Clear Sky Science · en
A lncRNA and radiomics-based model for predicting the response of non-small cell lung cancer to chemo- and radio-therapy
Why predicting treatment success matters
For people diagnosed with advanced non-small cell lung cancer, chemotherapy and radiotherapy are often the main options when surgery or targeted drugs are not possible. Yet doctors still have no simple way to tell in advance who will benefit from these harsh treatments and who will not. This study explores whether a blood-based molecule and tiny patterns hidden in CT scans can work together as a kind of early warning system, helping to match patients with the treatments most likely to help them.
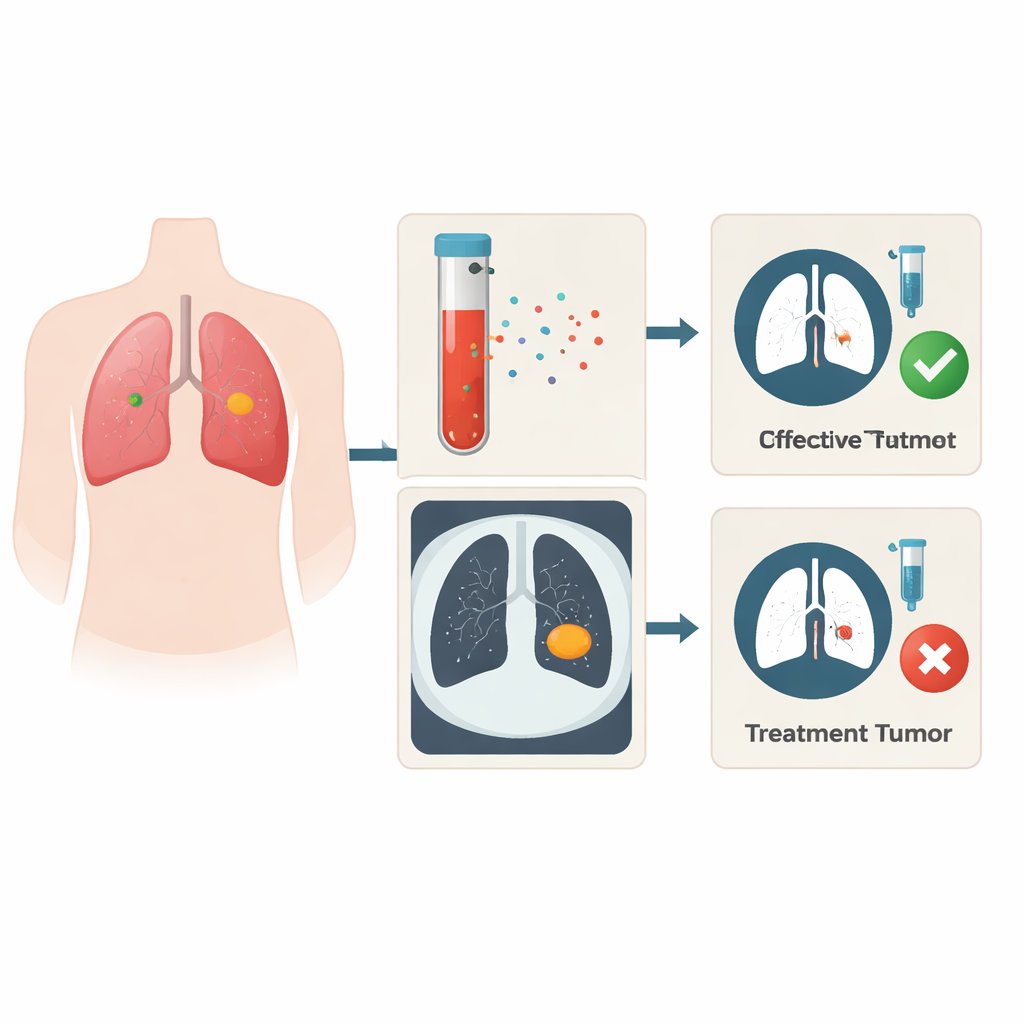
A signal hiding in the blood
The researchers focused on a type of genetic material called long non-coding RNA, or lncRNA, which does not make proteins but can strongly influence how cancer cells behave. These molecules can be packaged into microscopic bubbles released by tumors into the bloodstream, meaning they can be measured with a simple blood draw. By sequencing lncRNAs from blood samples of lung cancer patients who either responded well or poorly to chemo- and radiotherapy, and by cross-checking these results with large public cancer databases, the team homed in on one standout molecule: an lncRNA named MIF-AS1. Patients and cell lines that resisted standard treatment consistently showed higher levels of MIF-AS1.
How this molecule may fuel stubborn tumors
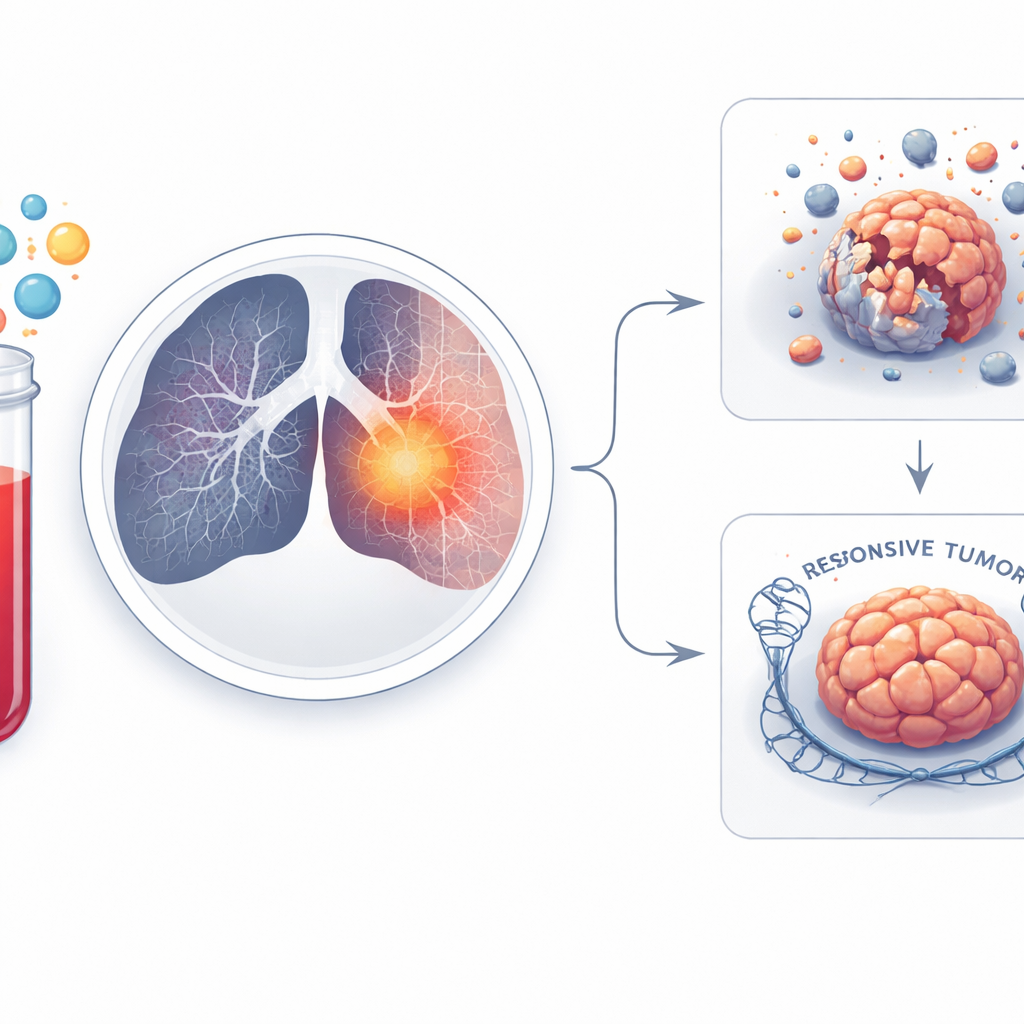
To understand whether MIF-AS1 was simply a marker or an active troublemaker, the scientists reduced its levels in lung cancer cells grown in the lab. When MIF-AS1 was switched off, the cells grew more slowly, spread less, and became easier to kill with the common chemotherapy drug cisplatin. The team then used computational methods to map how MIF-AS1 might interact with other RNA molecules and genes. This analysis pointed to RAD21, a protein that helps repair damaged DNA, as a likely partner. In tumor samples, higher MIF-AS1 went hand-in-hand with higher RAD21. When MIF-AS1 was knocked down, RAD21 levels dropped, and cells became more vulnerable to treatment, suggesting that this RNA–protein pair may help cancer cells patch up the DNA damage caused by chemo- and radiotherapy.
Reading tumors through their CT "fingerprints"
Modern CT scans capture far more detail than the human eye can see. Radiomics is a technique that turns these images into thousands of numerical features describing shape, brightness, and texture. In this study, the team analyzed 1,409 such features from CT scans of lung cancer patients. Using a statistical method that selects only the most informative signals, they narrowed this huge list down to just two key features linked to whether tumors shrank or persisted after treatment. One feature tended to be higher in patients whose tumors responded, while the other was higher in those whose tumors resisted therapy, indicating that resistant tumors may have distinctive structural or textural patterns on scans.
Uniting blood tests and imaging into one prediction tool
The researchers then combined the blood level of MIF-AS1 with the two CT-based features into a single prediction model. Tested first on a smaller training group and then on an independent group of 124 patients, this model did a better job of separating likely responders from non-responders than any single measure alone. Its accuracy, expressed by a standard metric called the area under the curve, reached 0.808 in the validation group—higher than using MIF-AS1 alone and noticeably better than a commonly used blood marker, CEA. Importantly, MIF-AS1 levels were not strongly tied to age, sex, smoking history, tumor size, or stage, suggesting that this signal reflects something specific about treatment sensitivity rather than general disease severity.
What this could mean for future patients
In plain terms, the study suggests that a simple blood test, combined with a smart readout of routine CT images, could help doctors foresee whether a person’s lung tumor is likely to bow to chemo- and radiotherapy or resist it. The lncRNA MIF-AS1 emerges both as a promising non-invasive marker and as a potential driver of resistance through enhanced DNA repair, working hand-in-hand with the RAD21 protein. Although the work needs to be confirmed in larger, multi-center trials and across different lung cancer subtypes, it points toward a future in which treatment plans are guided not just by what tumors look like to the naked eye, but by the hidden molecular and imaging signatures that reveal how they are likely to behave.
Citation: Ye, F., Yin, Y., Wang, J. et al. A lncRNA and radiomics-based model for predicting the response of non-small cell lung cancer to chemo- and radio-therapy. Sci Rep 16, 8337 (2026). https://doi.org/10.1038/s41598-026-39560-x
Keywords: non-small cell lung cancer, treatment resistance, liquid biopsy, radiomics, lncRNA biomarker