Clear Sky Science · en
Optimization and mechanistic insights into SiO2 nanoparticle–CTAB surfactant pickering emulsions for water mobility control
Why slowing down water can boost oil recovery
In many aging oil fields, companies inject water underground to push out more oil. But the water often takes the easiest shortcuts through the rock, racing through large open pathways and leaving much of the oil behind. This study explores a clever way to "thicken" and redirect that injected water using specially designed mixtures of tiny particles and soap-like molecules, creating long‑lived emulsions that can block these fast lanes and force water to sweep through oil‑rich zones instead.
Building stable mixtures of oil, water, and tiny particles
The researchers focused on a type of emulsion called a Pickering emulsion, where solid particles sit at the boundary between oil and water and act like armor around droplets. Here, they used very small silica particles together with a common surfactant (CTAB), which behaves like a detergent. By adjusting how much particle, how much surfactant, and how much water versus oil they mixed, they could tune how stable the droplets were and whether the continuous phase was oil or water. Using a statistical design tool, they ran a limited but carefully chosen set of experiments and built a mathematical model that predicts how these three knobs control the stability of the mixture.
Finding the sweet spot for long‑lasting droplets
The team quantified stability by tracking how much of each sample remained well mixed after days of resting and by measuring droplet sizes under a microscope. They found that particle concentration was the most powerful lever: more particles built a stronger protective shell around droplets and slowed their tendency to merge. The surfactant had a strong supporting role, helping the particles spread out and grab the oil–water boundary more effectively. The ratio of water to oil mattered in a non‑obvious way. Too little or too much water led to less stable mixtures, while an intermediate water share, around 60/40 water‑to‑oil, gave the most robust emulsions. The researchers captured these trends in a predictive equation that matched their measurements very well.
Heat, flow, and how the fluid behaves under stress
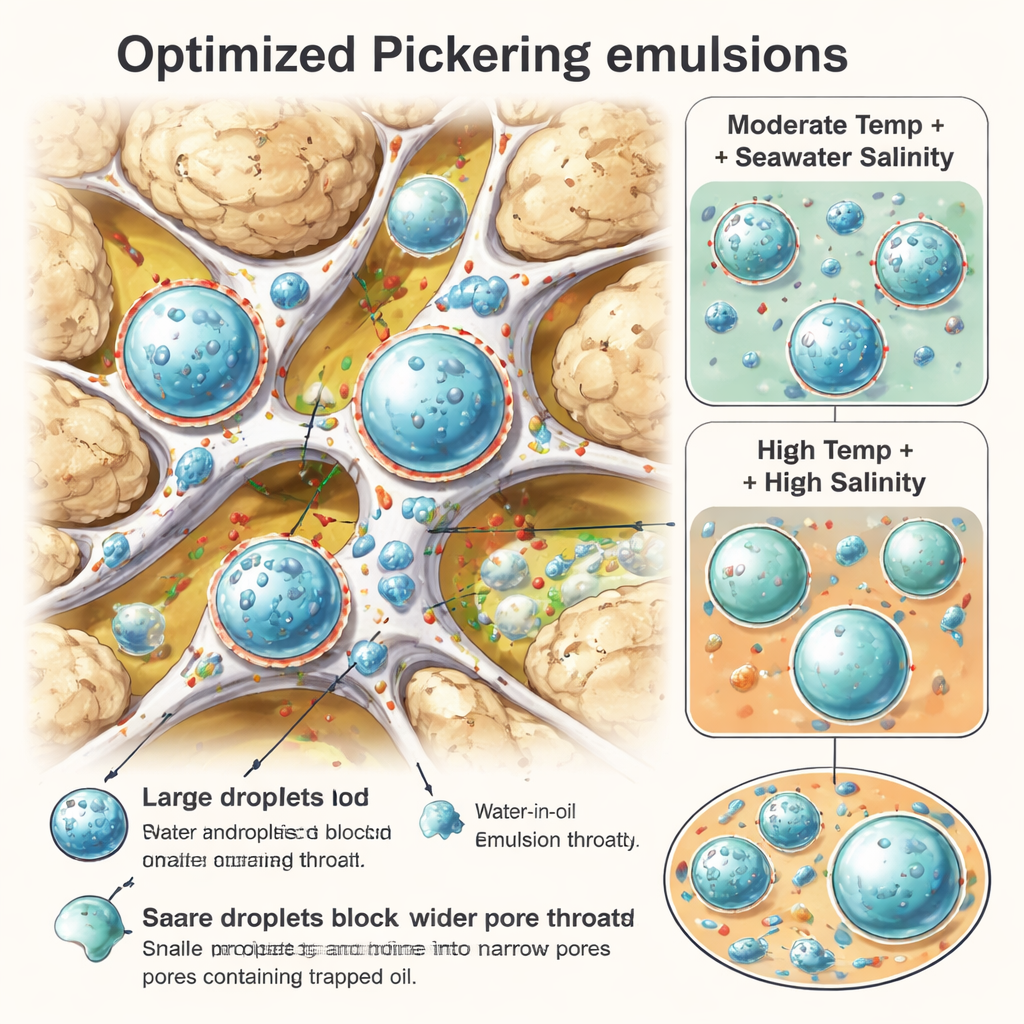
Real oil reservoirs are hot, so the group tested how their best formulations behaved from room temperature up to 120 °C. Up to roughly 80 °C, the emulsions stayed fairly stable, with only modest growth in droplet size. At higher temperatures, droplets swelled dramatically, a sign that they were merging and the protective shells were breaking down, and overall stability dropped. When they probed how these fluids flowed in a viscometer, they found that the emulsions were "shear‑thinning": thick and viscous when moving slowly, but thinning out as they were sheared faster. Adding more water increased the apparent thickness at low shear but also made the structure more fragile, consistent with a crowded network of droplets that can rearrange under stress.
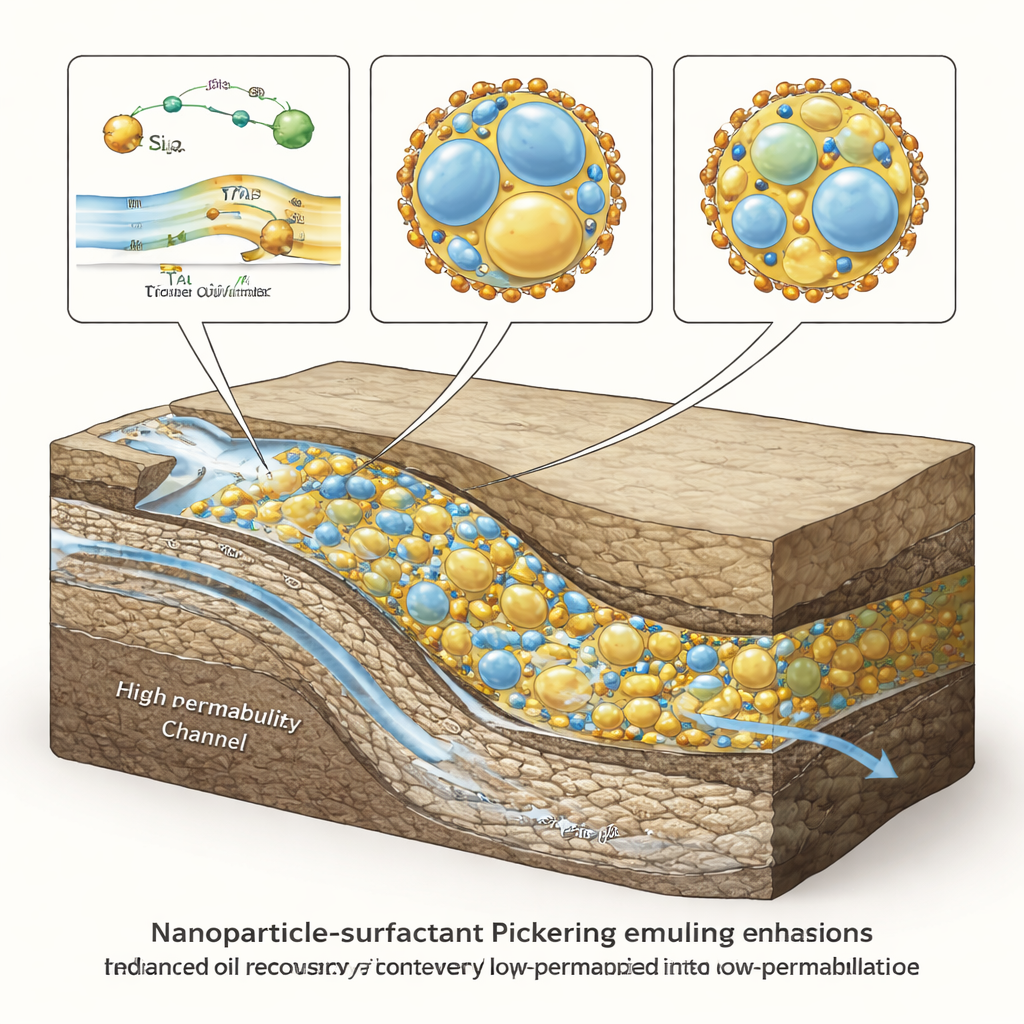
Watching emulsions redirect water inside rock
To see whether these lab‑made fluids could actually improve oil production, the scientists etched a glass model of a rock with both wide, easy‑flow channels and tighter, harder‑to‑reach pores. After saturating the model with crude oil, they injected brine alone and saw water quickly finger through the high‑permeability pathway, leaving most oil untouched. When they followed with the optimized Pickering emulsions, the picture changed: droplets jammed in the wider throats, increasing flow resistance in the easy path and forcing the injected water into the smaller pores. With seawater salinity and a high water content in the emulsion (about 75% water), the oil recovery climbed to about two‑thirds of the oil originally in place. Under much saltier conditions, however, the emulsions became less stable, flow diversion weakened, and recovery dropped to roughly one‑third.
What this means for real oil fields
For a non‑specialist, the key lesson is that how we package water and oil together can dramatically change how they move underground. By wrapping droplets in a coat of nanoparticles and surfactant, this work shows it is possible to create mixtures that are stable at realistic temperatures, flow like a thick but flexible fluid, and selectively block the rock’s natural fast lanes. When tuned to the right recipe and salinity, these emulsions can slow the injected water just enough to sweep more thoroughly through the reservoir, unlocking significantly more oil without major changes to existing infrastructure.
Citation: Ahmadi, B., Sahraei, E. Optimization and mechanistic insights into SiO2 nanoparticle–CTAB surfactant pickering emulsions for water mobility control. Sci Rep 16, 7802 (2026). https://doi.org/10.1038/s41598-026-39583-4
Keywords: Pickering emulsions, nanoparticles, enhanced oil recovery, water mobility control, porous media flow