Clear Sky Science · en
Hypoxia-inducible factor 1α exerts dual roles in bladder cancer progression through TIMP3-mediated regulation of angiogenesis and invasion
Why oxygen-starved tumors matter
Cancer cells often grow so fast that they outrun their blood supply, leaving parts of a tumor starved of oxygen. This low-oxygen, or hypoxic, environment forces cells to switch on survival programs that can make cancers harder to treat. In bladder cancer—a common tumor of the urinary system—scientists have long suspected that one such oxygen-sensing program, controlled by a protein called HIF-1α, mainly helps tumors grow and spread. This study takes a closer look and finds a surprising twist: under certain conditions, turning down HIF-1α can actually make bladder cancer cells more aggressive, and a second protein, TIMP3, sits at the heart of this paradox.
A closer look at bladder cancer cells
The researchers worked with two well-established human bladder cancer cell lines and exposed them either to normal oxygen levels or to hypoxia, mimicking the oxygen-poor pockets inside real tumors. They then used genetic tools to dial down HIF-1α in these cells and measured how this change affected basic cancer behaviors: how fast the cells multiplied, how easily they died, and how well they could move, invade through a barrier, and encourage new blood vessels to form. In parallel, they examined bladder tumor samples from patients to compare levels of HIF-1α and TIMP3 in cancerous tissue versus nearby normal bladder tissue.
A surprising role reversal under low oxygen
Patient samples confirmed that HIF-1α is indeed higher in bladder tumors than in normal tissue, consistent with its reputation as a tumor-promoting factor. Yet, in the cell experiments under hypoxia, reducing HIF-1α had the opposite effect of what many would expect. When HIF-1α was knocked down and oxygen was scarce, cancer cells grew faster over time, were less likely to undergo programmed cell death, and became better at migrating and invading through model barriers. They also released signals that encouraged blood vessel cells to form denser, more extensive tube-like networks, a hallmark of increased tumor blood supply. In short, with less HIF-1α in a low-oxygen setting, bladder cancer cells behaved in a more malignant way.
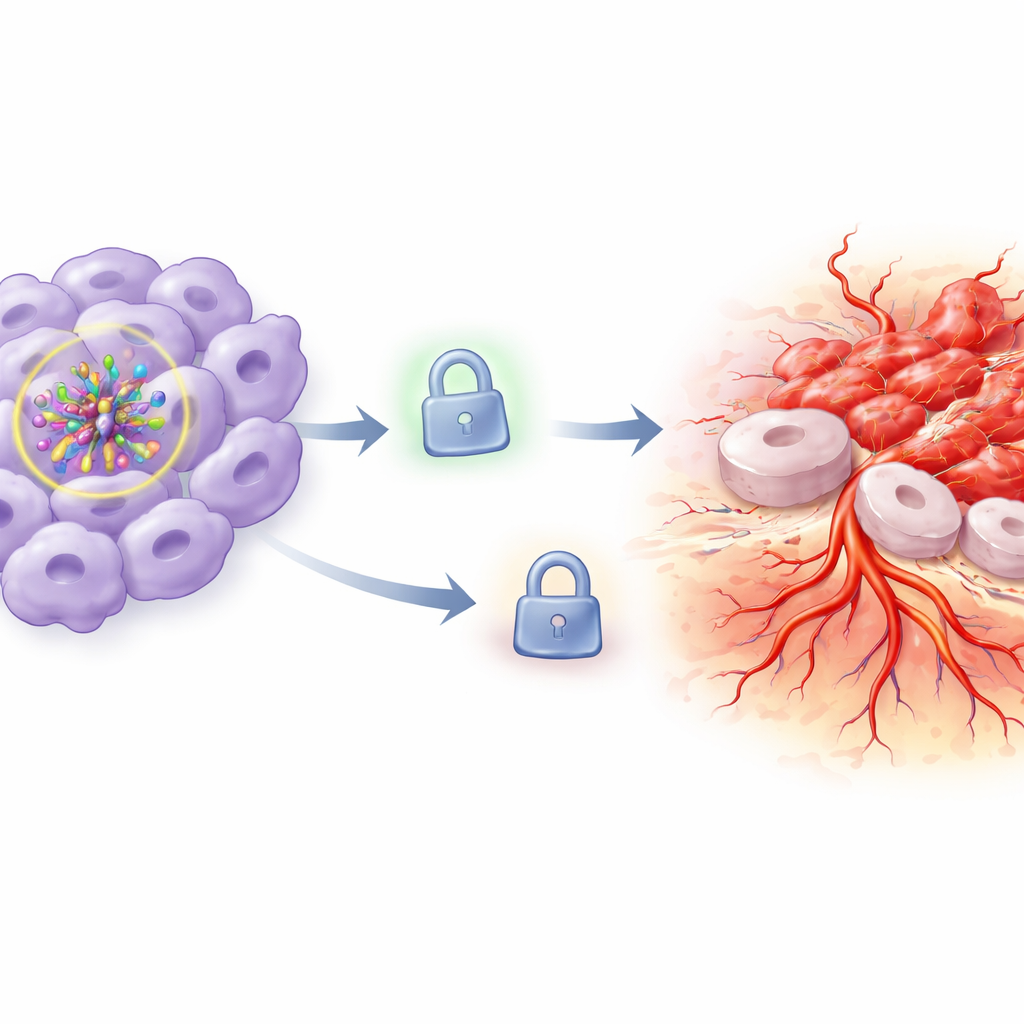
The missing brake on tumor spread
To understand why this happened, the team focused on TIMP3, a protein that normally acts as a brake on tissue breakdown and new vessel growth by blocking enzymes that chew through the surrounding matrix. In HIF-1α–reduced cells kept under normal oxygen, TIMP3 levels remained relatively high. But when oxygen levels dropped, TIMP3 protein fell sharply. This loss of TIMP3 fit well with the observed increase in invasion and vessel formation: with the brake weakened, cancer cells could more easily remodel their surroundings and expand. The researchers then flipped the switch by forcing cells to make extra TIMP3 while HIF-1α was still knocked down under hypoxia. Restoring TIMP3 partially or almost completely reversed the aggressive behavior: cells grew less, died more readily, moved and invaded less, and triggered fewer and shorter vessel-like tubes.
What this means for future treatments
These findings suggest that HIF-1α has a dual personality in bladder cancer. On one hand, its overall levels are higher in tumors and it helps drive known cancer-friendly programs. On the other, under severe oxygen shortage it appears to help maintain the protective presence of TIMP3. Removing HIF-1α in that context can unintentionally lower TIMP3 and unleash more invasive, blood-vessel–promoting behavior. For therapy, this means that simply blocking HIF-1α might backfire unless doctors also find ways to keep TIMP3, or similar brakes, in place. The work highlights the importance of understanding not just single targets, but the broader network of checks and balances that shape how a tumor responds to its harsh, oxygen-poor environment.
Citation: Wang, X., Guo, J., Zhang, R. et al. Hypoxia-inducible factor 1α exerts dual roles in bladder cancer progression through TIMP3-mediated regulation of angiogenesis and invasion. Sci Rep 16, 8425 (2026). https://doi.org/10.1038/s41598-026-39635-9
Keywords: bladder cancer, tumor hypoxia, HIF-1α, TIMP3, angiogenesis