Clear Sky Science · en
Targeting of specific neuronal types in the non-human primate brain by using a murine CD25-specific recombinant immunotoxin
Why this brain study matters
Understanding how specific groups of brain cells control movement, emotion, and behavior is key to treating disorders such as Parkinson’s disease and autism. But the brain is a dense forest of intertwined cells, and most tools cut down many trees at once. This study presents a way to remove only a chosen type of neuron in a monkey’s brain, with minimal collateral damage, opening the door to much sharper experiments on how particular circuits shape behavior and disease.
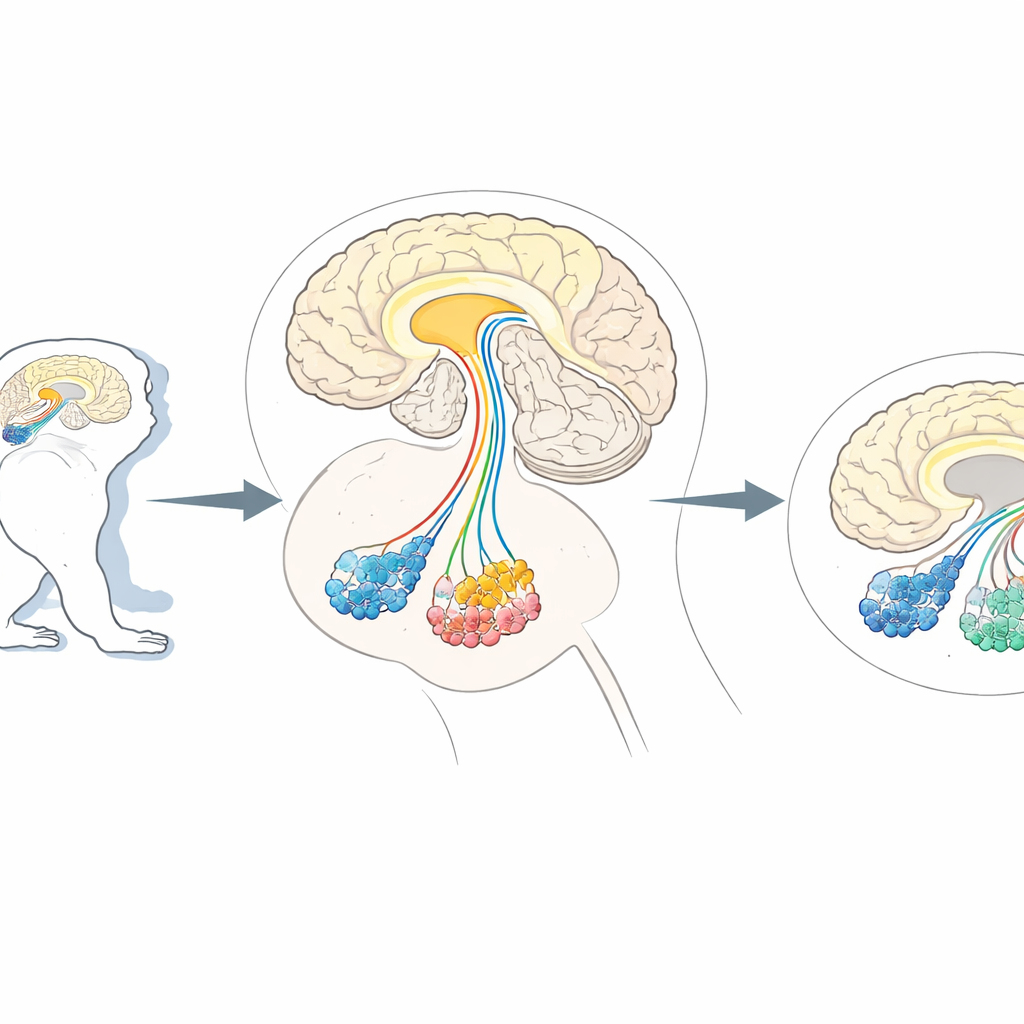
A need for precise brain "cell pruning"
Researchers often turn to non-human primates, like marmosets and macaques, because their brains are organized much like ours. These animals are invaluable models for conditions that affect higher brain functions, from Parkinsonian movement problems to autism-like changes in social behavior. Existing methods can silence or stimulate specific pathways, but actually removing just one type of neuron in these complex brains without harming others has remained difficult. Earlier work in mice used a trick: selected neurons were engineered to display a human cell-surface marker called CD25, which can be recognized by a designer toxin that kills only those marked cells. However, this same marker naturally occurs in primate immune and possibly brain cells, raising the risk that the toxin would attack the wrong targets in monkeys.
Designing a safer molecular scalpel
The authors set out to build a new molecular scalpel tailored for use in primates. Instead of targeting the human version of CD25, they focused on the mouse version, which is much less similar to the natural CD25 found in monkeys. They first immunized a rabbit with the mouse CD25 protein and used a chip-based method to fish out individual rabbit cells that produced antibodies binding strongly to mouse CD25 but not to the human form. From these, they identified a standout antibody, called RMAb-52, with very high binding strength. They then stitched together the key parts of this antibody with a toxic fragment from the bacterium Pseudomonas to create a single engineered "immunotoxin" protein, named anti-mCD25-PE38.
Putting the tool through its paces
In test-tube experiments, the new immunotoxin grabbed onto mouse CD25 much more tightly than human CD25, confirming its selectivity. When applied to cultured cells engineered to carry mouse CD25, the toxin sharply reduced their survival at very low doses, while leaving human CD25-bearing cells unharmed. Next, the team moved to living marmosets. Using a special viral vector that travels backward along nerve fibers, they delivered the mouse CD25 gene into dopamine-producing neurons that send signals from a deep brain region called the substantia nigra to the striatum, a pathway crucial for movement control. After giving the virus time to switch on the marker in these neurons, they injected the anti-mCD25-PE38 protein directly into the target midbrain area.
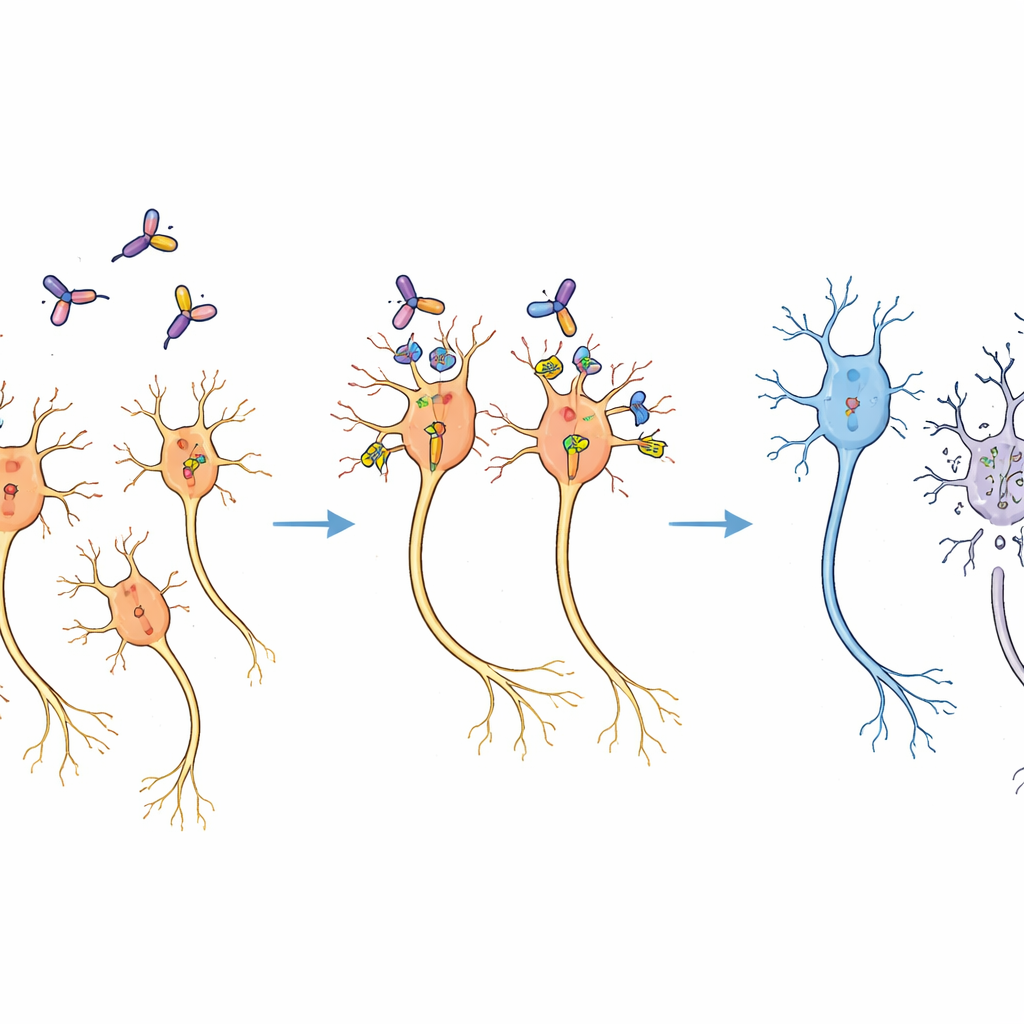
Selective loss without widespread damage
Two weeks after treatment, brain sections from the marmosets told a clear story. On the side that received the immunotoxin, the number of dopamine neurons was reduced to about two-thirds of that on the untreated side, as shown by staining for a dopamine-making enzyme. Yet the surrounding brain tissue looked normal under the microscope, and other brain regions that also received the viral vector showed no obvious loss of cells. Pilot experiments had already established a dose range that avoided non-specific tissue injury, and the chosen dose fell well within this safe window. The pattern of loss matched the expected spread of virus and toxin, indicating that neurons were eliminated specifically because they had been engineered to display mouse CD25, not because the toxin was harming cells indiscriminately.
What this means for future brain research
To a lay reader, the key message is that the researchers have built a highly selective delete button for chosen neuron populations in primate brains. By combining a retrograde viral delivery system with a mouse-specific immunotoxin, they can remove defined pathways—for example, dopamine circuits implicated in Parkinson’s disease—while sparing neighboring cells. This strategy avoids dangerous cross-reactions with the animal’s own CD25 and should be especially useful when that natural receptor is present or elevated in disease. In the long run, this approach will help scientists map how individual routes in the brain’s wiring contribute to movement, decision-making, and psychiatric symptoms, bringing us closer to targeted treatments that adjust faulty circuits instead of broadly affecting the whole brain.
Citation: Kobayashi, T., Kato, S., Kimura, S. et al. Targeting of specific neuronal types in the non-human primate brain by using a murine CD25-specific recombinant immunotoxin. Sci Rep 16, 8247 (2026). https://doi.org/10.1038/s41598-026-39662-6
Keywords: non-human primate neuroscience, immunotoxin targeting, dopamine neurons, marmoset brain circuits, Parkinson’s disease models