Clear Sky Science · en
CPAP and high-flow nasal oxygen also reduce lung, diaphragm, and accessory muscle injury in experimental self-inflicted lung injury
Why breathing support matters beyond the ICU
When someone struggles to breathe because their lungs are badly injured, every breath can become a workout that quietly damages the lungs and breathing muscles even more. This study in rats asks a question that matters for many patients with severe pneumonia or COVID‑like illness: can gentler forms of breathing support, like common hospital devices that push air through a mask or soft nasal prongs, actually protect the lungs and the main breathing muscle—the diaphragm—from this self‑inflicted harm?
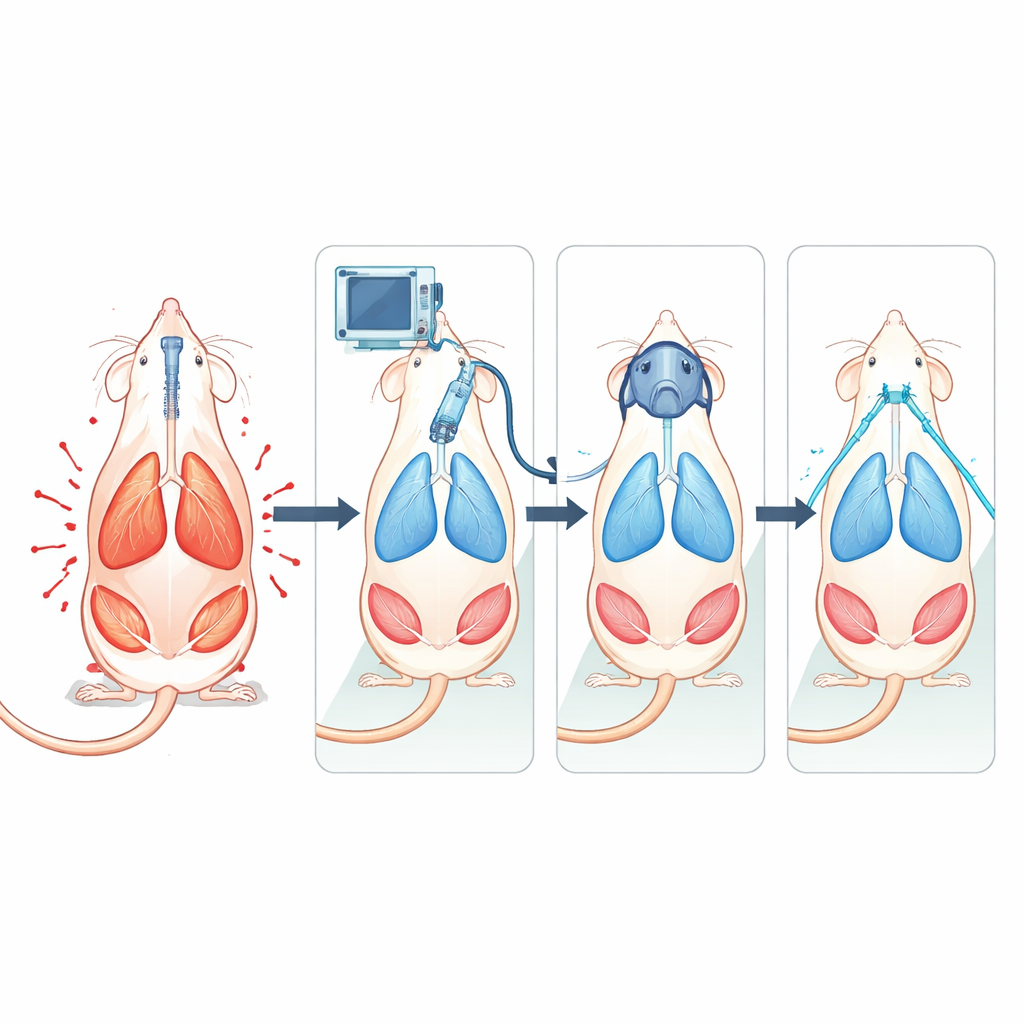
When hard breathing becomes a second hit
The researchers focused on a problem doctors increasingly recognize: in very sick lungs, the effort of breathing itself can act like a “second hit.” As injured lungs become stiff and oxygen levels fall, the body responds with stronger, faster breaths, pulling harder with the diaphragm and the smaller helper muscles in the chest and abdomen. Those powerful swings in pressure and repeated stretching can tear delicate air sacs and strain blood vessels, a process called patient self‑inflicted lung injury. At the same time, the muscles doing this extra work can suffer microscopic damage, a kind of overuse injury inside the chest.
Testing common hospital supports in a controlled model
To probe these effects, the team created a controlled lung injury in anesthetized rats by washing out the natural surfactant that normally keeps tiny air sacs from collapsing. After this “first hit,” animals were assigned to one of several types of oxygen support for three hours. One group breathed on their own with simple low‑flow oxygen through a mask, mimicking minimal support. Others received continuous positive airway pressure (CPAP) through a snug mask, high‑flow nasal oxygen (HFNO) through warmed, humidified gas, or fully controlled mechanical ventilation that took over the work of breathing. The scientists carefully tracked how hard the animals were breathing using pressure probes in the esophagus, ultrasound of the diaphragm, and electrical recordings from abdominal muscles, along with bedside‑style lung ultrasound and blood tests.
What happened inside the lungs and muscles
After the breathing support period, the team examined lung tissue and a set of breathing muscles under the microscope: the diaphragm, chest wall muscles between the ribs, neck muscles that help lift the chest, and abdominal muscles that drive forceful exhalation. Animals left to breathe on their own with simple oxygen showed the greatest damage: thickened and sometimes ruptured air sacs, small hemorrhages, fluid around blood vessels, and early inflammatory changes. Their breathing muscles showed more swelling, fragmentation of fibers, and other signs of strain. All three support therapies—mechanical ventilation, CPAP, and HFNO—reduced this structural damage in both lungs and muscles. Protection formed a clear gradient in the lungs: strongest with mechanical ventilation, intermediate with CPAP, and weakest with HFNO. Notably, CPAP offered the best shield for the diaphragm, even better than full ventilation, while mechanical ventilation most strongly protected the helper muscles.
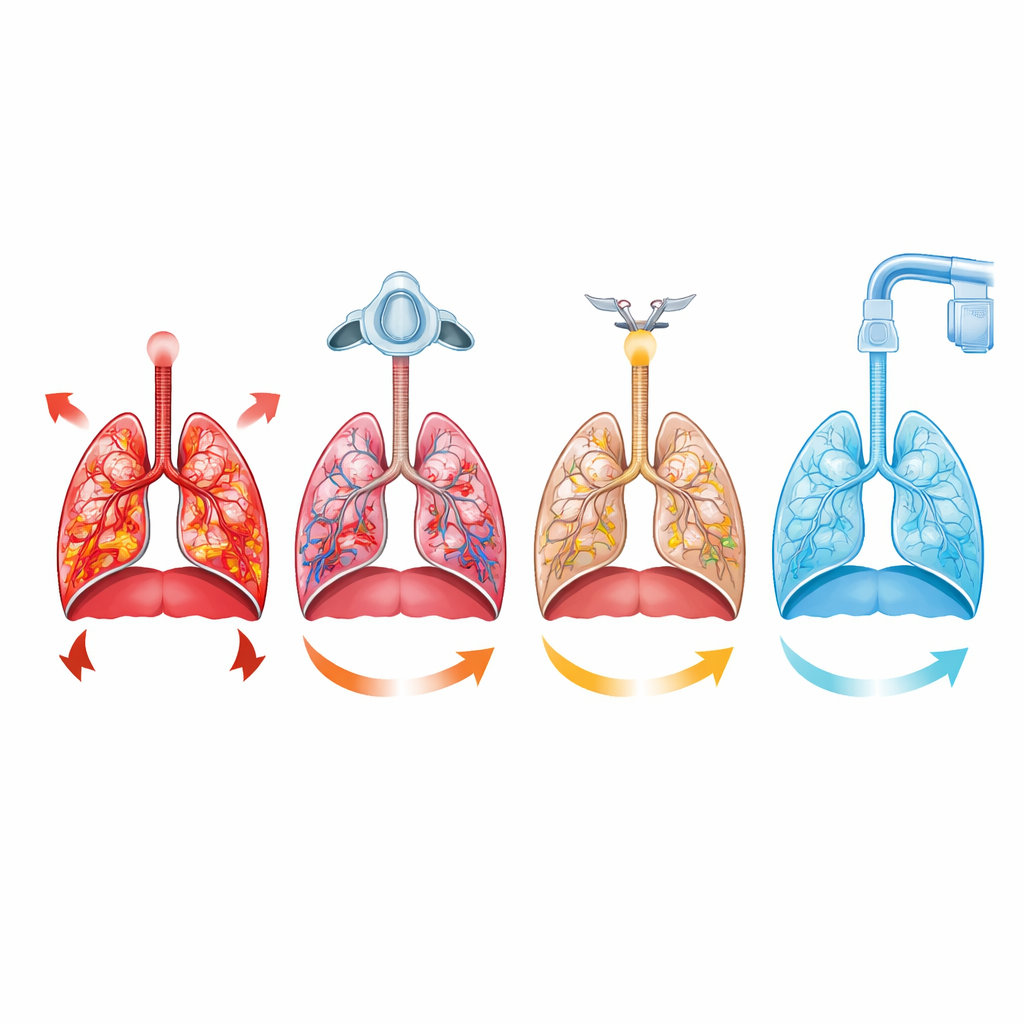
How effort, support, and injury are linked
The monitoring data helped connect these tissue changes to what was happening in real time. Compared with simple oxygen or HFNO, CPAP and mechanical ventilation cut down both the effort to inhale and the active push to exhale, reduced the visible use of neck and abdominal muscles, and limited how far the diaphragm had to move each breath. Lung ultrasound showed that CPAP helped prevent the loss of air‑filled lung regions over time, suggesting that gentle continuous pressure kept air sacs from repeatedly collapsing and reopening. Statistical analysis revealed that higher inspiratory and expiratory efforts were directly associated with more lung and accessory muscle injury, reinforcing the view that unchecked breathing drive can worsen fragile lungs. Surprisingly, diaphragm injury correlated more with forceful expiration than with inspiration, hinting that the way the diaphragm behaves while exhaling may also matter for long‑term muscle health.
What this could mean for patient care
For a general reader, the takeaway is that not all forms of “breathing help” are equal, and the goal is not just to raise oxygen levels but to tame the hidden costs of hard breathing. In this rat model, any structured support—mask pressure, high‑flow nasal gas, or full mechanical ventilation—limited the cascading injury to lungs and breathing muscles compared with simple oxygen alone. Mechanical ventilation best protected lung tissue overall but risks over‑resting the diaphragm; CPAP struck a promising balance, easing the load on the lungs while keeping the main breathing muscle safely active. While animal results cannot be applied directly to patients, the study supports a growing idea in critical care: carefully chosen noninvasive support, started early and monitored with simple bedside tools, may help prevent the body from literally breathing itself into deeper trouble.
Citation: Reveco, S., Llancalahuen, F.M., Caviedes, P. et al. CPAP and high-flow nasal oxygen also reduce lung, diaphragm, and accessory muscle injury in experimental self-inflicted lung injury. Sci Rep 16, 8399 (2026). https://doi.org/10.1038/s41598-026-39564-7
Keywords: acute lung injury, noninvasive ventilation, CPAP, high-flow nasal oxygen, respiratory muscle injury