Clear Sky Science · en
fln-2 isoform-specifically regulates Caenorhabditis elegans health span by affecting pharyngeal function
Why Mouthparts Matter for Living Longer
We usually think of aging as something that happens deep inside our cells, but this study shows that the way an animal chews its food—and the microbes that come with it—can also shape how long it stays healthy. Using the tiny roundworm Caenorhabditis elegans, a classic model for aging research, the authors discovered that specific versions of a single structural gene can tune how well the worm’s “mouth” grinds bacteria. Better grinding means fewer bacterial infections, a sturdier gut, and ultimately a longer, healthier life, all without eating less.
A Gene with Many Faces
The work centers on a gene called fln-2, the worm counterpart of human filamin A, a protein that helps give cells their shape and links them to their surroundings. The fln-2 gene is unusually complex: it can produce up to 27 slightly different protein forms, or isoforms, from the same stretch of DNA. Earlier studies had hinted that a naturally occurring mutation in fln-2 could extend worm lifespan by reducing deadly infections in the feeding organ, the pharynx. However, it was unclear which of the many isoforms mattered, and why losing them might sometimes shorten lifespan instead of lengthening it.
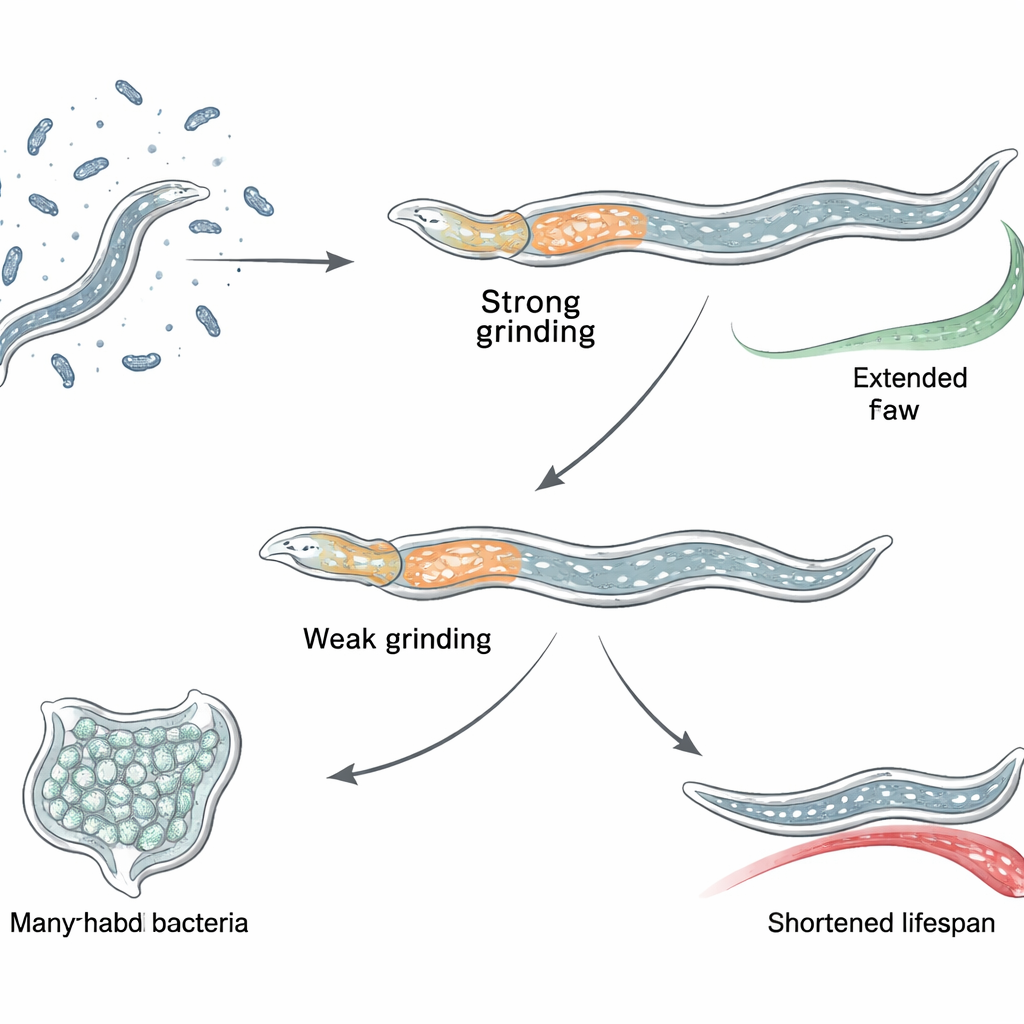
Long Versions Help, Short Versions Hurt When Lost
Using precise genome editing, the researchers created a panel of worm strains in which selected groups of fln-2 isoforms were disabled. They found a striking pattern: when only the longest isoforms were disrupted, the worms lived longer and stayed healthier; but when all isoforms were removed, including shorter ones expressed more broadly in the body, lifespan actually shrank. Imaging showed that the long isoforms are concentrated along the inner surface of the pharynx, especially in a toothed structure called the grinder. This layout suggests that different isoforms play opposing roles: losing the pharynx-focused long versions boosts survival, whereas losing widely used shorter versions causes harmful side effects that overwhelm any benefit.
Sharper Grinding, Fewer Germs, Stronger Guts
The team then asked how missing the long isoforms could make worms more robust. They showed that worms lacking these isoforms grind their bacterial food more thoroughly, leaving fewer intact microbes to pass into the intestine. With age, normal worms accumulate living bacteria in the gut and can develop widespread infection in the pharynx, both of which limit lifespan. In contrast, fln-2 long-isoform mutants had much less bacterial build‑up in both regions. Microscopy with fluorescent bacteria and RNA staining confirmed substantially lower colonization. These worms also maintained a tighter intestinal barrier, as tested with a dye that leaks into the body cavity when the gut wall is damaged. This protection depended on an internal recycling process called autophagy: if key autophagy genes were disabled, the lifespan extension and barrier benefits disappeared.
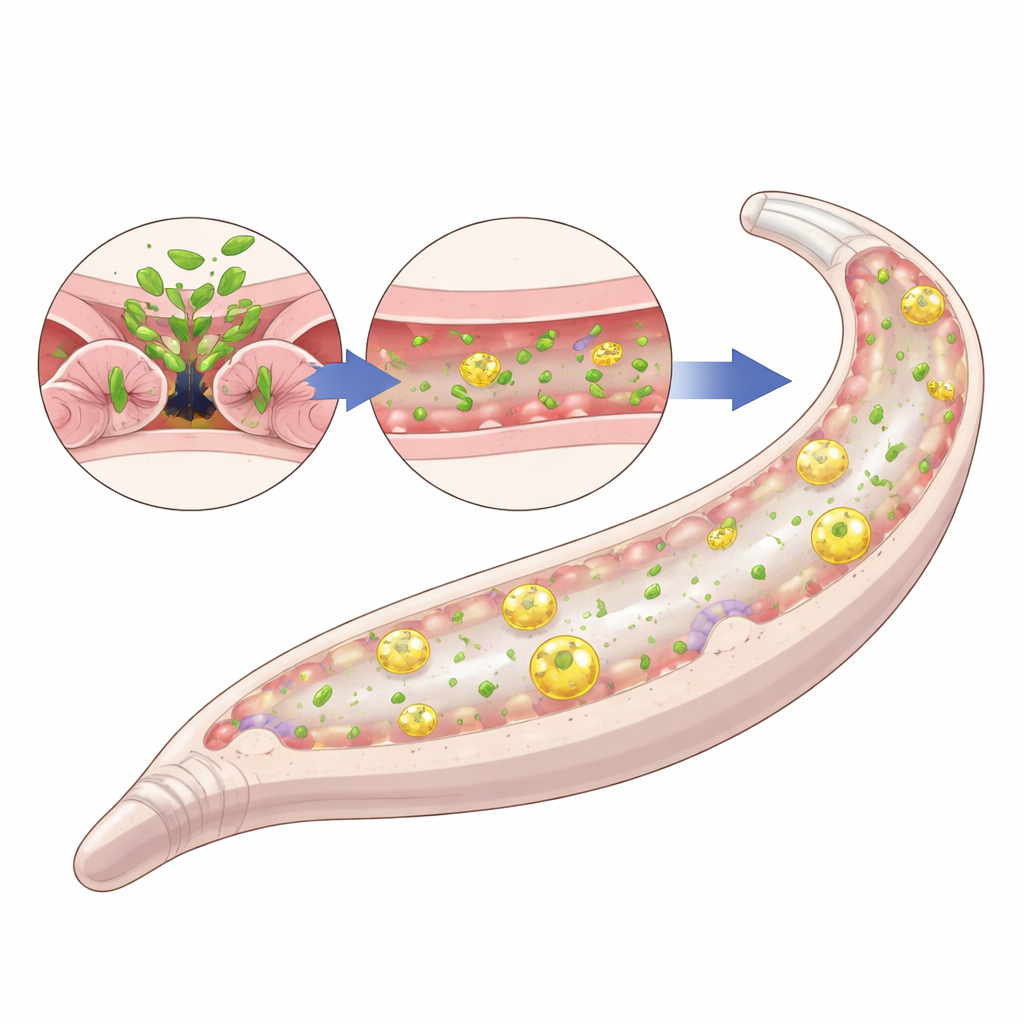
Not Just Eating Less: The Role of Live Bacteria
Many long-lived worm mutants work by eating fewer calories, but that was not the case here. The fln-2 mutants pumped food through the pharynx at normal rates and consumed similar amounts of bacteria as standard worms. Genetic tests showed that their longevity did not rely on well-known hormone-like aging pathways either. Instead, the benefits vanished when worms were fed heat‑killed bacteria. Without live microbes to infect them, fln-2 mutants no longer lived longer than normal worms, even though both groups showed high levels of autophagy. This indicates that the key advantage of losing the long isoforms is context‑dependent: it only pays off when worms face the constant challenge of living, replicating bacteria.
What This Means for Aging and Infection
In simple terms, this study reveals a tug‑of‑war within one gene between different protein versions. Removing the long versions fine‑tunes the worm’s mouthparts so they crush bacteria more effectively, lowering infection pressure and triggering protective cleanup inside gut cells. But removing every version of the gene destabilizes vital tissues and shortens life. Together, the findings highlight how aging can be steered not only by deep molecular switches but also by the physical design of organs that interact with microbes. While worms are far from humans, the work suggests that subtle changes in structural proteins related to filamin A—and in how our bodies manage the constant flow of microbes through the digestive tract—could play a broader role in determining how long and how well we live.
Citation: Chang, YH., Chi, AQ., Ren, YC. et al. fln-2 isoform-specifically regulates Caenorhabditis elegans health span by affecting pharyngeal function. Sci Rep 16, 8363 (2026). https://doi.org/10.1038/s41598-026-39461-z
Keywords: aging, C. elegans, gut microbiota, autophagy, pharyngeal function