Clear Sky Science · en
Proteomic profiling of bronchoalveolar lavage following human segmental endotoxin challenge—a potential exacerbation model
Why a short burst of lung inflammation matters
Chronic lung diseases like chronic obstructive pulmonary disease (COPD) often flare up suddenly when the airways become acutely inflamed, usually due to infections. These episodes, called exacerbations, send many people to the hospital and can permanently worsen breathing. Studying them directly in patients is difficult and risky. This study uses a carefully controlled, short‑lived irritation of the lung in healthy volunteers to mimic some features of these flare‑ups, then measures hundreds of proteins in lung fluid to see which signals switch on. The findings help researchers understand the chemistry of lung inflammation and may speed the search for new treatments.
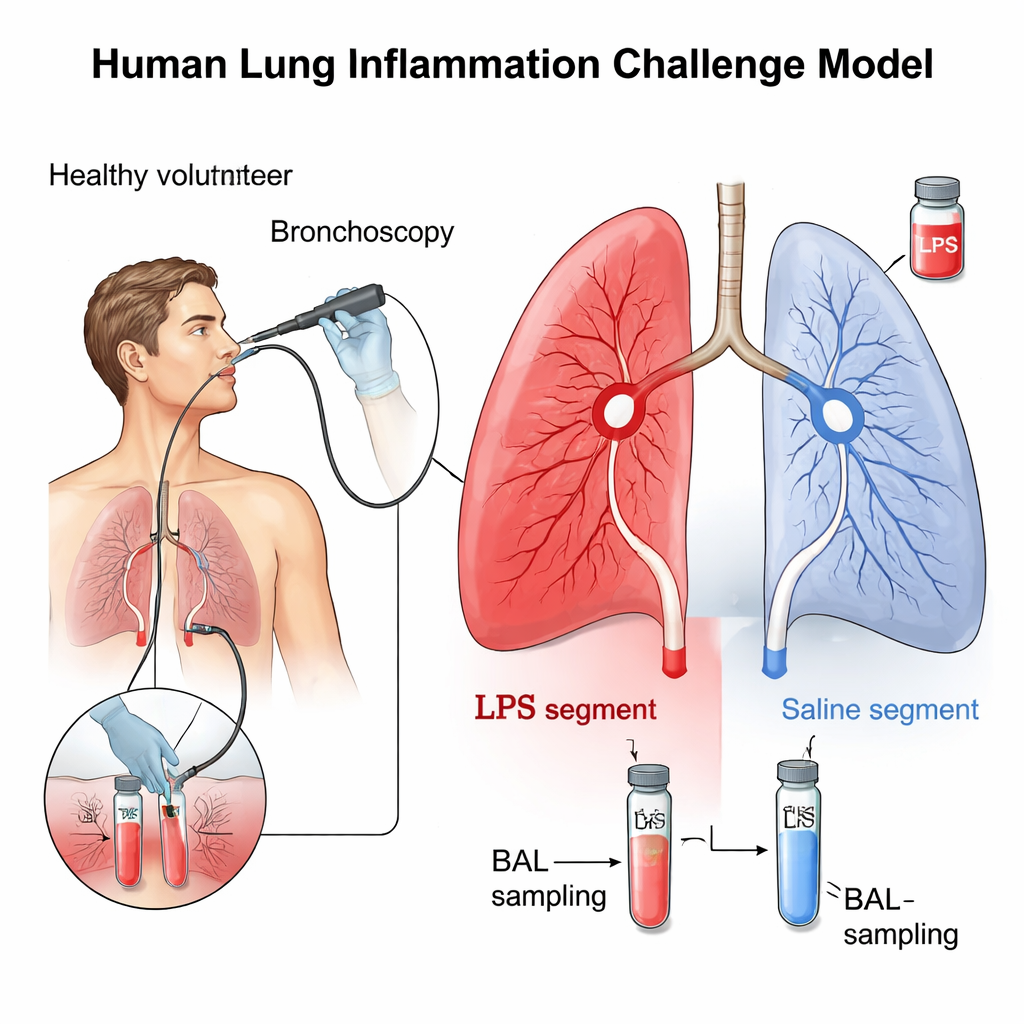
Creating a safe mini “lung flare‑up”
The researchers used a well‑established technique that briefly and locally irritates a small region of the lung. Ten healthy, non‑smoking adults underwent bronchoscopy, where a thin flexible tube is guided into the airways. Through this tube, the team placed a tiny dose of bacterial endotoxin, known as lipopolysaccharide (LPS), into one lung segment and simple saline into a matching segment on the other side as a control. LPS is a natural component of certain bacteria that strongly activates the body’s defenses. After 24 hours, the same areas were washed with salt solution, a procedure called bronchoalveolar lavage (BAL), to collect the fluid and cells from deep inside the lungs for detailed analysis.
Taking a protein census in lung fluid
Rather than looking at just a few familiar markers, the team applied a high‑throughput technology (the SomaScan platform) that can measure about 1,500 different proteins at once in the BAL fluid. They compared samples taken before any challenge, after saline alone, and after LPS. As expected, pre‑challenge and saline samples looked very similar, confirming that the procedure itself did not provoke inflammation. In contrast, the LPS‑treated segments showed a dramatic shift: 599 proteins were significantly increased and only four were decreased. Many of the rising proteins are well‑known inflammatory messengers, including IL‑6 and IL‑8, enzymes such as myeloperoxidase (MPO) and MMP9, and acute‑phase proteins like C‑reactive protein (CRP) and von Willebrand factor (VWF). Principal component and clustering analyses showed that LPS samples formed a clearly separate group, underlining how strongly this local challenge changes the lung environment.
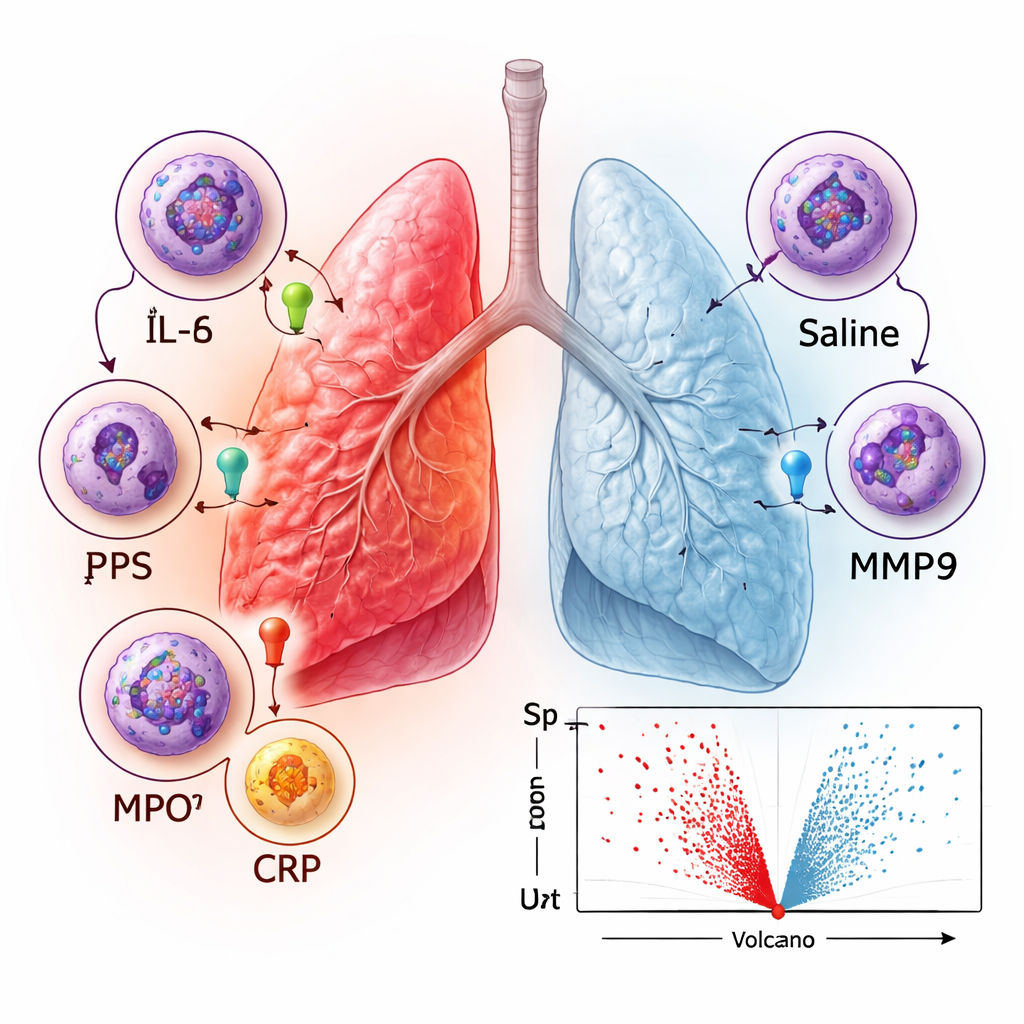
What the protein patterns reveal about lung defenses
To move beyond single markers, the team examined which types of biological processes were most enriched among the proteins that rose after LPS. The strongest signals involved general immune activity and responses to external or chemical stimuli, along with processes linked to cell movement and programmed cell death. Network analysis of how proteins interact with each other highlighted clusters of chemokines (molecules that attract immune cells), tissue‑remodeling enzymes called matrix metalloproteinases, and components of the immunoproteasome, a cellular machine that helps process foreign proteins for immune recognition. These patterns match what is known about how lungs respond to infection: immune cells such as neutrophils are drawn into the airways, harmful microbes are attacked, and the surrounding tissue is reshaped and, if needed, repaired.
Connecting the model to real COPD flare‑ups
The authors then asked how well this short‑term LPS model reflects real‑world COPD exacerbations. They compared their lung fluid data with a published list of blood biomarkers that rise when people with COPD experience a flare. Of 17 evaluable markers, 16—including IL‑6, IL‑8, CRP, fibrinogen, and several chemokines—were also elevated in BAL after LPS. Only one (sTREM‑1) did not change, in line with earlier mixed results on its usefulness. This strong overlap suggests that many of the same inflammatory pathways are engaged when a healthy lung is briefly challenged with LPS and when a vulnerable COPD lung undergoes a serious exacerbation. At the same time, the authors note that the LPS response is temporary and does not fully reproduce the chronic, structural damage seen in long‑term disease.
What this means for future treatments
In plain terms, this study shows that a carefully controlled, short‑lasting irritation of part of the lung in healthy volunteers can trigger a chemical storm that closely resembles what happens during COPD flare‑ups. By mapping hundreds of proteins that rise or fall during this response, researchers gain a detailed picture of which signals and pathways might be most important to target. Because the model is safe, repeatable, and already used in early drug trials, it offers a powerful testing ground for new anti‑inflammatory medicines and for identifying more precise biomarkers that could one day help doctors predict, detect, and better treat sudden worsening of chronic lung disease.
Citation: Gress, C., Müller, M. & Hohlfeld, J.M. Proteomic profiling of bronchoalveolar lavage following human segmental endotoxin challenge—a potential exacerbation model. Sci Rep 16, 6145 (2026). https://doi.org/10.1038/s41598-026-39528-x
Keywords: lung inflammation, COPD exacerbation, bronchoalveolar lavage, endotoxin challenge, proteomics