Clear Sky Science · en
Genomic and in vitro characterization of two lytic bacteriophages infecting multidrug-resistant Erwinia sp. strain AnSW2-5
Why Crop-Killing Bacteria Matter to Everyone
Across the globe, farmers are battling bacterial diseases that rot fruits, shrivel leaves, and devastate harvests. Many of these outbreaks are now caused by strains that no longer respond to common antibiotics, threatening both food supplies and the livelihoods that depend on them. This study explores an emerging alternative: using viruses that attack bacteria, called bacteriophages, to safely disarm a particularly stubborn plant pathogen found in soil linked to heavy antibiotic use.
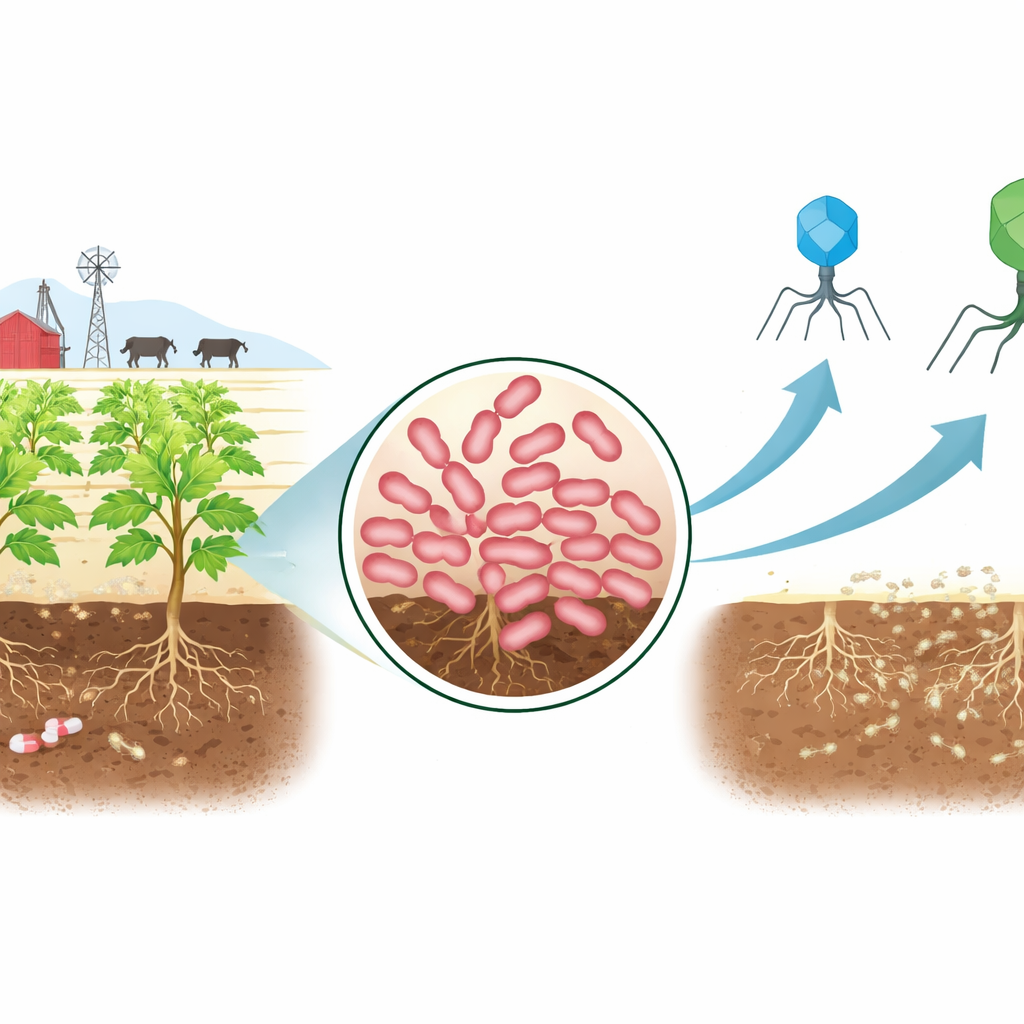
A Tough New Farmyard Invader
The researchers began by sampling soil from a cattle enclosure known to receive livestock waste and antibiotics. From this challenging environment they isolated a strain of Erwinia, a bacterial group that includes notorious culprits behind fire blight in apples and soft rot in potatoes. Tests showed that this strain, named AnSW2-5, could withstand multiple major antibiotic families, including drugs used against a broad range of infections. Genetic analysis confirmed why: its DNA carried several known resistance genes and powerful molecular pumps that eject antibiotics from the cell. At the same time, genome comparisons revealed that this strain is distinct from previously described Erwinia species, making it a useful model for studying how to control new, hard-to-treat plant pathogens.
Two Tiny Predators with Different Strengths
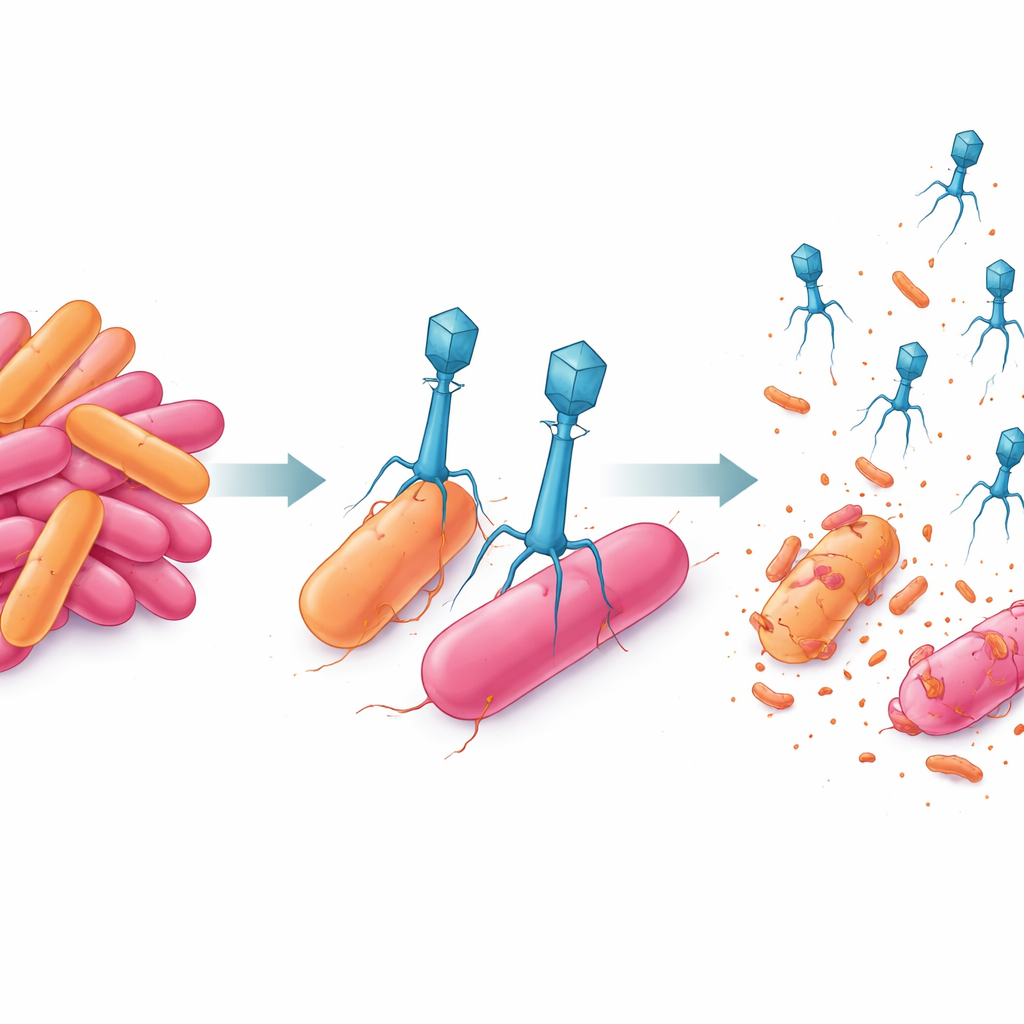
To find natural enemies of this multidrug-resistant bacterium, the team turned to freshwater from the same site and enriched it for bacteriophages that could infect AnSW2-5. They isolated two promising candidates, labeled P-A and P-K. Under the electron microscope, P-A appeared compact with a short tail, a design linked to fast, direct infection. P-K, in contrast, carried a longer, contractile tail with a complex base structure, suggesting a more forceful way of drilling into its host. Sequencing their genomes showed that both phages are strictly lytic: they invade, replicate, and burst the cell open, rather than hiding quietly in bacterial DNA. Yet they do so with very different genetic toolkits—P-A’s streamlined genome supports rapid attack, while P-K’s larger genome encodes more elaborate structural and replication machinery.
How the Viral Tag Team Attacks
When the scientists tracked each phage’s life cycle in the lab, they found that P-A strikes quickly: it needs only about 20 minutes before new viral particles begin to appear, and each infected bacterium releases around 70 offspring. P-K takes longer—about 35 minutes before it starts to produce new phages—but each infected cell yields roughly 110 new particles. In co-culture tests where bacteria and phages grow together over three days, both phages alone slowed bacterial growth but did not fully prevent it. The bacteria eventually bounced back, reflecting the appearance of resistant survivors. However, when P-A and P-K were combined in a single cocktail, they reduced the bacterial population by more than 80% and kept it suppressed for the full 72-hour experiment.
Keeping Resistance from Gaining a Foothold
One of the most striking findings was how the phage pair affected the rise of resistant mutants. When exposed to either P-A or P-K alone, a small fraction of bacteria—roughly one in a million to one in ten million—managed to escape and grow despite phage attack. But when both phages were used together, resistant colonies became so rare that they fell below the experiment’s detection limit. This suggests that the two viruses likely latch onto different targets on the bacterial surface or disrupt the cell in complementary ways. For a single bacterium to survive, it would need to overcome both at once, a vanishingly unlikely event. In practical terms, this makes the cocktail far more robust against the evolutionary tricks that often doom antibiotics.
What This Could Mean for Future Harvests
Taken together, the work shows that carefully chosen pairs of bacteriophages can do more than just knock down dangerous plant bacteria—they can also box in the bacteria’s ability to evolve resistance. By combining a fast-acting phage with one that produces larger numbers of offspring, the researchers created a viral tag team that kept a multidrug-resistant Erwinia strain in check for days while preventing detectable escape variants. Although these trials were done in the lab rather than in orchards or fields, they point toward a future in which targeted phage mixtures become part of standard integrated pest management, helping farmers protect crops with far less dependence on traditional antibiotics.
Citation: Baek, K., Goh, J. & Choi, A. Genomic and in vitro characterization of two lytic bacteriophages infecting multidrug-resistant Erwinia sp. strain AnSW2-5. Sci Rep 16, 8172 (2026). https://doi.org/10.1038/s41598-026-39563-8
Keywords: bacteriophage biocontrol, plant bacterial diseases, antibiotic resistance in agriculture, Erwinia phage therapy, phage cocktails