Clear Sky Science · en
A zebrafish multimodal toolbox to study the blood-brain barrier in health and disease
Why leaky brain vessels matter
When blood vessels in the brain or eye start to leak, fluid and proteins seep into the surrounding nerve tissue, causing swelling that can blur vision or impair thinking. This kind of "waterlogging"—known as vasogenic edema—plays a role in diabetic vision loss, stroke, brain trauma, and some forms of dementia. Yet scientists still lack simple, affordable ways to watch this protective barrier fail in real time. This study introduces a versatile zebrafish model that lets researchers see, measure, and probe how high blood sugar weakens the brain’s vessel barrier, potentially speeding up the search for new treatments.
A tiny fish with a clear advantage
Zebrafish larvae offer a unique window into the living brain. Unlike mammals, their bodies are transparent in early life, and their brain blood vessels can be made to glow under the microscope. The authors took advantage of this by focusing on the blood–brain barrier, the tight seal between blood and brain tissue that resembles the inner blood–retinal barrier protecting our eyes. Because the same cell types and many of the same protective proteins are shared between brain and retina, the team used the brain barrier in zebrafish as a stand-in to study diseases like diabetic macular edema and other neurovascular disorders, but in a faster, more ethical, and less expensive system than traditional rodent models.
Raising blood sugar in baby zebrafish
To mimic the high blood sugar seen in diabetes, the researchers raised zebrafish larvae in water containing extra glucose between three and five days after fertilization—a critical window when the brain barrier is still maturing. They confirmed that this external sugar bath drove up internal glucose levels several-fold without killing the larvae or altering their overall shape. With the fish still alive and swimming, they injected fluorescent dyes of different sizes into the circulation and used high-resolution confocal microscopes to track how much dye seeped from brain blood vessels into the surrounding tissue. After two days of high-glucose exposure, both small and large dye molecules leaked more into the brain, especially at the higher sugar dose, indicating that the barrier had become more permeable.
What happens to the vessel wall
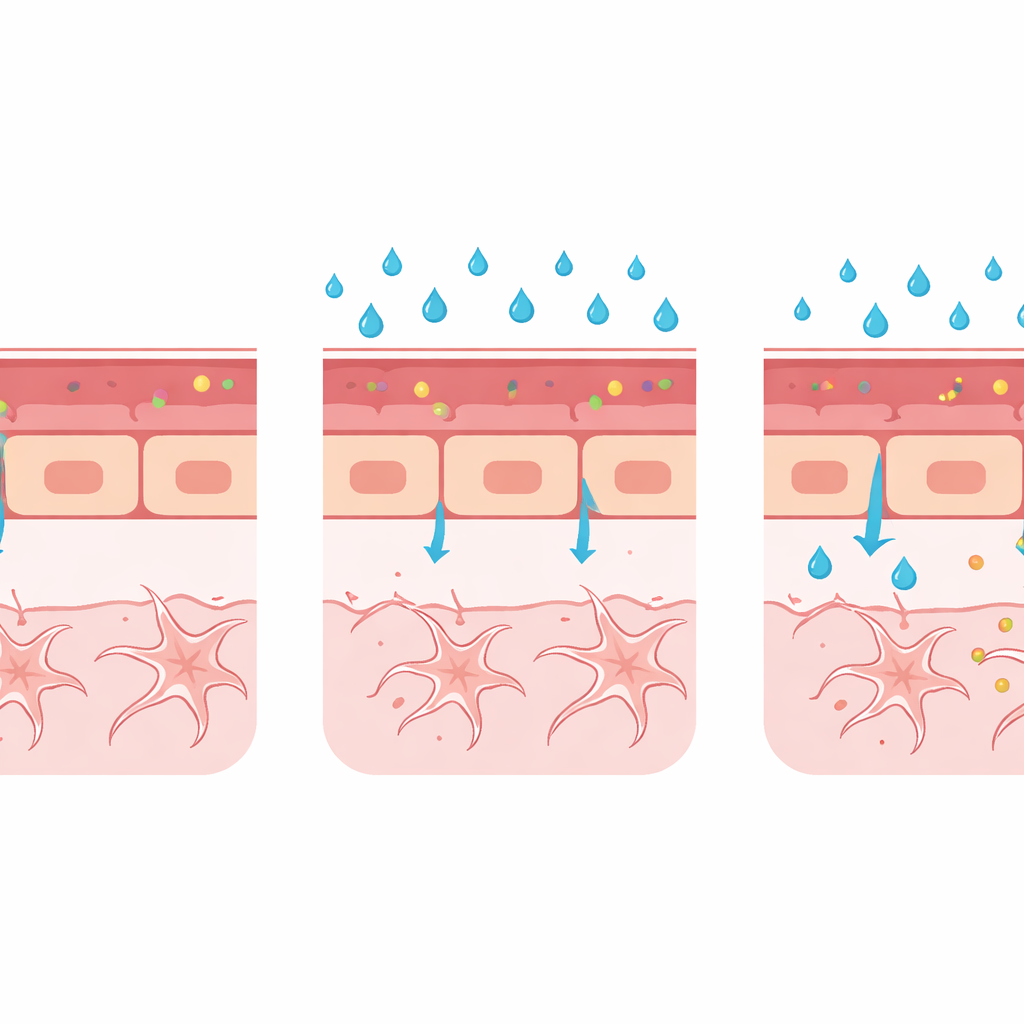
Beyond simply measuring leakage, the team built a multimodal "toolbox" to examine what was going wrong in the vessel wall itself. They measured the width of a key brain artery and found that high glucose caused the vessel to dilate—an early change also seen in people with diabetic eye disease. Using zebrafish engineered to light up specific proteins, they showed that high glucose reduced levels of claudin-5, a major component of the tight seals between neighboring vessel cells, and increased levels of PLVAP, a protein associated with immature, leak-prone vessels that shuttle material across the cell layer. Electron microscopy, which can reveal structures at the nanometer scale, confirmed subtle widening at junctions between cells, although the tiny transport pockets called caveolae were too sparse to quantify reliably in this first pass.
A flexible platform for future disease research
Taken together, these changes—more dye leakage, wider vessels, weaker cell junctions, and increased PLVAP—paint a consistent picture: high blood sugar can delay or derail proper barrier formation in the developing brain. The model does not yet capture the breakdown of a fully mature barrier, as occurs in chronic human disease, but it excels at showing how harmful conditions during development can keep the barrier from ever sealing properly. Because the techniques are compatible with a range of fluorescent reporter lines and advanced imaging, the same platform can be expanded to study other triggers of barrier failure, such as inflammatory molecules, altered support cells like pericytes and glia, or disruptions in key signaling pathways.
What this means for patients
For non-specialists, the core message is that this work delivers a practical, living testbed for watching the brain’s protective wall fail under stress, using tiny transparent fish instead of larger mammals. By following how high blood sugar reshapes vessel structure and function in real time, researchers can more quickly pinpoint which molecules and cell types to target to keep the barrier intact. Ultimately, such insights could guide the development of new drugs or treatment strategies to prevent or reduce fluid leakage in the eyes and brain—helping to preserve sight and protect cognitive function in people with diabetes and other neurovascular diseases.
Citation: Bakker-van Bugnum, N., Snijders, E.E., Hogendorp, E.F. et al. A zebrafish multimodal toolbox to study the blood-brain barrier in health and disease. Sci Rep 16, 9422 (2026). https://doi.org/10.1038/s41598-026-39616-y
Keywords: blood-brain barrier, zebrafish model, diabetic complications, vascular leakage, neurovascular disease