Clear Sky Science · en
Comparative analysis of natural and synthetic surfactant adsorption by quartz minerals: an experimental study
Why this matters for everyday energy
Much of the world’s oil is still locked underground, even after modern drilling and pumping. One promising way to nudge more of that oil out is to use soap-like molecules called surfactants that help oil and water mix. But if these surfactants stick too strongly to the rock instead of going to the oil–water boundary where they are needed, the process becomes wasteful and expensive. This study compares how a common synthetic surfactant and a plant-based alternative behave on quartz-rich sandstone, the rock that hosts many oil reservoirs, to see whether greener options can realistically compete.
Soap-like helpers in oilfields
Surfactants act a bit like dish soap in a greasy pan: they lower the tension between oil and water so trapped droplets can move. In enhanced oil recovery, water mixed with surfactant is pushed through the rock to sweep out more oil. However, mineral surfaces in the reservoir can “steal” surfactant molecules by adsorbing them, lowering the amount left in the flowing water. The authors focused on quartz, the main ingredient of sandstone, and studied two surfactants. One is sodium dodecyl sulfate (SDS), a widely used synthetic cleaning agent. The other is an extract from the leaves of the Ziziphus spina-christi tree (ZSC), rich in natural soap-like compounds called saponins. ZSC is attractive because it is inexpensive, locally available in many regions, and friendlier to the environment.
How the rock and surfactants were tested
The team first prepared powdered quartz from sandstone, carefully washing, sieving, and drying it to remove clays and other minerals so that only quartz behavior was measured. They characterized the particles’ surface area and pores and determined that the quartz surface carries a negative electrical charge in water. Solutions of SDS and ZSC at different strengths were then mixed with the quartz. After controlled mixing and resting periods, the liquid was separated and analyzed. Electrical conductivity and ultraviolet–visible light measurements revealed how much surfactant remained in solution, and thus how much had stuck to the rock. The researchers also used infrared spectroscopy to see which chemical groups were present on the quartz surface before and after contact with the surfactants, confirming that surfactant molecules really were attaching.
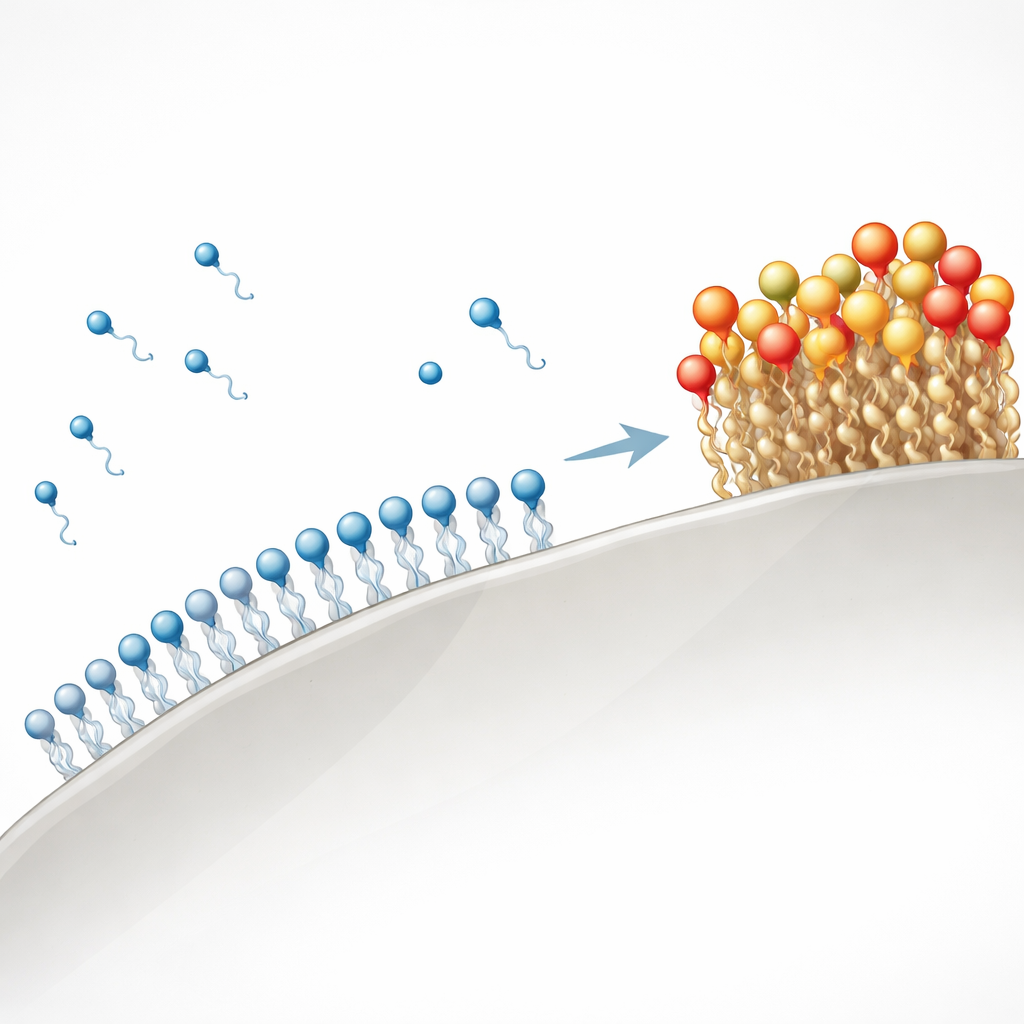
How much sticks and why
The measurements show a clear contrast between the two surfactants. Under the same conditions, SDS reached a maximum adsorption of about 3 milligrams per gram of quartz, while ZSC reached about 25 milligrams per gram—roughly eight times higher. In both cases, adsorption rose with concentration up to a characteristic point where surfactant molecules begin forming tiny clusters in water (the critical micelle concentration) and then leveled off. The quartz surface is negatively charged, and SDS is also negatively charged, so its adsorption is limited by electrostatic repulsion and relies mainly on weaker forces such as van der Waals interactions and the tendency of its oily tails to associate with the surface. ZSC, by contrast, is made of larger, more complex molecules with many oxygen- and nitrogen-containing groups that can form multiple hydrogen bonds with the silanol groups on quartz. These extra “sticky spots” help ZSC pack densely on the surface, even though the bonding is still mostly physical rather than forming new chemical bonds.
Fitting the patterns with simple models
To describe these behaviors in a way that engineers can use, the authors compared their data with standard mathematical curves known as adsorption isotherms. They tested three of them—Langmuir, Freundlich, and Temkin. For both surfactants, the Langmuir model, which assumes a single, uniform layer of molecules on a fairly even surface, provided the best overall fit, with very high correlation numbers for SDS and ZSC. The other two models also fit reasonably well and suggested that the real quartz surface still has some variation and can host multilayer adsorption, especially at higher concentrations. Analysis of the Temkin model parameters pointed to relatively low adsorption energies, supporting the view that the surfactants are held by physical forces rather than by strong chemical bonding.
What this means for greener oil production
For practical oil recovery, very high adsorption is a double-edged sword. ZSC’s strong tendency to stick to quartz means more surfactant is lost to the rock and less is available where it does the most good, at the oil–water boundary. SDS, in contrast, wastes less in this way on quartz-rich rocks. The study therefore concludes that, in simple quartz systems, ZSC is much more strongly adsorbed than SDS, and both follow primarily monolayer, physical adsorption described well by the Langmuir model. While this may limit ZSC’s direct use on its own, earlier work suggests that adding alkalis or nanoparticles can curb adsorption for both natural and synthetic surfactants. The present results provide a solid baseline for designing such improved, more environmentally friendly formulations and will help guide future tests in more realistic sandstones that also contain clays.
Citation: Shirali, A., Ebrahimi, M., Hemmati-Sarapardeh, A. et al. Comparative analysis of natural and synthetic surfactant adsorption by quartz minerals: an experimental study. Sci Rep 16, 7852 (2026). https://doi.org/10.1038/s41598-026-39608-y
Keywords: enhanced oil recovery, surfactant adsorption, natural surfactant, sandstone reservoir, quartz minerals