Clear Sky Science · en
Lethal effects of ivermectin structures on malaria vectors and in silico analysis of interactions with their glutamate-gated chloride ion channels
Turning a Deworming Drug into a Mosquito Killer
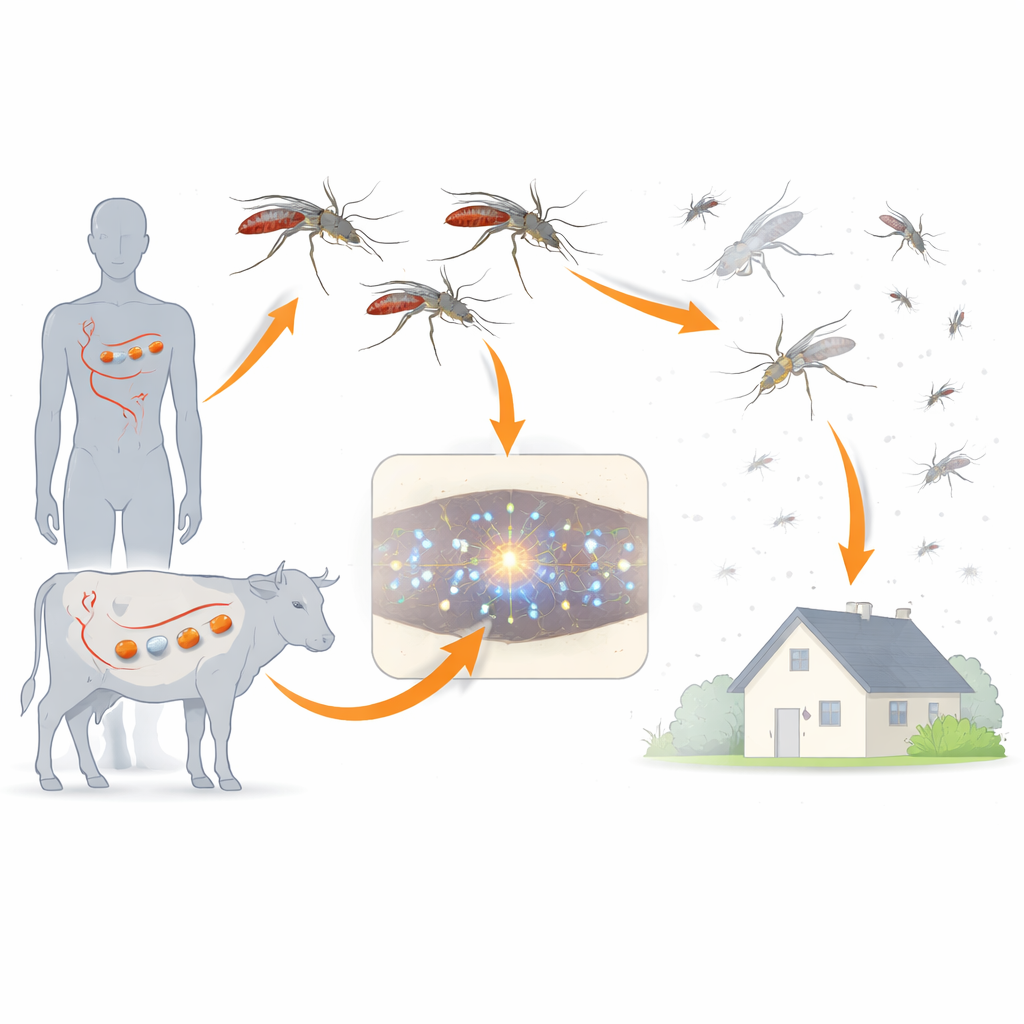
Ivermectin is best known as a medicine against parasitic worms in humans and livestock, but it has a surprising side effect: mosquitoes that drink treated blood often die. That raises an intriguing idea for malaria control—could giving people or animals ivermectin thin out local mosquito populations enough to cut transmission? This study digs deep into how ivermectin and related molecules kill malaria-carrying Anopheles mosquitoes, and what changes might someday let mosquitoes escape its effects.
Why Killing Mosquitoes Through Blood Matters
Traditional malaria control relies on bed nets and insecticide sprays, which target mosquitoes from the outside. Ivermectin offers a different strategy: when humans or animals take the drug, their blood becomes deadly to blood-feeding mosquitoes. The researchers focused on two important Southeast Asian malaria vectors, Anopheles dirus and Anopheles minimus, which differ widely in how easily ivermectin kills them. By comparing these species, the team hoped to understand why the drug is more lethal in some mosquitoes than others and how its chemical structure influences that lethal punch.
Testing Which Parts of the Drug Matter
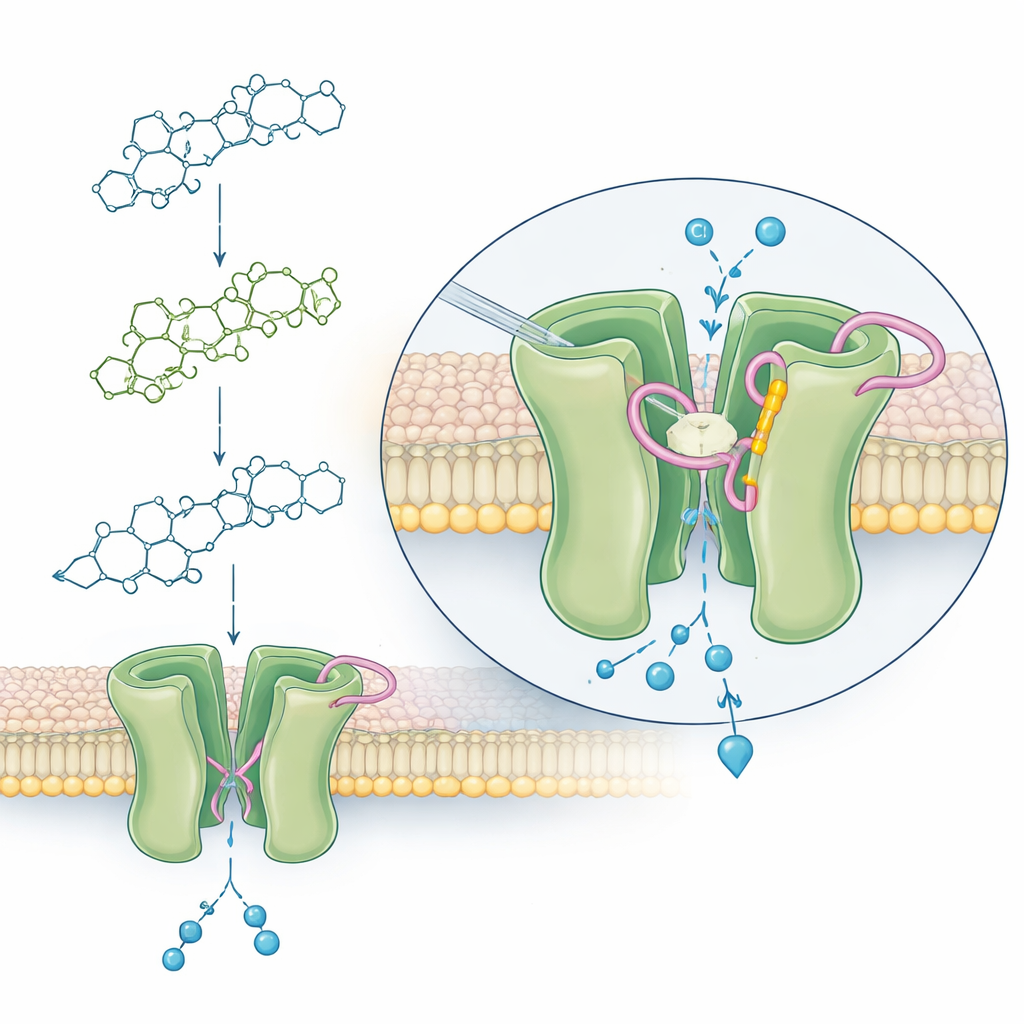
Ivermectin is a bulky molecule built around a large ring with two attached sugar rings. The team compared full ivermectin (with both sugars) to a version with only one sugar ring (called a monosaccharide) and to a stripped-down core with no sugars (aglycone). They fed these compounds, at different concentrations, to thousands of mosquitoes in blood meals and tracked survival for ten days. Full ivermectin was highly lethal, especially to An. minimus, while the monosaccharide was much weaker and the aglycone had almost no killing power at realistic doses. In other words, removing one sugar ring turned a powerful mosquito killer into a much milder one, and removing both nearly erased the effect.
Peeking Inside the Mosquito’s Nerve Gate
Ivermectin acts on a tiny gate in nerve and muscle cells called the glutamate-gated chloride (GluCl) channel. When this gate is forced open, chloride ions rush in, electrical activity collapses, and the mosquito becomes paralyzed and dies. Using advanced protein-structure prediction and computer docking, the researchers built three-dimensional models of the Anopheles GluCl channel and simulated how ivermectin and its variants nestle into the channel. They found that, in mosquitoes, a specific part of the protein loop near the pore—containing an amino acid called threonine at position 304—can form a hydrogen bond with the second sugar ring of ivermectin. This bond, plus nearby weak attractions, appears to stabilize a drug-bound shape that keeps the channel open.
Why Some Versions Kill and Others Do Not
The simulations showed a consistent pattern: full ivermectin and three major ivermectin breakdown products found in humans extend deeply into the channel, allowing their second sugar ring to interact closely with the key loop. These forms all created strong predicted binding and, in earlier work, proved just as lethal to mosquitoes as the parent drug. The monosaccharide, lacking that second sugar ring, could still touch the loop weakly but could not form the same stabilizing bond, matching its much lower killing power. The aglycone did not contact the loop at all, aligning with its failure to kill mosquitoes in feeding tests. Across all structures, one shared interaction with another part of the channel (in the opposing subunit) also emerged as important, suggesting several contact points work together to lock the gate open.
Looking Ahead to Resistance and Better Tools
The findings suggest that the second sugar ring of ivermectin—and its ability to bond with a specific loop of the mosquito GluCl channel—is central to its mosquito-killing effect. This improved picture of how the drug fits into the channel highlights potential weak spots where future mutations could reduce binding and lead to resistance. It also hints at ways chemists might tweak ivermectin-like molecules to keep or enhance their power against mosquitoes. While the study relies on computer models that still need experimental confirmation, it strengthens the case for ivermectin-based strategies as part of the malaria-control toolbox and points to molecular features that will be crucial to watch as such strategies are rolled out.
Citation: Nguyen, M.N., Jones, A.K., Hotwagner, D. et al. Lethal effects of ivermectin structures on malaria vectors and in silico analysis of interactions with their glutamate-gated chloride ion channels. Sci Rep 16, 8141 (2026). https://doi.org/10.1038/s41598-026-39698-8
Keywords: ivermectin, malaria vectors, Anopheles mosquitoes, ion channels, insecticide resistance