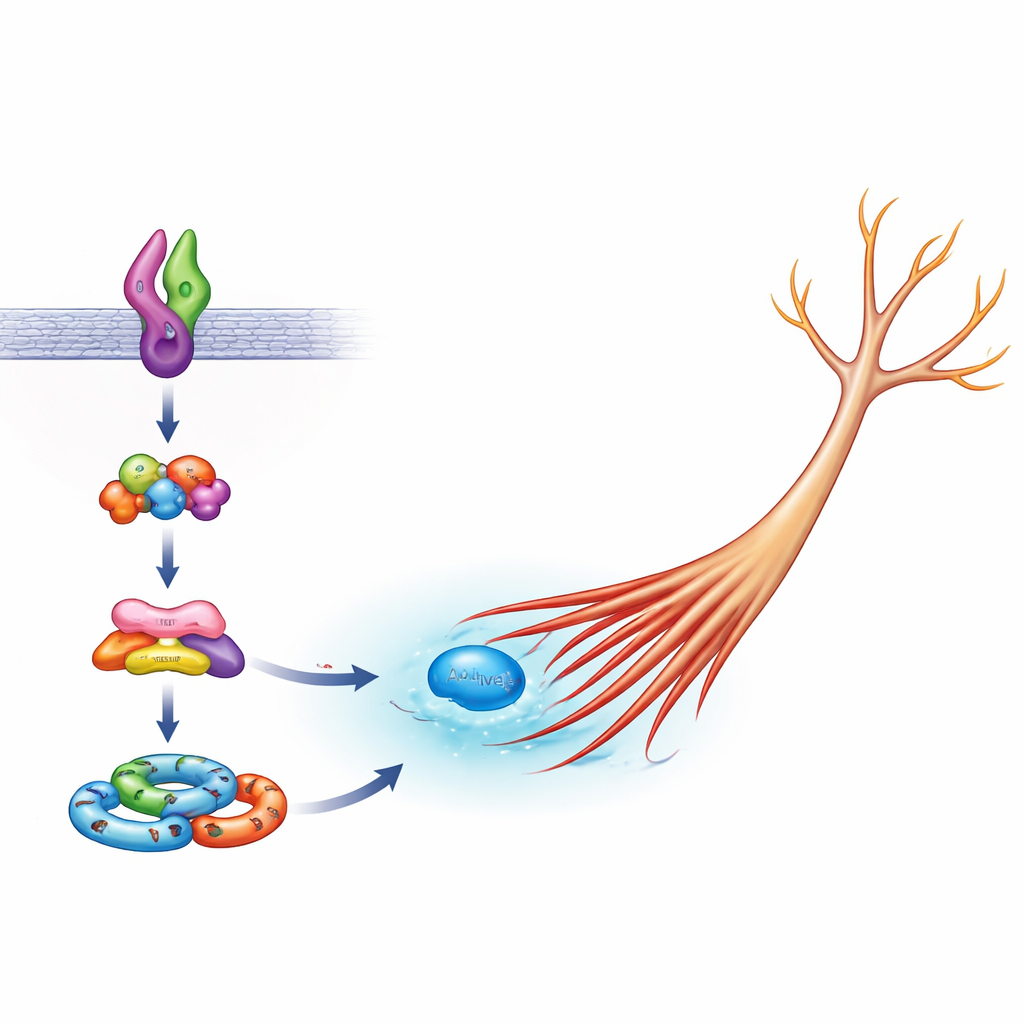
Why This Microscopic Chain Matters
To a non-specialist, the alphabet soup of protein names can seem remote from the lived experience of autism. Yet this study offers a concrete bridge: it traces how a precise genetic change in Sema5A can over-activate a specific chain of helper molecules, causing nerve cells to extend their branches too far and potentially wire the brain in atypical ways. By pinpointing Arf6, FE65, and the ELMO2 signal complex as crucial links in this chain, the work highlights potential future drug targets. In principle, medicines that gently tune down this overactive pathway could one day help correct the underlying cell-shape changes associated with Sema5A-related forms of autism, adding an important piece to the larger puzzle of neurodevelopmental disorders.
Citation: Takahashi, M., Yako, H., Miyamoto, Y. et al. Autism spectrum disorder-associated Sema5A p.Arg676Cys drives Arf6/FE65 signaling and aberrant cell morphogenesis. Sci Rep 16, 9423 (2026). https://doi.org/10.1038/s41598-026-39722-x
Keywords: autism spectrum disorder, Sema5A mutation, neuronal morphogenesis, Rac1 signaling, Arf6 FE65 pathway