Clear Sky Science · en
Boosting visible-light photocatalysis with MWCNT-modified TiO2/SiO2/g-C3N4: efficient tetracycline removal in pure and hard water
Cleaning Tough Water Problems
Antibiotics and brightly colored dyes that wash out of homes, farms, and factories can linger in rivers and drinking water, where they harm wildlife and help drug‑resistant bacteria spread. This paper reports a new light‑powered material that can both grab these pollutants from water and then break them apart using visible light, including in hard water that usually makes cleanup difficult.
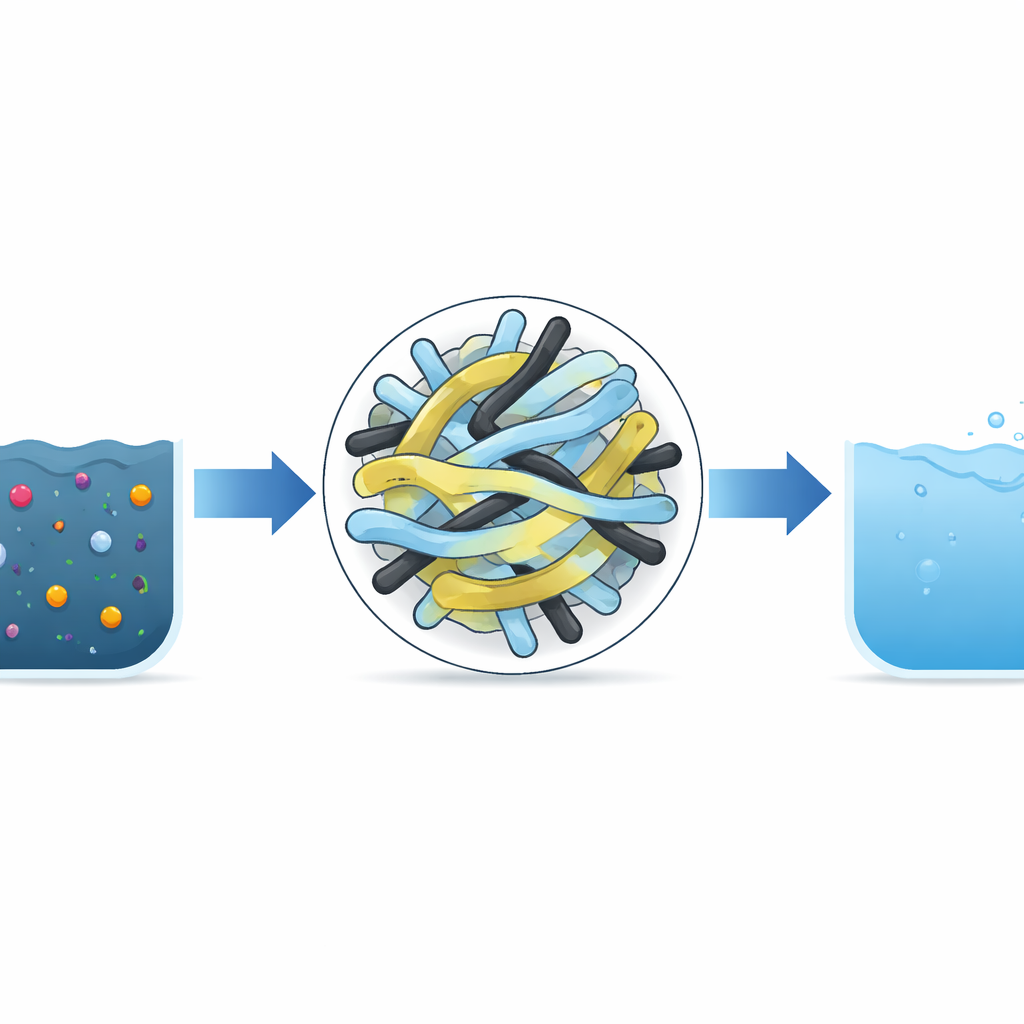
A New Kind of Cleaning Powder
The researchers built a powdered material that combines several known ingredients into one tiny, flower‑like structure. Titanium dioxide and silicon dioxide form sturdy spherical clusters, while very thin sheets of a carbon‑based solid called g‑C3N4 wrap around them. Threaded through this framework are multi‑walled carbon nanotubes, which are hollow carbon cylinders thousands of times thinner than a human hair. Together, these components create a highly porous architecture with a large internal surface area where pollutant molecules can stick before light‑driven reactions occur.
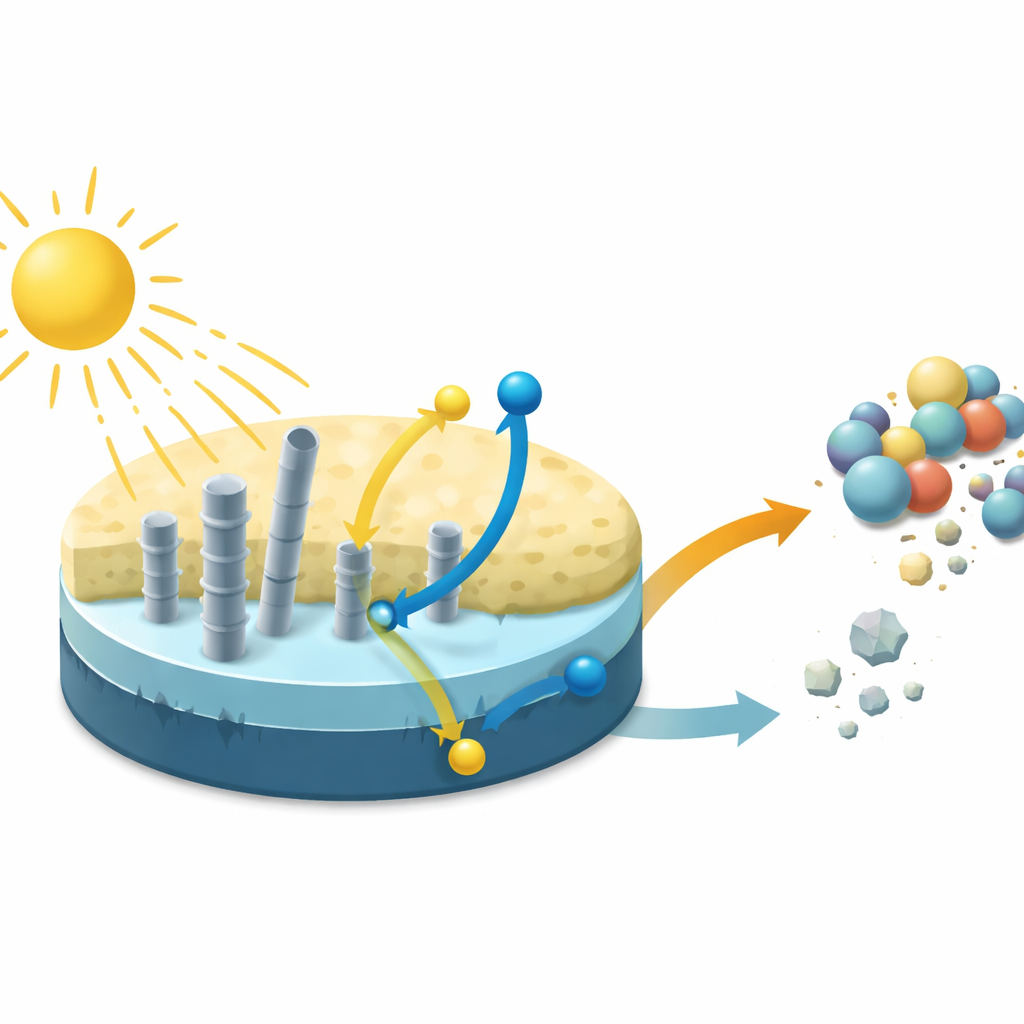
Using Light Instead of Harsh Chemicals
When visible light shines on this composite, it acts as a photocatalyst: it absorbs light energy and uses it to generate short‑lived, highly reactive forms of oxygen. These reactive species attack complex molecules such as the dye methylene blue and the antibiotic tetracycline, cutting them down into smaller, less harmful fragments and, eventually, into carbon dioxide and water. The carbon nanotubes play a dual role. They broaden the range of light the material can absorb and also serve as fast pathways for electric charges inside the solid, preventing wasted recombination of positive and negative charges and leaving more energy available for breaking pollutants apart.
Working Even in Hard Water
Real‑world water often contains minerals like calcium carbonate that make it "hard" and can coat or neutralize many catalysts. The team tested their material in both pure water and water loaded with calcium carbonate to mimic hard groundwater or seawater. A version of the catalyst without nanotubes lost noticeable efficiency in hard water, because ions in the water competed with pollutants for surface sites and interfered with the reactive species. In contrast, the nanotube‑modified composite removed about 92% of tetracycline in both pure and hard water, showing that its design overcomes many of the usual drawbacks of mineral‑rich conditions.
From Colorful Dyes to Stubborn Antibiotics
Beyond antibiotics, the material was challenged with methylene blue, a common test dye that represents many industrial colorants. By fine‑tuning the amount of nanotubes, the scientists found an optimal formulation containing about 11% nanotubes by weight. This version degraded over 90% of the dye under visible light in two and a half hours, clearly outperforming the nanotube‑free version. Detailed measurements of light emission, electrical behavior, and surface area confirmed that the nanotubes create many local junctions inside the powder that speed up charge separation and transfer, which in turn boosts its cleaning power.
Staying Strong Through Reuse
To see if the catalyst could be practical outside the lab, the authors ran multiple cleaning cycles on tetracycline‑contaminated hard water. Even after four rounds, the material still removed more than three‑quarters of the antibiotic within the same reaction time, and its internal crystal structure remained intact. Advanced chemical analyses of the treated water showed that the parent antibiotic peak nearly vanished and only smaller fragments remained, supporting the idea that the molecules were thoroughly broken down rather than just hidden on the particle surfaces.
What This Means for Safer Water
Overall, this work shows that carefully combining different nanoscale building blocks can yield a light‑driven powder that both captures and destroys stubborn water pollutants, and keeps working even in hard, mineral‑rich water. By harnessing visible light—the largest part of sunlight—such materials could form the basis of future treatment systems that quietly scrub antibiotics and dyes from wastewater before it returns to rivers, lakes, and taps.
Citation: Mohammaddarvish, S., Masoudi, A.A. & Hosseini, Z.S. Boosting visible-light photocatalysis with MWCNT-modified TiO2/SiO2/g-C3N4: efficient tetracycline removal in pure and hard water. Sci Rep 16, 7848 (2026). https://doi.org/10.1038/s41598-026-39505-4
Keywords: photocatalytic water treatment, antibiotic removal, titanium dioxide nanocomposite, carbon nanotubes, hard water pollution