Clear Sky Science · en
Cold inducible RNA binding protein promotes fibroblast activation and its inhibition represents a potential therapeutic target in pulmonary fibrosis
Why lung scarring matters
Pulmonary fibrosis is a condition in which the lungs slowly turn from soft, stretchy tissue into stiff, scar‑like material. As the scarring spreads, breathing becomes difficult and oxygen levels fall, and many patients die within a few years of diagnosis. Current medicines can slow the damage but rarely change the long‑term outlook. This study explores a stress‑responsive protein called CIRBP and asks a simple question with big implications: does this protein help drive lung scarring, and if so, can blocking it protect the lungs?
A stress signal hiding in plain sight
CIRBP is normally produced inside many kinds of cells when they are exposed to stress, such as cold, low oxygen, or toxic injuries. Inside the cell it helps stabilize RNA, the messages that tell cells which proteins to make. But CIRBP can also leak or be secreted outside cells, where it behaves more like an alarm signal, stirring up inflammation. Earlier clinical work showed that people with idiopathic pulmonary fibrosis have more CIRBP in their scarred lung tissue and in their blood, and that higher levels tracked with worse outcomes. The new study set out to test whether CIRBP is simply a bystander that reflects tissue stress or an active player that pushes the disease forward.
Putting CIRBP to the test in diseased lungs
The researchers used a standard mouse model of lung fibrosis in which the chemotherapy drug bleomycin is delivered directly into the airways, triggering injury and scarring over several weeks. They compared normal mice with animals genetically engineered to lack CIRBP. After bleomycin exposure, normal mice showed a strong rise in CIRBP, especially in the most fibrotic regions of the lung. In contrast, mice without CIRBP survived longer, had less visible scarring on tissue sections, and showed lower levels of collagen and other fibrotic markers. These findings suggest that CIRBP is not only present but actively contributes to the build‑up of scar tissue.
How CIRBP pushes fibroblasts into overdrive
To zoom in on the mechanism, the team isolated primary fibroblasts, the connective‑tissue cells that lay down collagen and are central to fibrosis. When they added purified CIRBP to these cells in culture, the fibroblasts secreted more collagen, multiplied faster, and moved more readily—behaviors that together favor expansion of scar tissue. Interestingly, CIRBP did not clearly boost classic markers of fully transformed "myofibroblasts" inside the cells, implying that it mainly tunes their activity rather than completely changing their identity. Gene‑expression analyses showed that CIRBP‑treated fibroblasts ramped up many immune and inflammatory genes, and protein measurements highlighted one molecule in particular: the cytokine IL‑6, which rose sharply in response to CIRBP.
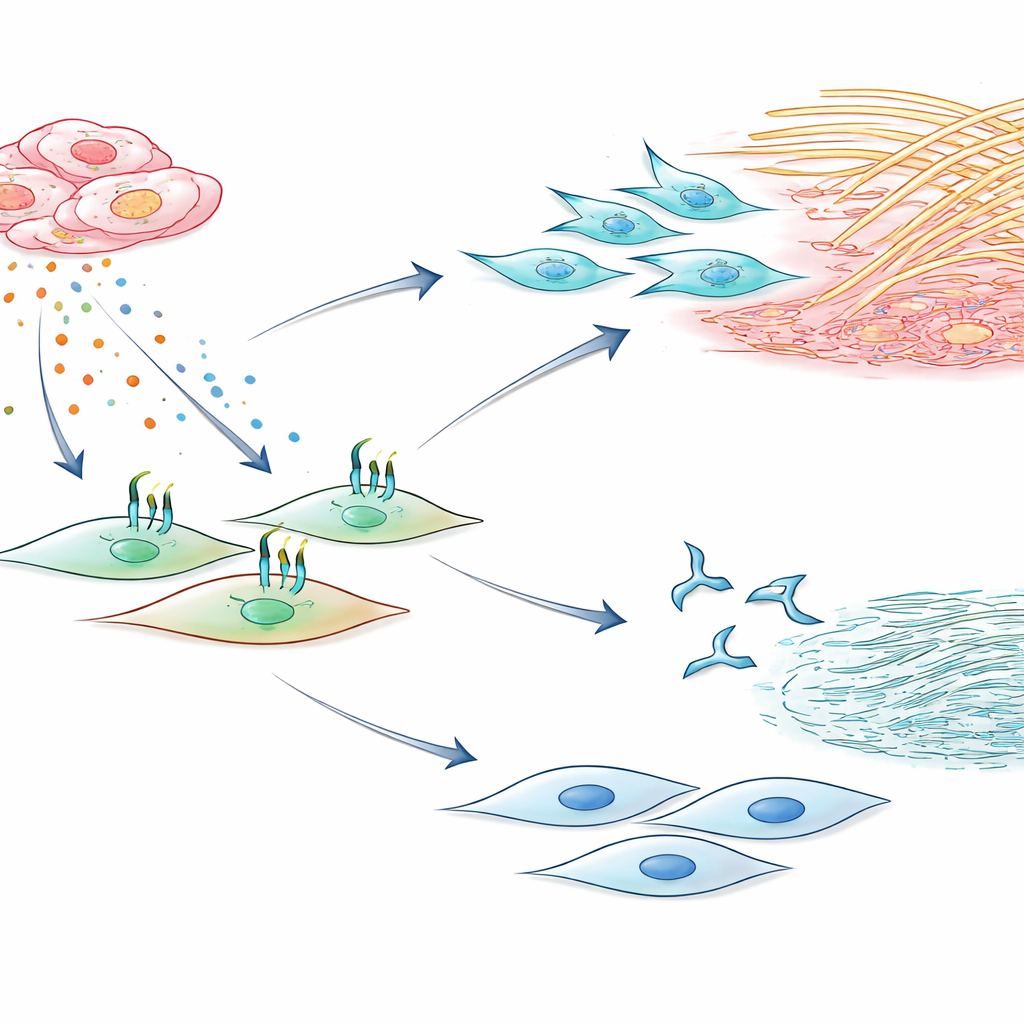
A signaling chain that can be interrupted
The study then mapped the signaling chain that links CIRBP to fibroblast activation. CIRBP outside the cell appears to engage two innate‑immune receptors on fibroblasts, known as TLR2 and TLR4. When these receptors sensed CIRBP, the cells secreted more IL‑6, which in turn acted back on the same fibroblasts in an autocrine loop to further enhance collagen release and cell growth. Blocking IL‑6 with neutralizing antibodies cut down collagen secretion and proliferation, confirming its central role in this loop. Likewise, drugs that inhibit TLR2 or TLR4 weakened CIRBP’s effects on IL‑6, collagen, and cell division. Most strikingly, a short peptide called C23, derived from CIRBP itself and designed to compete for binding to these receptors, strongly reduced IL‑6 production and the downstream fibrotic behaviors in cultured fibroblasts.
Peptide treatment that softens the blow
Finally, the scientists tested whether targeting CIRBP could help animals with established lung injury. In the bleomycin model, they began giving mice C23 injections a week after the initial insult, at a time when damage is already underway. Compared with placebo, C23‑treated mice survived better, had milder scarring on histology, and showed lower lung collagen content and reduced levels of fibrosis‑related proteins. These results indicate that blocking CIRBP signaling can meaningfully blunt the progression of lung fibrosis, even when treatment starts after the injury has occurred.
What this means for future lung therapies
For non‑specialists, the key message is that the study identifies CIRBP as a stress‑induced alarm protein that helps drive lung scarring by switching fibroblasts into a more aggressive, collagen‑secreting state through a TLR2/TLR4–IL‑6 signaling loop. Interrupting this chain, particularly with the C23 peptide, eased fibrosis and improved survival in mice. While much work remains before such an approach could reach patients, CIRBP now stands out as a promising upstream target: dialing down its signal might slow or prevent deadly lung scarring while leaving much of the immune system’s normal defenses intact.
Citation: Mochizuka, Y., Hozumi, H., Watanabe, H. et al. Cold inducible RNA binding protein promotes fibroblast activation and its inhibition represents a potential therapeutic target in pulmonary fibrosis. Sci Rep 16, 8324 (2026). https://doi.org/10.1038/s41598-026-39649-3
Keywords: pulmonary fibrosis, fibroblasts, CIRBP, IL-6 signaling, lung scarring