Clear Sky Science · en
Correlation between carbon percentage and nanocomposite performance in commodity and engineering thermoplastics (ABS, HIPS, PP, and PC)
Why tiny sheets of carbon matter for everyday plastics
From car bumpers and phone cases to clear safety glasses, many familiar products are made from a handful of workhorse plastics. A new study asks a deceptively simple question: if you sprinkle in a tiny amount of graphene—ultra-thin sheets of carbon—do all of these plastics get stronger in the same way? By comparing four common plastics under identical conditions, the researchers show that the answer depends not just on how much carbon they contain, but on how that carbon is arranged in their molecular makeup.
The four plastics that shape modern products
The team focused on four widely used thermoplastics: ABS, HIPS, PC, and PP. ABS, used in car interiors and 3D-printed parts, is tough and easily molded. HIPS, common in packaging and appliance housings, is an impact-modified form of polystyrene. PC (polycarbonate) is famous for its combination of transparency and extreme toughness, making it a go-to for safety gear and lenses. PP (polypropylene) is a lightweight, chemically resistant plastic used in everything from food containers to automotive components. These materials differ not only in strength and stiffness but also in how their molecules pack together—some are mostly disordered, others form crystalline regions—and in how much carbon they contain relative to other atoms like oxygen and nitrogen.
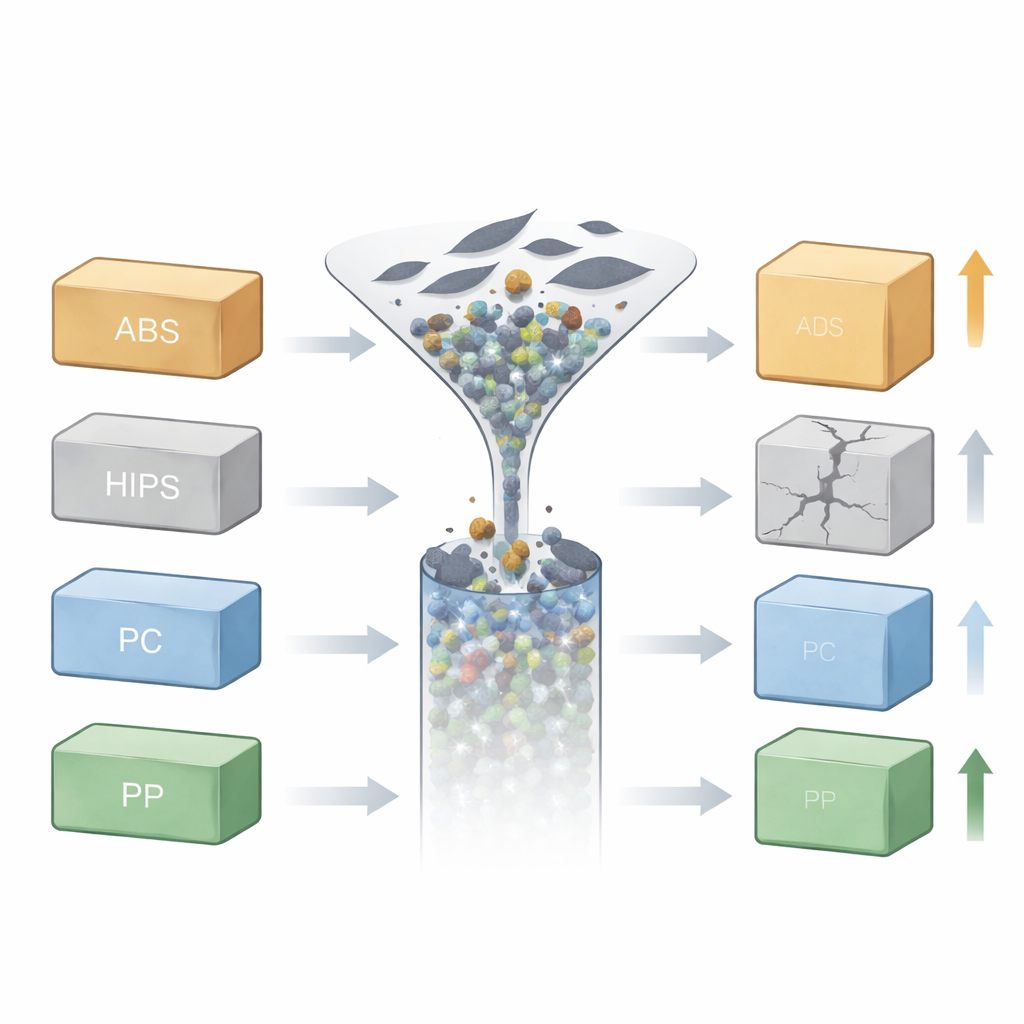
Adding graphene the same way, across the board
To make a fair comparison, the researchers mixed the same small amount of graphene nanoplatelets (0.7% by weight) into each plastic using melt processing, then formed standard test bars by injection molding. They did not tune the recipe for each polymer; instead, they deliberately held the graphene level and processing route constant so that any differences in performance would mainly reflect the underlying plastic. They then examined the samples using scanning electron microscopy to see how well graphene dispersed, X-ray diffraction to probe changes in molecular ordering, and mechanical tests to measure hardness and impact strength. Statistical models, built with a factorial experimental design, linked these measurements to each polymer’s overall carbon percentage and its interaction with graphene.
What happens inside the plastic
Microscope images revealed that how graphene spreads through the plastic is crucial. In ABS and PP, fracture surfaces showed fibrous, stretched regions and only modest graphene clumping, signs of ductile failure and good stress sharing between the filler and the polymer. In PP, X-ray patterns indicated that graphene acted as a nucleating agent, sharpening crystalline peaks and implying more ordered regions that help stiffen the material. PC remained largely amorphous, with smooth fracture features and limited but acceptable graphene dispersion; its already high toughness left little room for further improvement. HIPS told a different story: bright, clustered regions of graphene and a granular, brittle fracture texture pointed to poor mixing. Instead of helping carry load, the graphene clumps acted as weak spots where cracks could easily start and spread.
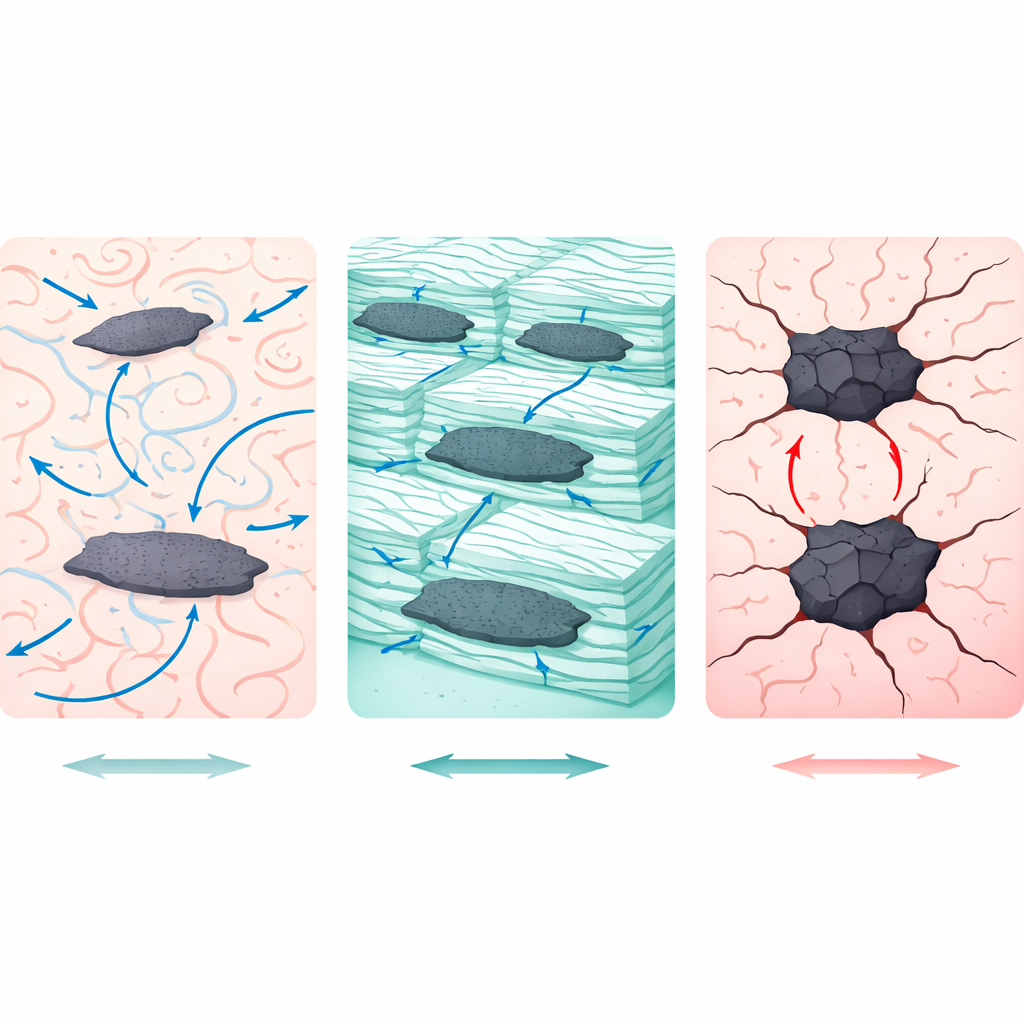
How strength and toughness actually changed
These internal differences showed up clearly in the mechanical tests. ABS saw the largest hardness gain, rising by nearly 40% with graphene, alongside a modest increase in impact strength. PP’s hardness and impact resistance improved slightly, consistent with better crystallinity but limited bonding to the non‑polar polymer chains. PC began with by far the highest impact energy absorption of all four plastics—around an order of magnitude greater—and graphene barely changed that value, suggesting a “ceiling” effect where the material is already so tough that a small filler addition makes little difference. In HIPS, hardness and impact strength both declined slightly after graphene addition, underscoring that poor dispersion can outweigh the inherent strength of the nanofiller. Statistical analysis confirmed that the base polymer’s carbon-related chemistry explained most of the variation, with graphene content and its interaction with that chemistry providing smaller but significant contributions.
What this means for choosing better materials
For non-specialists, the key message is that adding a high-tech ingredient like graphene is not a universal shortcut to stronger plastics. The same tiny carbon sheets can toughen one plastic, barely change another, and even weaken a third, depending on how well they “get along” with the host material at the molecular level. In this study, ABS and PP gained useful hardness and some impact benefits, PC was already so tough that graphene had little effect, and HIPS suffered from graphene clumping and performance loss. Rather than treating graphene loading alone as the design knob, the authors argue that engineers should consider the plastic’s carbon-based chemistry, polarity, and internal structure when selecting matrices for graphene nanocomposites, and use compatibilizers or surface treatments where needed to unlock graphene’s full potential.
Citation: Essam, M.A., Nassar, A., Nassar, E. et al. Correlation between carbon percentage and nanocomposite performance in commodity and engineering thermoplastics (ABS, HIPS, PP, and PC). Sci Rep 16, 8492 (2026). https://doi.org/10.1038/s41598-026-39627-9
Keywords: graphene nanocomposites, engineering thermoplastics, polymer reinforcement, mechanical properties, material selection