Clear Sky Science · en
Smart CO2 capture and release and an insight into biological evolution based on its characteristics
Why carbon dioxide and plain water matter to all of us
Rising carbon dioxide (CO2) from burning coal, oil, and gas is heating the planet and driving extreme weather. Most proposed high‑tech solutions to pull CO2 back out of the air are costly or complex. This study explores a surprisingly simple ally in the fight against climate change: ordinary water. By carefully using water’s natural tendency to dissolve CO2, the authors outline a “smart” system that can capture, move, and release CO2 in a controllable way, potentially offering a cheaper and safer path to large‑scale cleanup of our atmosphere.
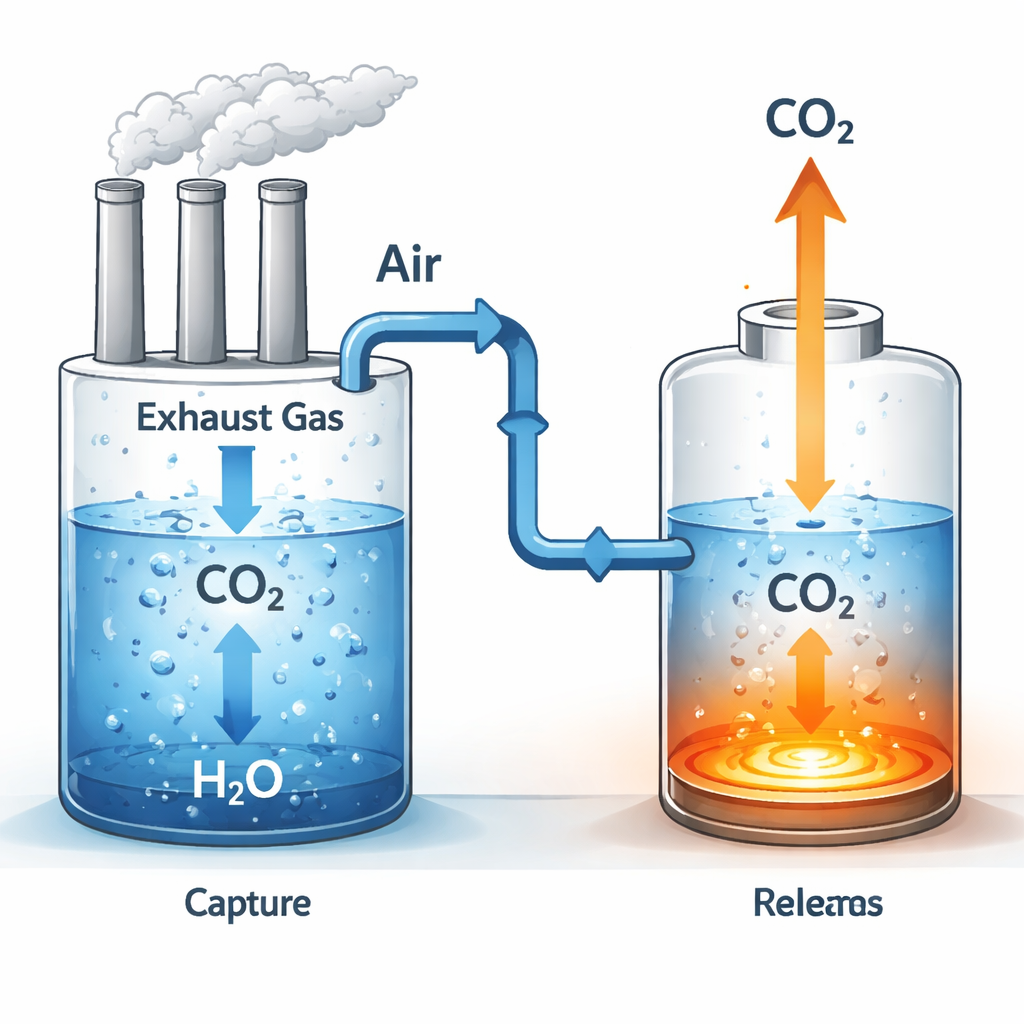
A fresh look at a familiar climate problem
The paper begins by revisiting how much our dependence on fossil fuels has altered the air. Even if countries meet their pledges to reach “net‑zero” emissions by mid‑century, the CO2 already in the atmosphere will keep warming the planet for decades. Existing capture methods often rely on synthetic chemicals called amines or on exotic materials and membranes. These approaches can work, but they tend to be expensive, energy‑hungry, and sometimes toxic, which limits how widely they can be used. The authors argue that to make a real dent in global CO2 levels, we need a capture method that is cheap, robust, and based on materials that are abundant and safe enough to deploy almost anywhere.
Letting water do what it naturally does
CO2 is unusual among common gases because it dissolves readily in water, while gases like oxygen, nitrogen, hydrogen, and methane barely do. The team set up simple experiments to show how powerful this effect can be. When a plastic drink bottle was partly filled with water and CO2 and then shaken, the CO2 rushed into the water so quickly that the gas volume dropped and the bottle visibly collapsed within seconds. Syringe experiments confirmed that much of the CO2 vanished from the gas space and ended up dissolved in the water, whereas hydrogen and methane hardly entered the water at all. The researchers also showed that cooler temperatures and higher pressures allowed more CO2 to be absorbed, while warming the water caused the gas to escape again.
Turning dissolved gas into a useful stream
Simply dissolving CO2 is only the first step. The authors designed a two‑stage system that uses pressure, temperature, and movement to cycle CO2 in and out of water in a controlled loop. In the first stage, exhaust gas from a factory or power plant is bubbled through cooled, circulating water under pressure. CO2 selectively enters the water, leaving behind a cleaner gas mixture with much less CO2. In the second stage, the CO2‑rich water is sent to a warm, low‑pressure chamber where agitation and heat drive the gas back out, creating a concentrated CO2 stream that can be handled, used, or stored. The same water is then cooled and pumped back to the first chamber to repeat the process, avoiding the need for large quantities of added chemicals.
From simple bottles to a smart capture plant
Building on these tests, the authors sketch a “smart” capture and release plant that could treat large flows of gas. Coiled pipes and spray or mixing systems increase the contact area between gas and water, boosting the rate at which CO2 is taken up. In some versions, a small amount of sodium hydroxide (NaOH) is added to the circulating water. This substance reacts with dissolved CO2 to form stable carbonate salts, locking the gas in place and preventing it from slipping back into the air until deliberately released with an acid. Because NaOH and related salts can be produced from common table salt and electricity, the whole system could be integrated with renewable power and existing industrial streams, and would avoid many of the safety issues linked to amine‑based systems.

What this could mean for climate and life
The study concludes that water’s everyday behavior—its ability to soak up CO2 at low temperature and pressure, and let it go again when warmed—can be engineered into a flexible tool for climate action. A well‑designed water‑based plant could strip CO2 from exhaust gases or even from the open air, using widely available materials and relatively modest energy inputs. While further scale‑up and testing are needed, the work suggests that an important part of our climate solution might rest not on exotic chemistry, but on harnessing a simple property that has shaped life on Earth for billions of years.
Citation: Sorimachi, K., Tsukada, T. Smart CO2 capture and release and an insight into biological evolution based on its characteristics. Sci Rep 16, 7392 (2026). https://doi.org/10.1038/s41598-026-39494-4
Keywords: carbon dioxide capture, water-based CO2 removal, climate change, carbon cycle, carbon storage