Clear Sky Science · en
Dose- and time-dependent relative biological effect of proton in different intracerebral cells
Why this brain radiation study matters
As proton therapy centers appear in more hospitals, many patients are told that proton beams can hit tumors harder while sparing healthy brain tissue better than conventional X‑ray radiation (photons). But how much gentler are protons on the living brain, really, and does that advantage change over time? This study used a detailed animal model to track how different types of brain cells respond to proton versus photon radiation over weeks, offering clues that could influence how safely we treat brain tumors and protect thinking and memory.
Looking inside the brain’s cell community
The brain is not a uniform sponge of nerve tissue; it is a community of specialized cells that react to injury in distinct ways. The researchers focused on three key players in rabbits: neurons, which handle signals and underlie memory; oligodendrocytes, which insulate nerve fibers so signals travel quickly; and microglia, the brain’s resident immune cells. They targeted the whole brain with either proton or photon radiation at several dose levels roughly comparable to strong clinical treatments, then examined two critical regions for thinking and information relay—the hippocampus and thalamus—over the course of two months.
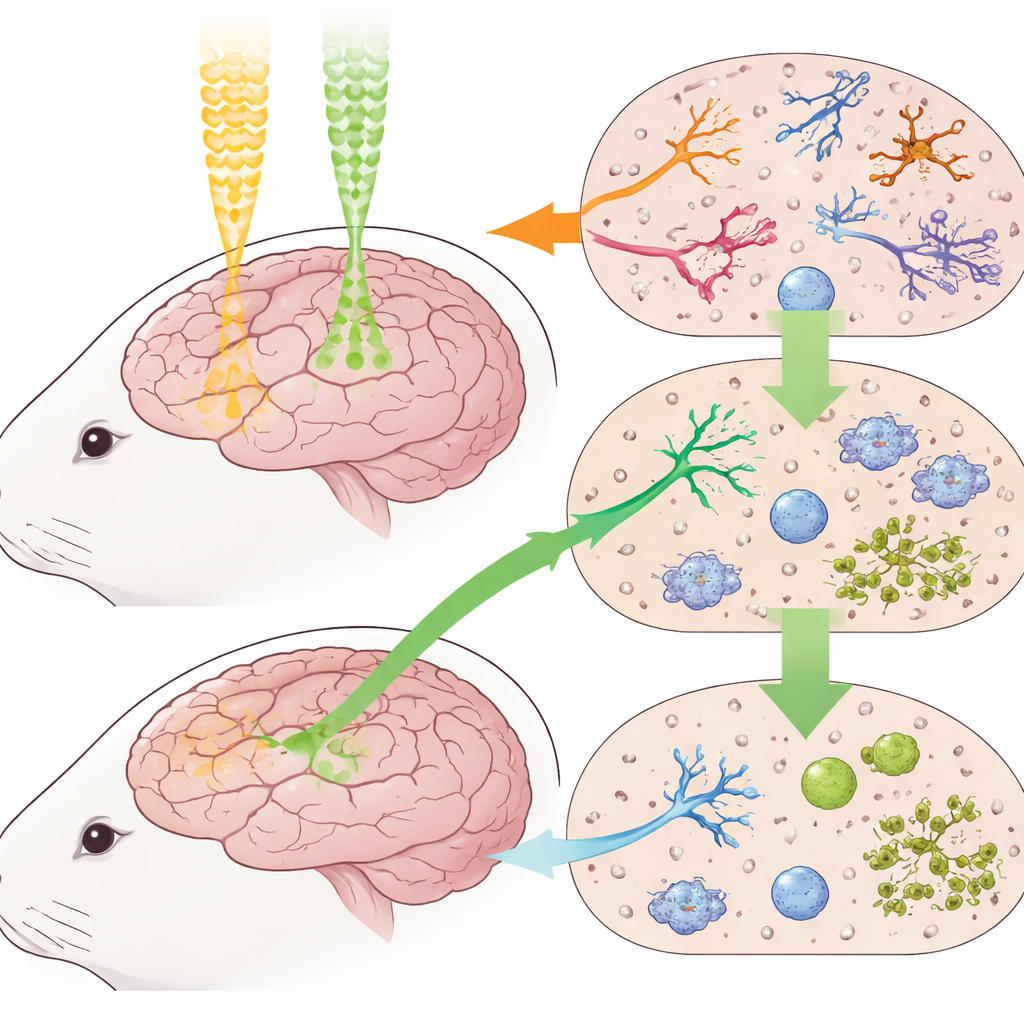
How the experiments were done
Groups of rabbits received single brain doses of 10, 20, 30, or 40 gray units of radiation, either as photons or as protons, while a control group received no radiation. Animals were then sacrificed at 2, 4, 6, or 8 weeks, and their brains were processed into thin slices. Standard tissue staining was used to count damaged versus healthy‑looking neurons, while special antibody stains highlighted nerve fibers, oligodendrocytes, and activated microglia. Using these counts, the team fit a widely used mathematical model of radiation response to calculate the so‑called relative biological effectiveness (RBE)—a measure of how potent protons are compared with photons—for each cell type, dose, and time point.
What happened to nerve cells and support cells
Both radiation types clearly injured neurons, and the damage accumulated over time. Yet, from 4 to 8 weeks after treatment, brains exposed to proton radiation consistently showed higher neuron survival and better‑preserved nerve fibers than those exposed to the same labeled doses of photons, especially at 10, 20, and 30 gray. Oligodendrocytes told a similar story: at modest doses and later time points, their numbers were often higher in proton‑treated brains than in photon‑treated ones, suggesting that the insulating white matter may weather proton exposure somewhat better. When these observations were translated into RBE values, the long‑term proton effect on neurons and oligodendrocytes generally fell below the commonly assumed value of 1.1, sometimes substantially so, implying that real brain tissue may tolerate higher physical proton doses than current planning rules presume.
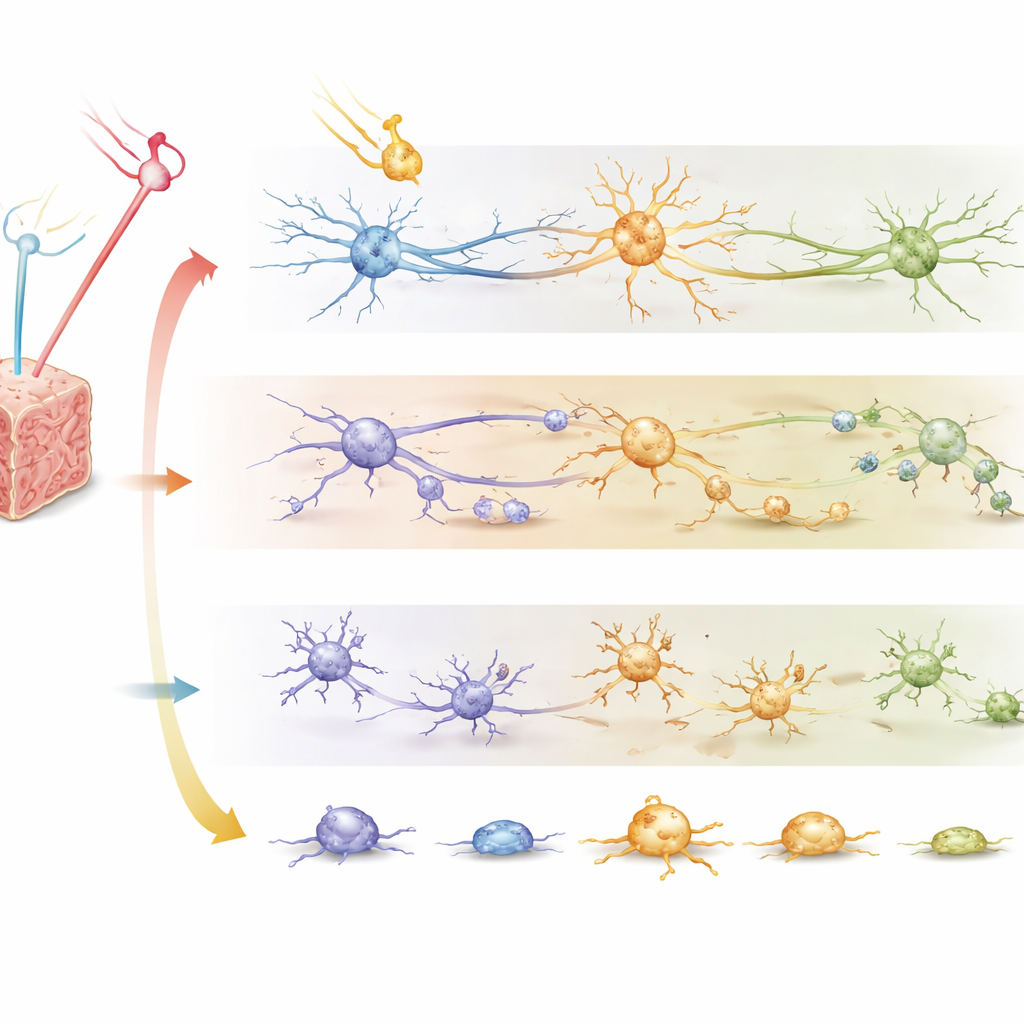
The brain’s immune response paints a different picture
Microglia behaved differently. These immune cells become “activated” when they detect injury, changing shape and releasing inflammatory molecules that can both help and harm. Across most doses and times, the level of microglial activation rose with dose and then slowly eased over weeks for both radiation types. However, at certain conditions—most notably four weeks after a mid‑range proton dose—protons triggered noticeably stronger microglial activation than photons. When the researchers calculated RBE for this immune response marker, many values exceeded 1.1, in contrast to the neuron and oligodendrocyte patterns. This suggests that while protons may spare nerve and support cells, they can provoke a more vigorous inflammatory reaction, a double‑edged sword that could influence both side effects and the success of combined treatments with immunotherapy.
What this means for future brain treatments
For patients and clinicians, the takeaway is that proton therapy’s biological impact in the brain is not a fixed number but a moving target that depends on cell type, dose, and time after treatment. In this rabbit model, neurons and their insulating partners ultimately fared better under proton radiation than under photons, supporting the idea that the brain may safely tolerate slightly higher or more precisely shaped proton doses than current conservative standards assume. At the same time, the heightened activation of microglia hints that protons may reshape the brain’s immune environment in complex ways, potentially opening doors for smarter combinations with immune‑based therapies. Together, these findings argue for more personalized radiation planning that looks beyond simple dose and into how different brain cells live, die, and repair themselves after proton versus photon therapy.
Citation: Wang, X., Guo, Y., Zhang, J. et al. Dose- and time-dependent relative biological effect of proton in different intracerebral cells. Sci Rep 16, 8984 (2026). https://doi.org/10.1038/s41598-026-39669-z
Keywords: proton therapy, brain radiation, neurons, microglia, radiation side effects