Clear Sky Science · en
γ-Fe2O3/MIL101(Fe)-NH2/COF-MT as a novel ternary photocatalyst for the selective oxidation of alcohols through a dual S-scheme pathway under sunlight irradiation
Turning Sunlight into Safer Chemistry
Chemists rely on reactions that turn simple starting materials into valuable ingredients for medicines, fragrances, and plastics. One of the most common steps is gently "tuning" alcohols into aldehydes, but traditional methods often use harsh, toxic oxidants and generate hazardous waste. This paper reports a new sunlight-driven catalyst that can perform the same transformation in a cleaner, more sustainable way, using air as the oxidant and a magnetically recoverable solid that can be reused many times.
A New Three-in-One Catalyst Particle
The research team built a tiny hybrid particle that combines three different materials into a single working unit: magnetic iron oxide, a metal–organic framework (MOF), and a covalent organic framework (COF). Iron oxide provides magnetism and also participates in light-driven chemistry. The MOF and COF are both highly porous, crystal-like networks assembled from metal nodes or organic building blocks, giving the catalyst enormous internal surface area where reactions can occur. By growing the COF as a thin outer layer on a modified iron-containing MOF anchored to iron oxide, the authors created a stable, mesoporous structure with channels that can host alcohol molecules and expose them efficiently to light and reactive sites. 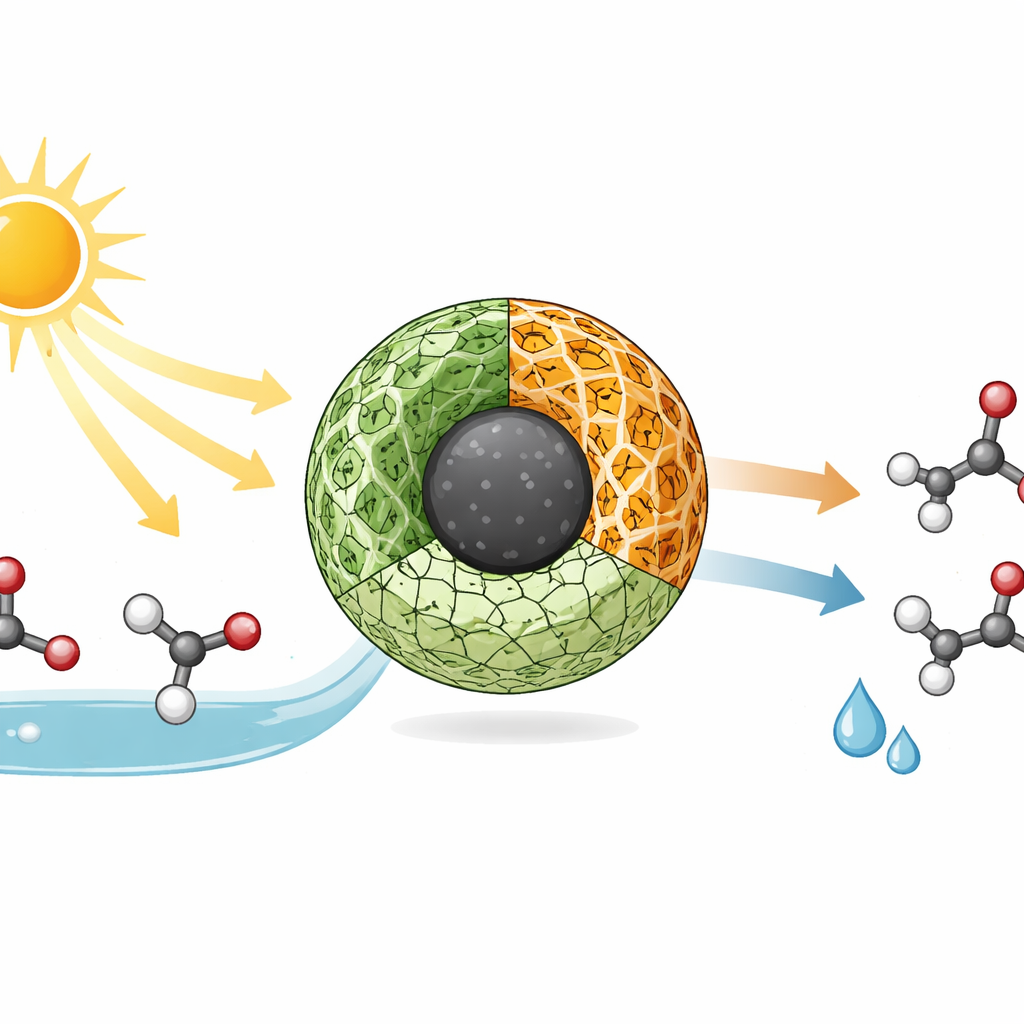
Catching Light and Moving Charges Efficiently
For a light-powered catalyst to work well, it must absorb visible light and keep the positive and negative charges generated by that light apart long enough to participate in chemical reactions. Detailed measurements of the new hybrid show that combining the three components broadens light absorption across most of the visible spectrum and narrows the effective energy gap between filled and empty electronic levels. Photoluminescence, impedance, and related tests reveal that the hybrid has a much lower rate of charge recombination and a smaller resistance to charge flow than any of its individual parts. In simple terms, when sunlight hits the material, the resulting charges travel along designed pathways inside the particle instead of quickly annihilating each other as heat.
A Gentle, Air-Breathing Reaction System
To test this catalyst, the researchers chose benzyl alcohol and a range of related alcohols that are common building blocks in fine chemicals. Using only a few milligrams of the solid, ethanol as a green solvent, air bubbles as the oxidant, and mild temperatures under simulated sunlight, they converted these alcohols selectively into their corresponding aldehydes or ketones in high yields. Control experiments showed that without light, without the catalyst, or under nitrogen instead of air, the reaction barely proceeds. Scavenger tests indicated that both positively charged "holes" in the catalyst and reactive oxygen species formed from air are key players in the oxidation step. Crucially, the magnetic iron oxide core allows the entire catalyst to be pulled out of the liquid with a simple magnet, washed, and reused at least seven times with almost no loss in activity or structural change. 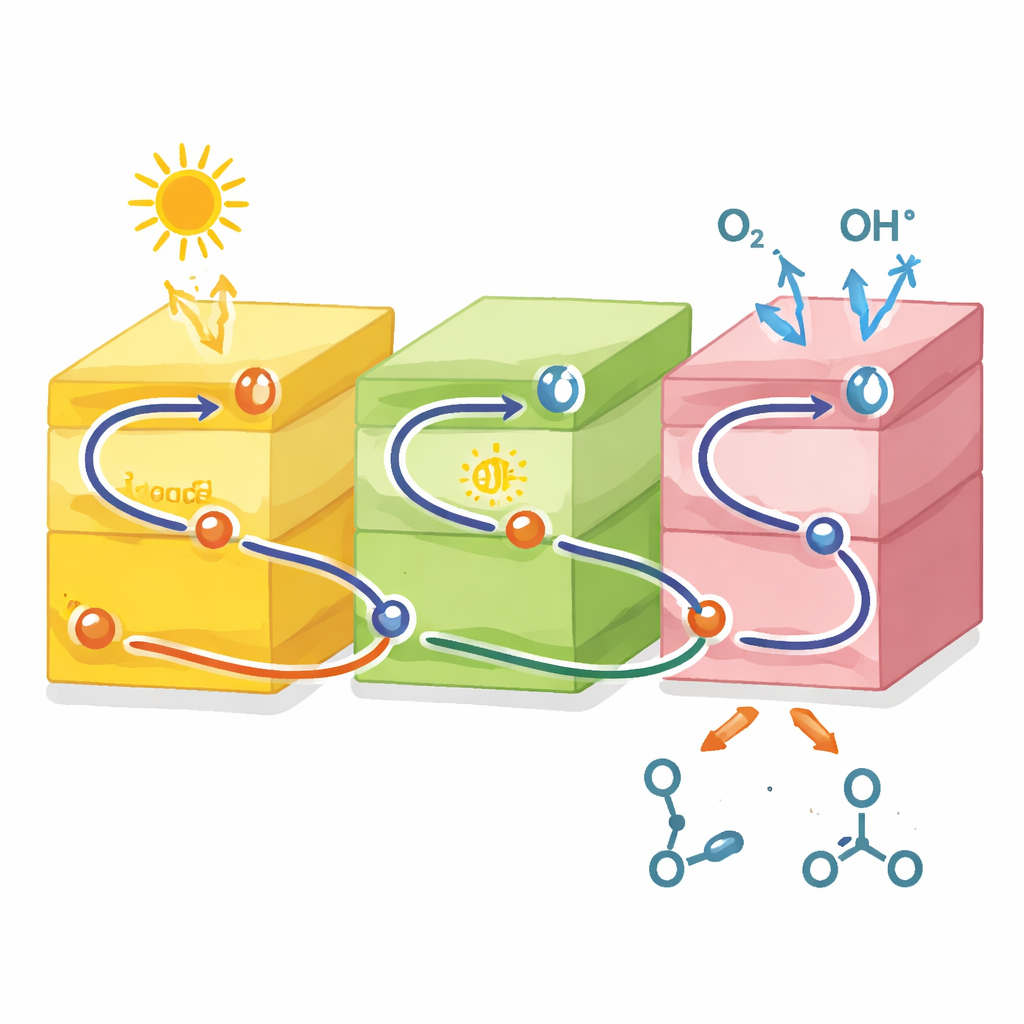
An S-Shaped Pathway Inside the Particle
The most intriguing finding is how the three components cooperate electronically. Based on electrochemical measurements and band-energy mapping, the authors rule out a simple, stepwise transfer of electrons between the materials. Instead, they propose a "dual S-scheme" pathway: under light, each component generates electrons and holes, but only the weaker charges recombine across interfaces, while the strongest oxidizing holes accumulate in the iron-based MOF and the most reducing electrons gather in the COF. This S-shaped route preserves the driving force needed to turn oxygen into reactive species on one side and alcohols into aldehydes on the other, while still minimizing wasteful recombination.
Cleaner Routes to Everyday Molecules
In practical terms, this work demonstrates a robust, magnetically recoverable catalyst that can use sunlight and air to perform an important industrial transformation under mild, environmentally friendly conditions. By carefully engineering how light-induced charges move through a three-part particle, the authors achieve both high selectivity and efficiency without resorting to toxic oxidants or high temperatures. For non-specialists, the key message is that smart materials design can make everyday chemical building blocks like aldehydes in ways that are kinder to people and the planet, pointing toward greener manufacturing processes in the future.
Citation: Sobhani, S., Bidokhti, H.K., Farrokhi, A. et al. γ-Fe2O3/MIL101(Fe)-NH2/COF-MT as a novel ternary photocatalyst for the selective oxidation of alcohols through a dual S-scheme pathway under sunlight irradiation. Sci Rep 16, 8138 (2026). https://doi.org/10.1038/s41598-026-39673-3
Keywords: photocatalysis, green chemistry, aldehyde synthesis, hybrid catalysts, solar-driven oxidation