Clear Sky Science · en
New benzimidazole-alkanesulfonate conjugates as cholinesterase inhibitors with in vitro and in silico validation
Why this research matters for memory and aging
Alzheimer’s disease slowly erodes memory, independence, and quality of life for millions of people worldwide, yet today’s medicines mostly ease symptoms without slowing the underlying damage. This study explores a fresh family of laboratory‑made molecules designed to better protect a key brain chemical involved in learning and memory, while also fighting harmful oxidative stress. By blending features of several successful drug types into a single structure, the researchers aim to move a step closer to more effective treatments for Alzheimer’s disease.
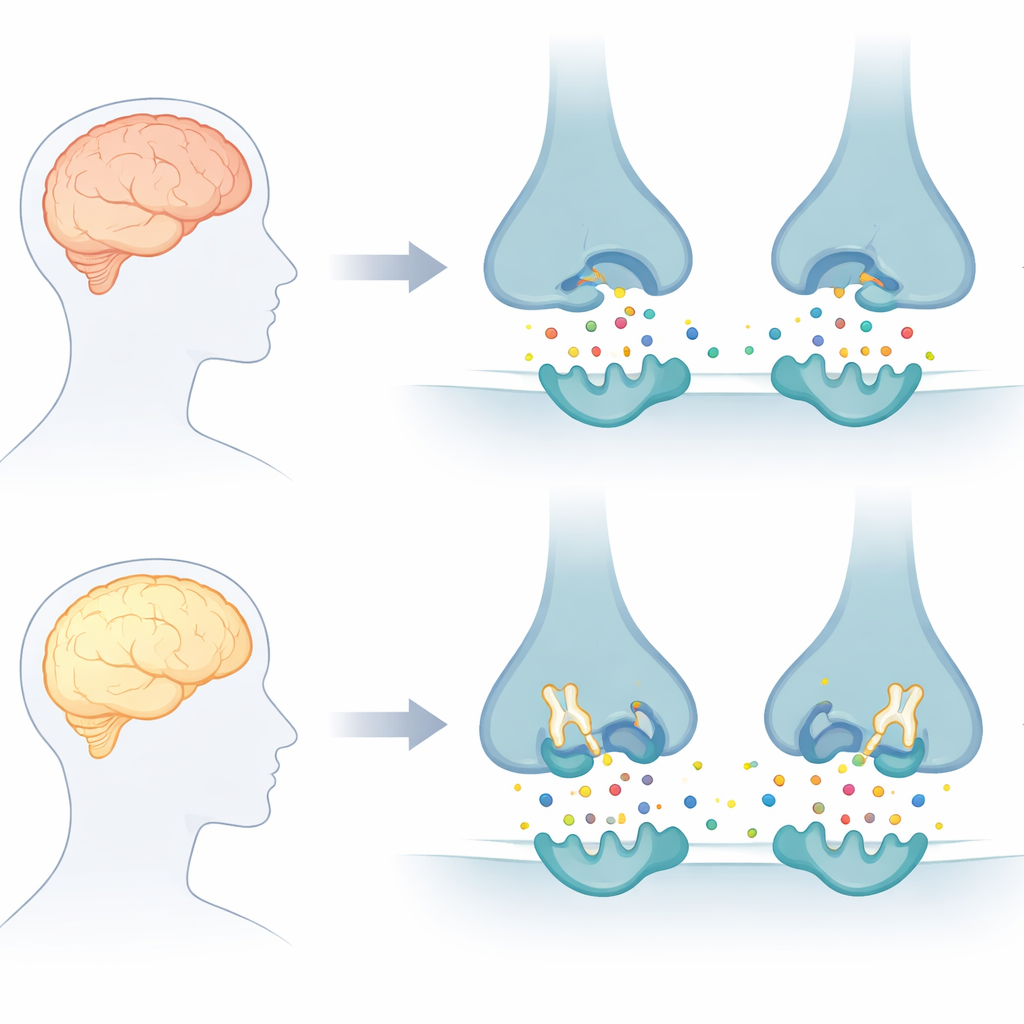
The brain signal that fades too fast
Healthy brain cells communicate using chemical messengers, one of the most important being acetylcholine. In Alzheimer’s disease, acetylcholine is broken down too quickly by two enzymes, acetylcholinesterase and butyrylcholinesterase, contributing to memory loss and confusion. Current drugs mainly block just one of these enzymes and only for a limited time, which is why they offer short‑term symptom relief rather than long‑term protection. The authors of this paper focused on designing “dual” blockers that could target both enzymes at once and potentially give acetylcholine a better chance to do its job.
Building a smarter molecular key
To craft these dual blockers, the team used a strategy called molecular hybridization: they stitched together pieces from three types of chemical building blocks already known to interact well with cholinesterase enzymes. These pieces included a benzimidazole ring often found in medicines, a sulfonate group that can bind strongly to enzyme pockets, and a hydrazone linker that helps connect the parts in a flexible way. The result was a series of related compounds, labeled 4a through 4r, whose structures were carefully confirmed using standard chemistry techniques such as infrared and nuclear magnetic resonance spectroscopy.
Testing enzyme blocking and antioxidant power
The new compounds were first screened in test‑tube experiments to see how well they could slow down the two cholinesterase enzymes. Most members of the series showed moderate to strong blocking effects, but five in particular—known as 4b, 4h, 4i, 4q, and 4r—stood out for their ability to inhibit acetylcholinesterase. When the researchers measured more precise potency values, several of these molecules matched or even outperformed donepezil, one of the main drugs currently prescribed for Alzheimer’s. The team also examined whether the best enzyme blockers could neutralize free radicals, unstable molecules that damage brain cells. Compounds 4q and 4r showed especially strong antioxidant activity across several different tests, approaching or surpassing the performance of the reference antioxidant vitamin C.
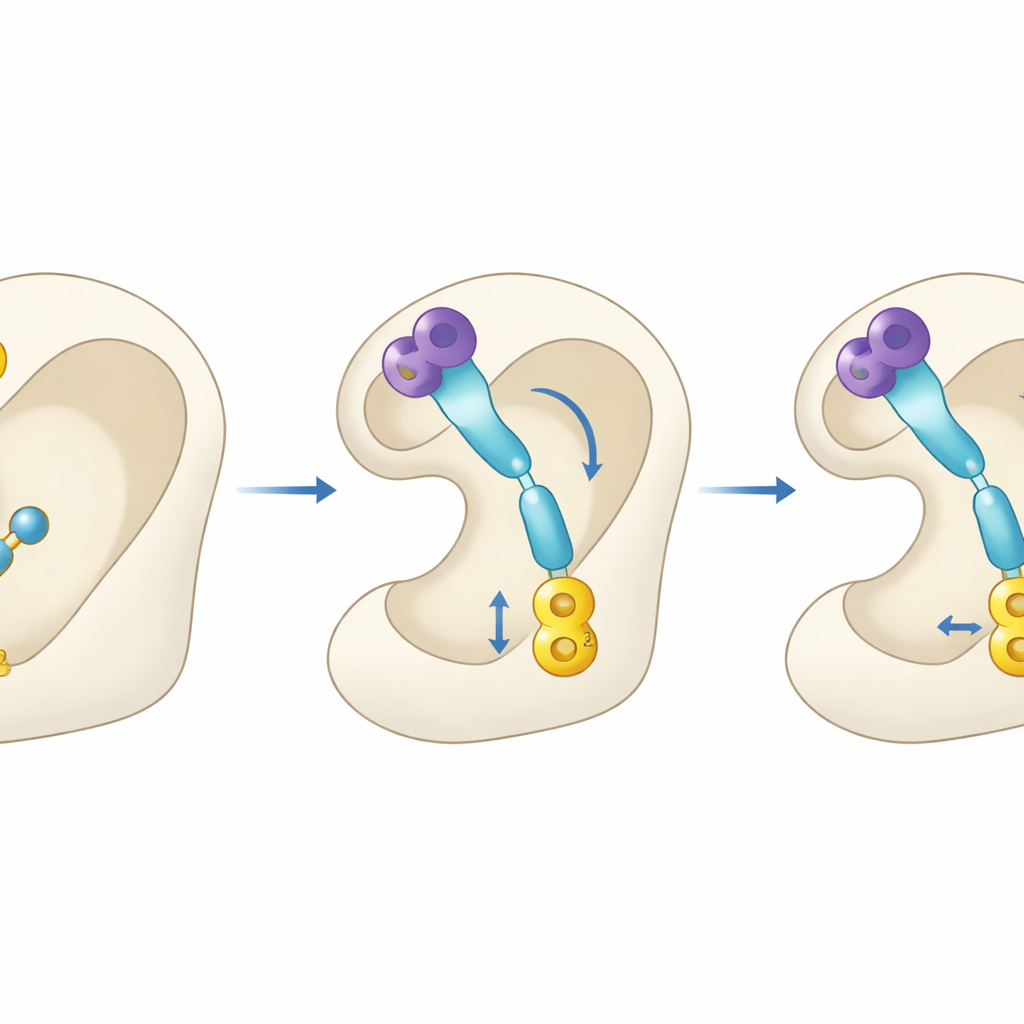
Peering into the enzyme’s lock
Because these experiments were done outside the body, the scientists turned to computer modeling to predict how the molecules might behave as medicines. Using widely adopted in silico tools, they estimated absorption, distribution, and metabolism properties, and then simulated how the compounds would fit into the three‑dimensional structure of acetylcholinesterase. The docking studies suggested that the benzimidazole portion of each molecule nests near the outer opening of the enzyme, while the sulfonate‑bearing end reaches deep into the catalytic center where acetylcholine is normally broken down. Notably, 4q and 4r formed several stabilizing interactions inside the enzyme pocket and showed higher calculated binding strength than donepezil itself, echoing their strong test‑tube performance.
What this means for future Alzheimer’s treatments
Taken together, the findings show that this new class of benzimidazole‑alkanesulfonate conjugates can strongly block acetylcholinesterase, show useful antioxidant behavior, and bind to their enzyme target in a way that is consistent with current drug design principles. While these molecules are far from ready for use in patients—they have not yet been tested in animals or humans—they offer a promising blueprint for next‑generation Alzheimer’s treatments that combine enzyme inhibition with protection against oxidative stress. With further refinement and biological testing, some members of this family could one day contribute to drugs that better preserve memory and thinking in the aging brain.
Citation: Omar, M.A., Al-Ashmawy, A.A.K., Abd El Salam, H.A. et al. New benzimidazole-alkanesulfonate conjugates as cholinesterase inhibitors with in vitro and in silico validation. Sci Rep 16, 8946 (2026). https://doi.org/10.1038/s41598-026-39534-z
Keywords: Alzheimer’s disease, acetylcholinesterase inhibitors, benzimidazole derivatives, antioxidant compounds, drug design