Clear Sky Science · en
Molecular docking and dynamics reveal novel CDK6 inhibitors for targeted glioblastoma therapy
Why this brain cancer study matters
Glioblastoma is one of the most aggressive forms of brain cancer, and for many patients today’s treatments still do not work well enough or long enough. This study explores a new way to attack glioblastoma by homing in on a single overactive control switch inside tumor cells, a protein called CDK6. Using advanced computer simulations instead of lab animals or test tubes, the researchers sifted through hundreds of drug‑like molecules and identified one especially promising candidate, called compound 21, that could one day help slow or stop this deadly disease.
A deadly tumor that dodges current treatments
Glioblastoma grows quickly, invades nearby brain tissue, and almost always comes back after surgery, radiation, and chemotherapy. Standard drugs such as temozolomide can shrink tumors at first, but many cancer cells adapt and become resistant, leading to relapse. One reason is that their internal cell‑division machinery is stuck in “go” mode. Proteins called CDKs act as timing gears that tell cells when to copy their DNA and divide. In glioblastoma, the CDK4/6 pathway is frequently overactive, pushing cells to multiply even in the face of radiation and drugs. That makes CDK6 an appealing target: if scientists can switch it off selectively in tumor cells, they might slow growth and make existing treatments work better.
Hunting for a better CDK6 blocker on a computer
Instead of starting in the lab, the team ran a large‑scale virtual search. They began with a known CDK6‑binding molecule and asked a screening program to find hundreds of similar structures from a drug database. From 400 initial candidates, 109 were chosen for detailed computer docking against the 3‑D structure of human CDK6. Docking is like trying many keys in a lock: the software predicts which molecules slide most snugly into the protein’s active pocket and how strongly they might bind. Forty‑eight candidates looked even better than the original reference molecule, but one, compound 21, stood out with the strongest predicted binding and especially favorable contacts at specific spots on CDK6 that are known to control selectivity.
Checking if the “virtual drug” behaves like a real medicine
Strong binding is only part of the story; a useful medicine must also reach the brain, dissolve well, and avoid harming healthy tissue. The researchers used additional programs to estimate how each top compound would be absorbed, carried through the body, and cleared. Compound 21 passed standard “drug‑likeness” rules, showed good predicted solubility, and, importantly for brain tumors, was forecast to cross the blood–brain barrier while remaining reasonably safe. A separate toxicity tool suggested that, aside from potential issues with very high oral doses, compound 21 was less likely than many others to irritate skin or eyes or cause severe acute damage. Another set of calculations examined how electrons are arranged within the molecule, giving clues to its chemical stability and tendency to form unwanted side reactions; these tests again favored compound 21 over the original reference drug.
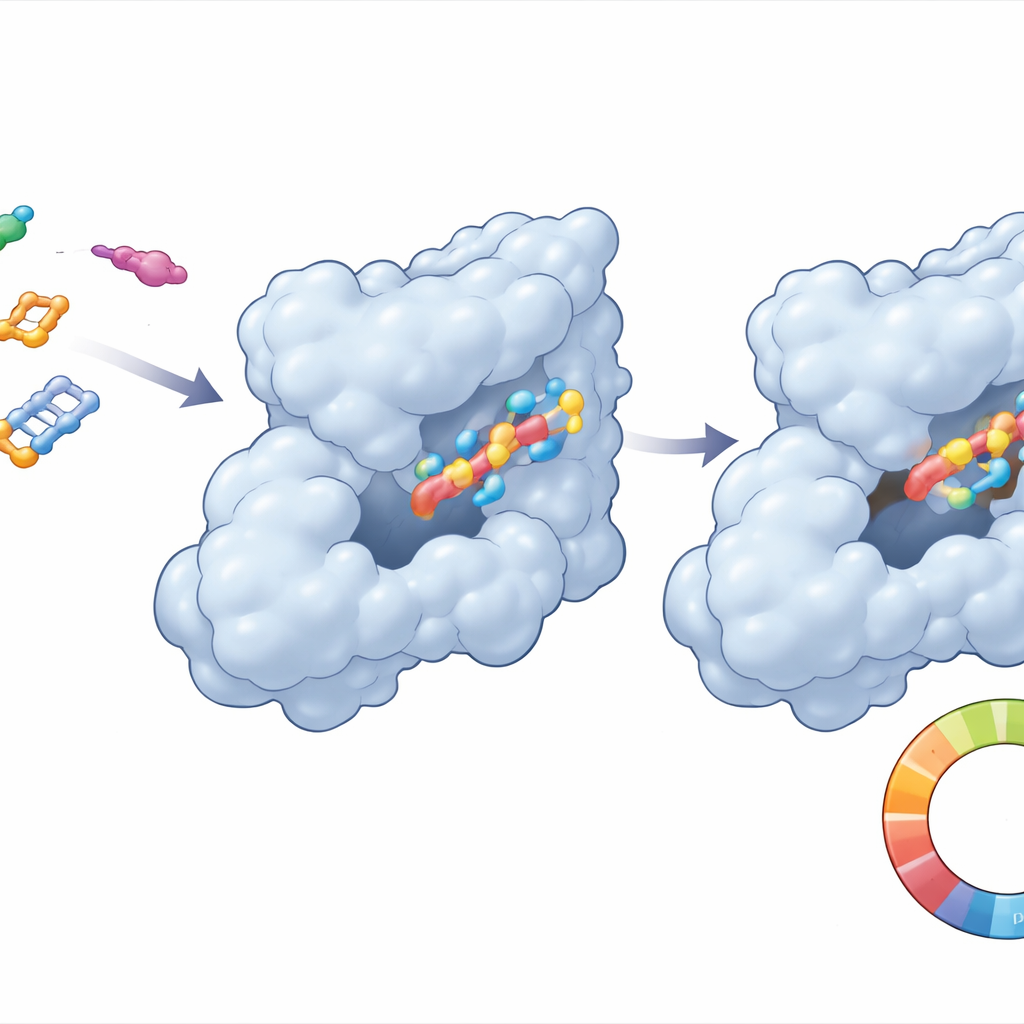
Watching the drug and its target move together
Because proteins and drug molecules are not rigid, the team ran long computer “movies” lasting 250 billionths of a second to see how CDK6 and each compound actually behave in a watery, body‑like environment. For compound 21, the complex settled quickly into a stable arrangement and wobbled only modestly, a good sign that the molecule would stay lodged in the CDK6 pocket. Key contact points between the drug and protein were maintained, while the overall shape of CDK6 remained compact. In contrast, the reference compound showed larger fluctuations and weaker, less consistent contacts. Additional energy calculations supported the idea that CDK6 prefers to bind compound 21 more tightly, mainly through snug hydrophobic (oil‑like) interactions and well‑positioned hydrogen bonds.
What this could mean for future brain cancer care
Taken together, these computer‑based tests build a strong case that compound 21 is a high‑quality starting point for a new glioblastoma drug aimed at CDK6. It appears more selective, more stable, and better suited to reach the brain than the reference molecule, while avoiding many common toxicity pitfalls. However, the work is still at a predictive stage: no cells or animals were treated in this study. The next steps will be to synthesize compound 21, test whether it really slows glioblastoma cell growth in the lab, and see how it behaves in animal models. If those studies confirm the simulations, this molecule—or improved versions of it—could eventually contribute to more precise, targeted therapies that give glioblastoma patients better odds and longer‑lasting control of their disease.
Citation: Khan, M.U., Munir, M., Manzoor, H. et al. Molecular docking and dynamics reveal novel CDK6 inhibitors for targeted glioblastoma therapy. Sci Rep 16, 9000 (2026). https://doi.org/10.1038/s41598-026-39629-7
Keywords: glioblastoma, CDK6 inhibitors, targeted therapy, virtual drug screening, brain cancer