Clear Sky Science · en
BMSCs exosomes regulate pulmonary microvascular endothelial apoptosis via circRNA_43350/miR-342-5p in COPD
Why protecting fragile lung blood vessels matters
Chronic obstructive pulmonary disease, or COPD, slowly robs people of breath and independence. Cigarette smoke not only irritates the airways but also injures the delicate web of tiny blood vessels that wrap around the air sacs where oxygen enters the bloodstream. In this study, scientists asked whether natural repair packages released by bone marrow stem cells could shield these vulnerable lung vessels from smoke damage, and why this protection seems weaker in COPD. Their findings reveal a tiny circular genetic loop that acts like a molecular sponge, soaking up a harmful signal and helping lung vessels survive.
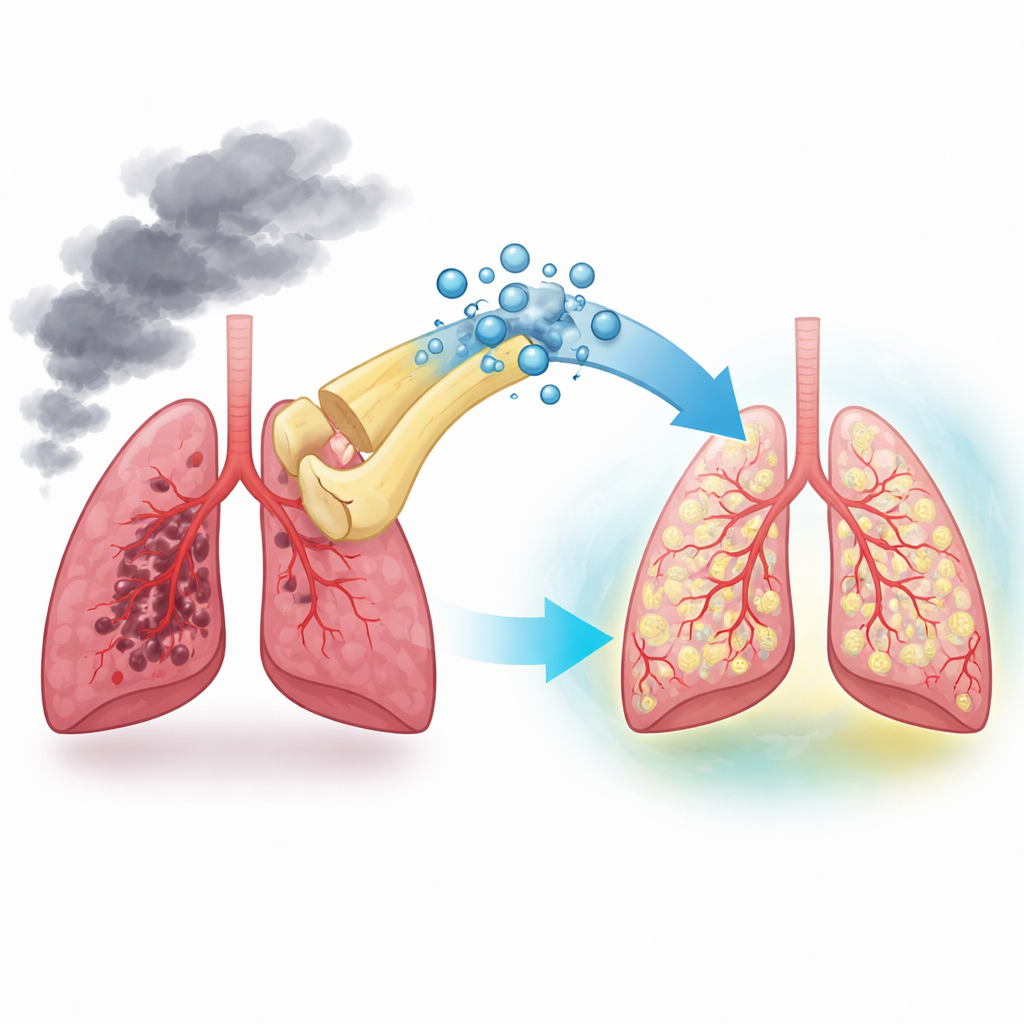
Small repair packages from stem cells
Bone marrow mesenchymal stem cells are known for their ability to calm inflammation and support tissue repair, not only by turning into other cell types but also by sending out microscopic bubbles called exosomes. These exosomes are loaded with proteins and genetic material and can be taken up by other cells as ready-made instructions. The researchers first isolated exosomes from mouse bone marrow stem cells and confirmed that they had the expected shape and size. They then showed that these exosomes were readily swallowed by lung microvascular endothelial cells—the cells that line the tiny lung blood vessels damaged in COPD.
Exosomes soften cigarette smoke injury in lungs
To mimic smoking-related COPD, the team exposed cultured lung vessel cells to cigarette smoke extract and created a mouse model of smoke-induced emphysema. In both systems, smoke strongly increased cell death in the vessel lining and led to enlarged, destroyed air sacs in the lungs. When exosomes from healthy stem cells were added, the balance of key survival proteins shifted toward protection, fewer endothelial cells underwent programmed death, and the emphysema-like damage in mouse lungs was partially reversed. These results showed that factors carried by healthy stem-cell exosomes can directly protect lung vessels and blunt structural lung injury.
A circular genetic loop at the heart of protection
Next, the scientists searched the exosome cargo for differences between healthy and COPD conditions, focusing on circular RNAs, a recently discovered class of stable, looped genetic molecules. Among nearly 14,000 circular RNAs detected, one called circRNA_43350 stood out: it was abundant in exosomes and lung tissue from healthy mice but markedly reduced in those with COPD. Artificially boosting circRNA_43350 levels in smoke-exposed vessel cells lowered their death rate and restored a healthier pattern of survival proteins. In COPD mice, delivering extra circRNA_43350 into the airways lessened emphysema-like changes and reduced cell death in the lung lining, pointing to a central protective role for this circular RNA.
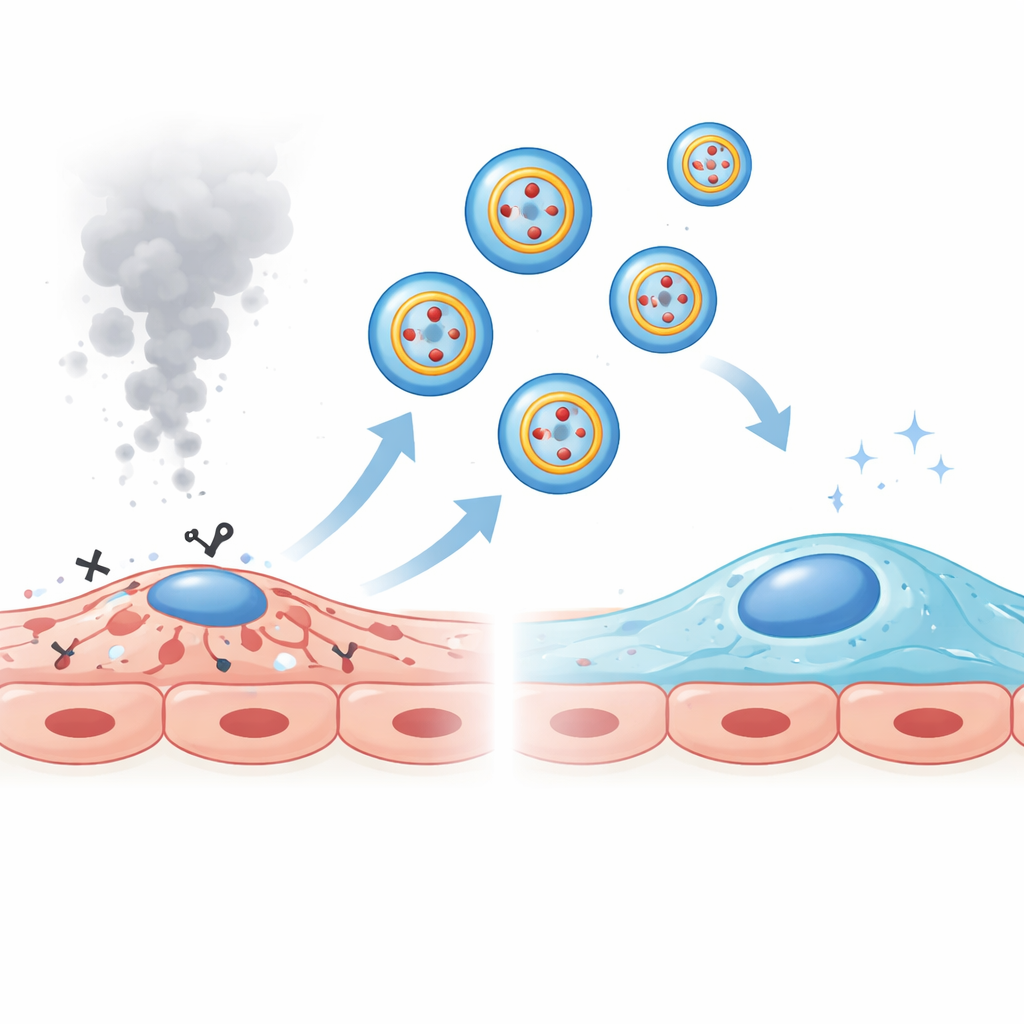
A molecular sponge that soaks up a harmful signal
Circular RNAs often work by binding small regulatory RNAs called microRNAs, preventing them from silencing their target genes. Using prediction tools and molecular reporter tests, the team found that circRNA_43350 directly binds a microRNA named miR-342-5p. In patients, COPD mice, and smoke-treated vessel cells, miR-342-5p levels were higher than normal, and blocking this microRNA reduced endothelial cell death. The researchers showed that circRNA_43350 and miR-342-5p restrain each other: extra circRNA_43350 drives miR-342-5p down and protects cells, whereas extra miR-342-5p weakens the benefit of circRNA_43350 and promotes cell death. This tug-of-war suggests that when circRNA_43350 is lost from exosomes in COPD, miR-342-5p is left free to push vulnerable lung vessel cells toward injury.
What this means for future COPD therapies
Together, these findings paint a picture in which healthy bone marrow stem cells ship protective exosomes loaded with circRNA_43350 to the lungs, where this circular RNA mops up excess miR-342-5p and helps the tiny blood vessels survive cigarette smoke stress. In COPD, exosomes carry less circRNA_43350, weakening this shield and allowing more damage and emphysema to develop. By pinpointing this microscopic repair loop, the work offers a new angle for therapy: engineered exosomes or other treatments that restore circRNA_43350 or restrain miR-342-5p could one day help preserve lung structure and breathing capacity in people living with COPD.
Citation: Zeng, Y., Song, Q., Yang, L. et al. BMSCs exosomes regulate pulmonary microvascular endothelial apoptosis via circRNA_43350/miR-342-5p in COPD. Sci Rep 16, 8183 (2026). https://doi.org/10.1038/s41598-026-39678-y
Keywords: COPD, lung endothelial cells, mesenchymal stem cell exosomes, circular RNA, microRNA